Patents
Literature
1467 results about "Reaction timing" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Method of growing oxide films
InactiveUS20030188682A1Easy to prepareExcellent dielectric propertiesPolycrystalline material growthSemiconductor/solid-state device manufacturingSilicon oxideOxygen
Abstract of Disclosure Process for producing silicon oxide containing thin films on a growth substrate by the ALCVD method. In the process, a vaporisable silicon compound is bonded to the growth substrate, and the bonded silicon compound is converted to silicon dioxide. The invention comprises using a silicon compound which contains at least one organic ligand and the bonded silicon compound is converted to silicon dioxide by contacting it with a vaporised, reactive oxygen source, in particular with ozone. The present invention provides a controlled process for growing controlling thin films containing SiO2, with sufficiently short reaction times.
Owner:ASM IP HLDG BV
Automatic playback overshoot correction system
InactiveUS6850691B1Television system detailsElectronic editing digitised analogue information signalsComputer scienceUser expectations
An automatic playback overshoot correction system predicts the position in the program material where the user expects to be when the user stops the fast forward or reverse progression of the program material. The invention determines the position where the program material was stopped. The media controller transitions to the new mode that the user selected, starting at the stopped position with an overshoot correction factor added or subtracted from it. The invention adapts to the user by remembering how much the user corrects after he stops the fast forward or reverse mode. Correction factors are calculated using the user's corrections and adjusting the correction factors if the user continues to make corrections. The invention also uses a prediction method to correctly place the user within the program upon transition out of either mode and determines if the speed of the fast forward or reverse modes and then automatically subtracts or adds, respectively, a time multiple to the frame where the transition was detected and positions the user at the correct frame. The time multiple is fine tuned if the user is consistently correcting after the fast forward or rewind mode stops. Another method initially tests the user's reaction time using a test video and asks the user to press the fast forward or reverse button on his control device during the test video and then asks the user to position the video to the place that he expected the system to have been. This time span is then used whenever the user uses the fast forward or reverse modes and is adjusted with a multiple for each speed. A final method allows the user to simply set a sensitivity setting that the system will use as a correction factor and a multiple is subtracted or added to the release frame whenever the user uses the fast forward or reverse modes, respectively.
Owner:TIVO SOLUTIONS INC
Dissolvable downhole tool, method of making and using
Disclosed herein is a dissolvable downhole tool. The tool includes, a dissolvable body constructed of at least two materials and at least one of the at least two materials is a reactive material, and a first material of the at least two materials being configured to substantially dissolve the dissolvable body and a second material configured to control reaction timing of the first material.
Owner:BAKER HUGHES INC
Method of producing ethyl acetate
InactiveUS20090005588A1Good dispersionWell mixedOrganic compound preparationRotary stirring mixersAcetic acidReaction temperature
Methods and systems for the production of ethyl acetate are described herein. The methods and systems incorporate the novel use of a high shear device to promote dispersion and mixing of a carbonyl co-reactant (e.g. acetic acid, acetaldehyde) with ethanol. The high shear device may allow for lower reaction temperatures and pressures and may also reduce reaction time with existing catalysts.
Owner:HRD CORP
Control device for automatic liquid handling system
InactiveUS7390460B2Bioreactor/fermenter combinationsAnalysis using chemical indicatorsTime segmentEngineering
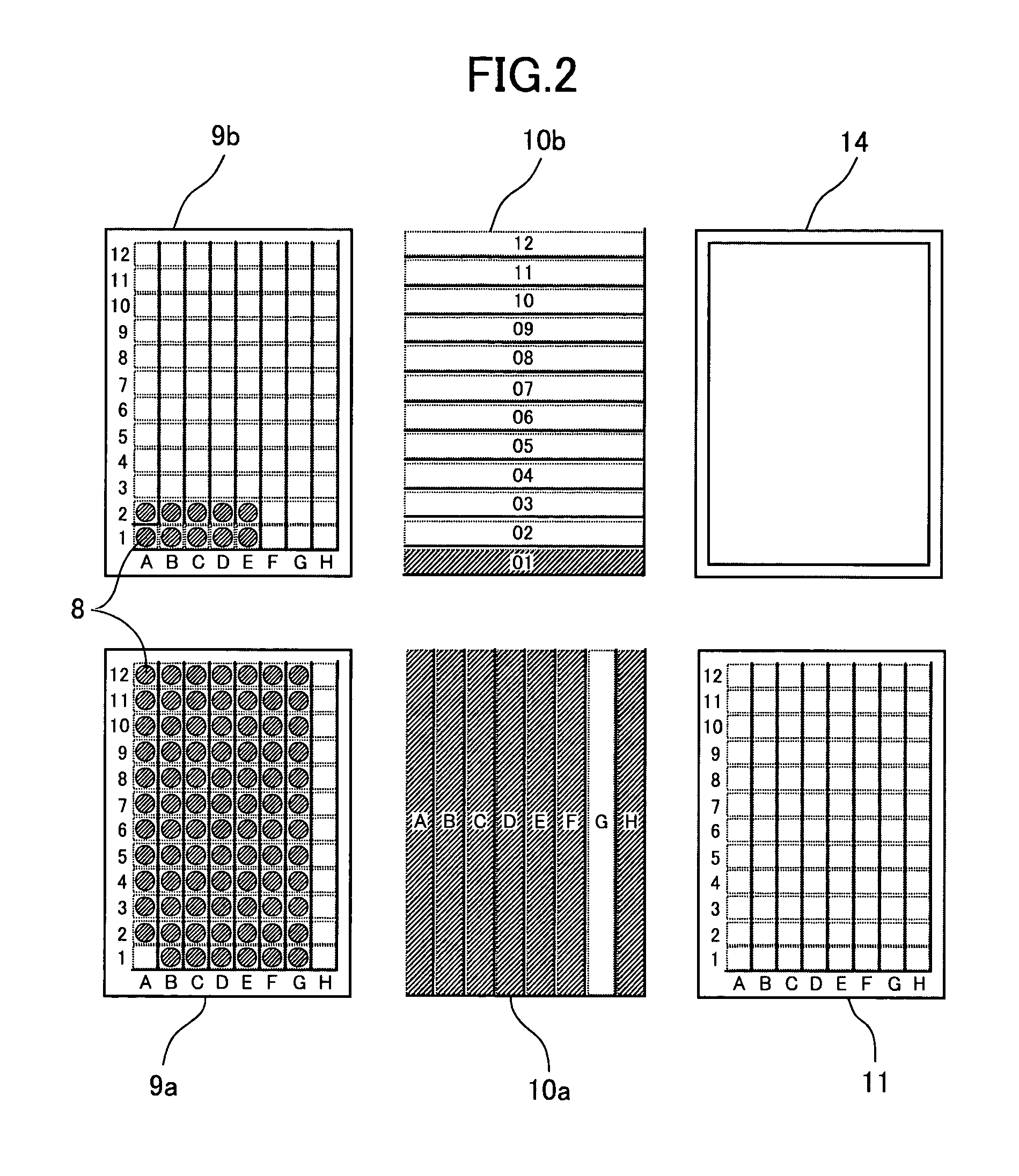
In reagent reaction experiments using microplates, a desired period of time can be set for determining the time to introduce a reaction stop solution after introduction of a reagent. The set time is measured starting from the introduction of the reagent and the reaction stop solution is introduced immediately after expiration of the set time. A control device for an automatic liquid handling system having the above-described reaction time managing feature is also provided with a self-diagnosing function that is capable of determining whether scheduled processes are executable or not during the time set before execution of the processes and informing an operator of the results of determination.
Owner:HITACHI KOKI CO LTD
Method of growing oxide thin films
InactiveUS20040065253A1Easy to prepareExcellent dielectric propertiesPolycrystalline material growthSemiconductor/solid-state device manufacturingSilicon oxideOxygen
Process for producing silicon oxide containing thin films on a growth substrate by the ALCVD method. In the process, a vaporisable silicon compound is bonded to the growth substrate, and the bonded silicon compound is converted to silicon dioxide. The invention comprises using a silicon compound which contains at least one organic ligand and the bonded silicon compound is converted to silicon dioxide by contacting it with a vaporised, reactive oxygen source, in particular with ozone. The present invention provides a controlled process for growing controlling thin films containing SiO2, with sufficiently short reaction times.
Owner:ASM IP HLDG BV
Dissolvable downhole tool, method of making and using
Disclosed herein is a dissolvable downhole tool. The tool includes, a dissolvable body constructed of at least two materials and at least one of the at least two materials is a reactive material, and a first material of the at least two materials being configured to substantially dissolve the dissolvable body and a second material configured to control reaction timing of the first material.
Owner:BAKER HUGHES INC
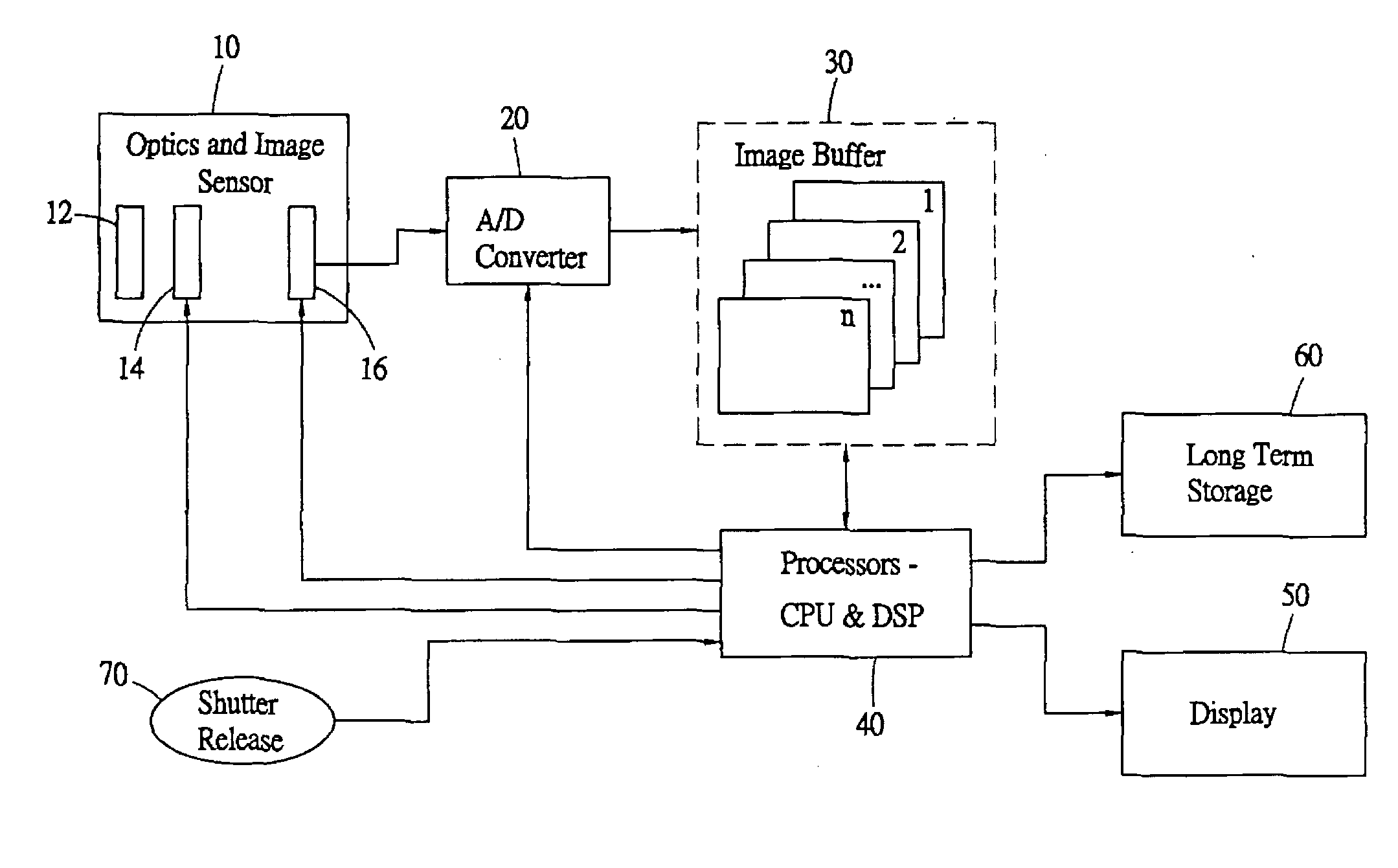
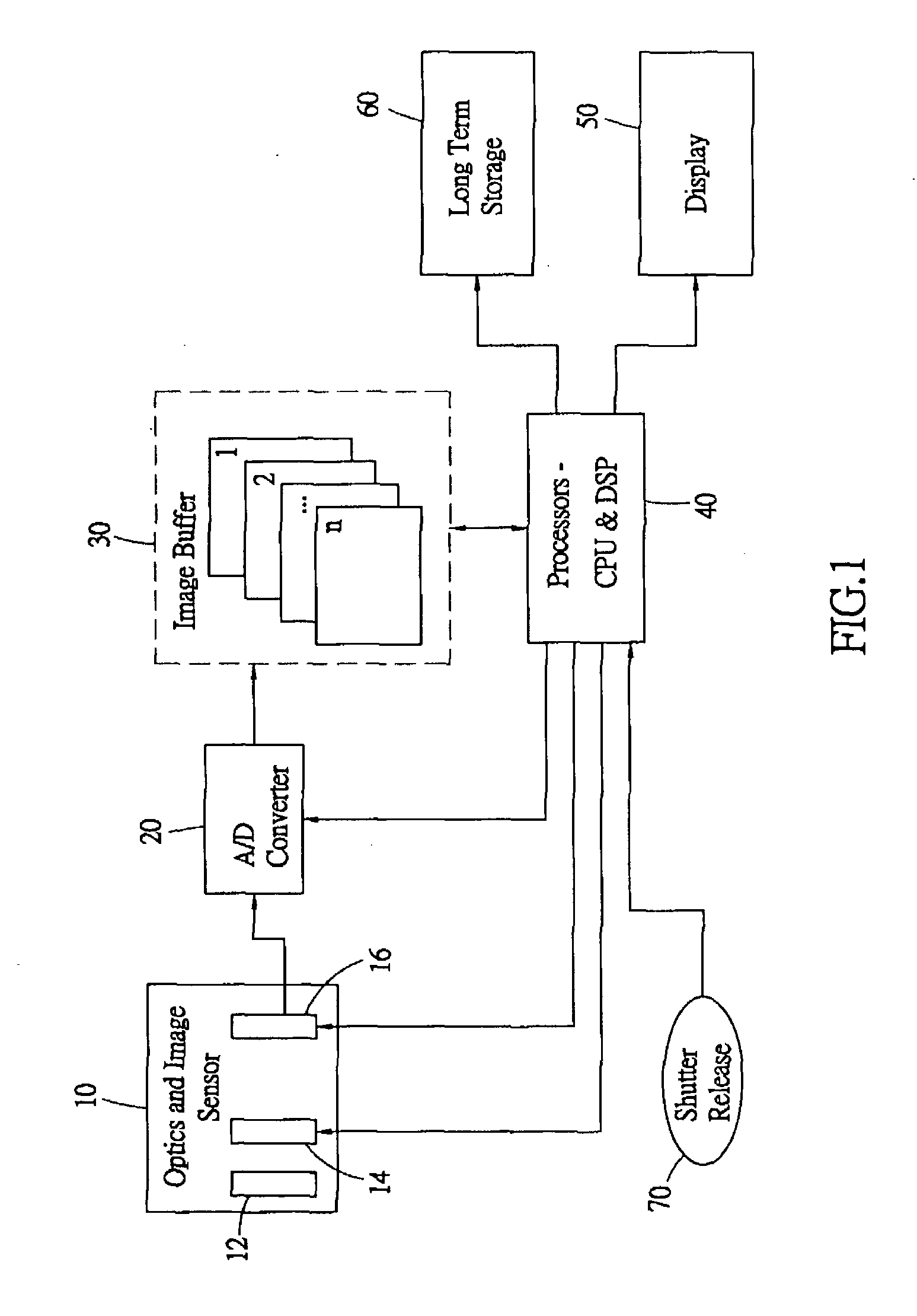
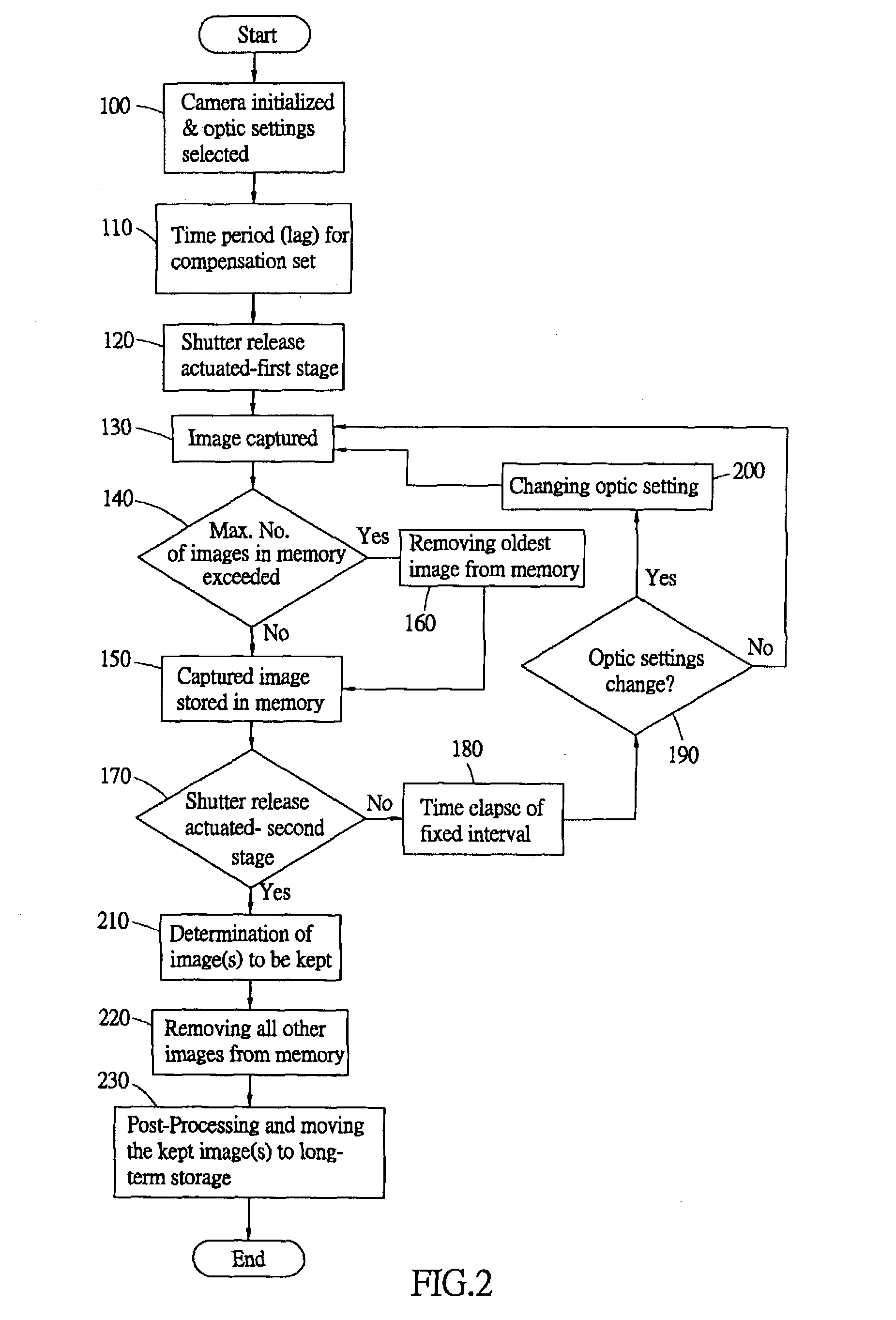
Compensating for delays inherent in digital still camera imaging
The delays in digital still cameras due to the lag between the press of the shutter release and the shutter opening and due to the reaction time of the photographer are compensated for by continuously capturing and storing images and by selecting a desired one of the images based on the lags. Upon processing an indication from the photographer, the camera begins capturing images at regular images and stores the most recent images in a first-in first-out buffer. At a second indication from the photographer, the camera selects, in accordance with the lags, the image in the buffer a fixed amount of time prior to the second indication for further processing. In this way, both the delay due to human reaction time and the delay due to shutter lag have been compensated for.
Owner:NUCAM CORP
Particle appearance regulatory lithium iron phosphate preparation method
ActiveCN101007630AGood for solid phase diffusionUniform size distributionCell electrodesPhosphorus compoundsLithium iron phosphatePhosphate ion
The invention discloses a modulating method of LiFePO4 particle shape, which comprises the following steps: blending one or more composition with lithium ion, ferric ion and phosphate radical ion; adding solvent and certain quantity of crystal growing inhibitor; proceeding solvent heat reaction under certain temperature; washing; filtering; drying; sintering to improve crystallizing property; obtaining the product with regular shape and size.
Owner:QINGHAI TAIFENG XIANXING LITHIUM ENERGY TECH CO LTD
Process for manufacturing 3-hydroxy-3-methylbutanoic acid
InactiveUS6090978ABatch size can be increasedImprove product qualityOrganic compound preparationChemical/physical/physico-chemical stationary reactors3-methylbutyric acidReagent
A method for manufacturing 3-hydroxy-3-methylbutanoic acid (HMB) in commercially viable amounts is disclosed. The reaction mixture cycles through an external heat exchanger while the primary reactant is added in a warmer so as to control and maintain a low temperature for the reaction mixture. The manufacturing process herein disclosed increases yield while decreasing reaction time from the synthetic processes currently practiced.
Owner:MET RX USA
Multiple extruder assembly and process for continuous reactive extrusion
Methods are disclosed for a novel and useful single pass extrusion process for the reactive extrusion and compounding of polymers. Traditional extruders utilized in reactive processes are of length to diameter ratios ranging from 30 to 1 to as high as 56 to 1. The process disclosed uses a series of sequential, very closely-coupled, independently driven screw extruders having a total effective length to diameter ratio much greater than 70 to 1 and as high as 132 to 1 or greater, and providing greatly extended reaction times, separate and multiple introductions of reactive and non-reactive agents and mechanical connections allowing for convenient screw changes and differential thermal expansion. The assembly is employed to economically produce grafted polyolefins, produce ionomers without employing the use of strong caustic agents, remove large volumes of unwanted polymer processing solvents and produce other reacted polymer species in one continuous pass.
Owner:ORREX PLASTICS CO LLC
Nano lithium iron phosphate material and preparation method thereof
InactiveCN101546830ASolve the real problemSmall particlesElectrode manufacturing processesChemical/physical/physico-chemical processesChemical compositionLithium iron phosphate
The invention relates to a nano lithium iron phosphate material and a preparation method thereof. A lithium source, an iron source, a phosphate radical source and a doped element compound are dissolved in water solution containing a complexing agent according to a stoichimetric proportion and added with high-conductive carbon nano tubes scattered by assistant to form a cladding material, the obtained solution is sintered in an inert atmosphere furnace, the reaction temperature is between 500 and 900 DEG C, and the reaction time is 3 to 16 hours. The method effectively controls the chemical components, phase components and grain diameters of LiFePO4; and the obtained lithium iron phosphate is the nano lithium iron phosphate clad by the carbon nano tubes, has fine and even grains with high purity, high charge / discharge capacity, good multiplying power performance and good cycle performance, and is an ideal material for manufacturing lithium ion batteries.
Owner:SHENZHEN DYNANONIC
Method for producing cross-linked hyaluronic acid
A method for producing a cross-linked hyaluronic acid in accordance with the present invention comprises: (a) cross-linking at least one polymer at a temperature, from about 35° C. to about 60° C., for a reaction time of from about 0.1 hour to about 72 hours with a cross-linking agent; and (b) lowering the temperature in step (a) to form about 10° C. to about 30° C. for a reaction time of from about 48 hours to about 28 days to obtain the cross-linked hyaluronic acid, whereby, a cross-linking agent content in a product of the method can be decreased so the product does not require purification.
Owner:SCIVISION BIOTECH
Continuous process and apparatus for preparing inorganic materials employing microwave
This invention relates to a continuous microwave synthesis process of inorganic materials and its apparatus and more particularly, to the process of synthesizing inorganic materials prepared in a manner such that after preparing a mixed solution of precursor materials for various inorganic materials such as porous molecular sieve, layered compounds and ceramics, this mixed solution is continuously added to a tube-type microwave reactor using a slurry pump for the synthesis and crystallization of inorganic materials. Thus the manufacturing process of this invention has the following advantages: (1) the reaction time is further shortened by several to tens of minutes for crystallization, compared to the conventional hydrothermal reaction requiring a prolonged time, (2) the continuous manufacturing and collection processes of this invention can give access to mass-scale production of inorganic materials with relatively small facility, compared to the conventional batch hydrothermal or microwave synthesis, and (3) less amount of organic templating agent can be required during the manufacture of porous molecular sieve.
Owner:KOREA RES INST OF CHEM TECH
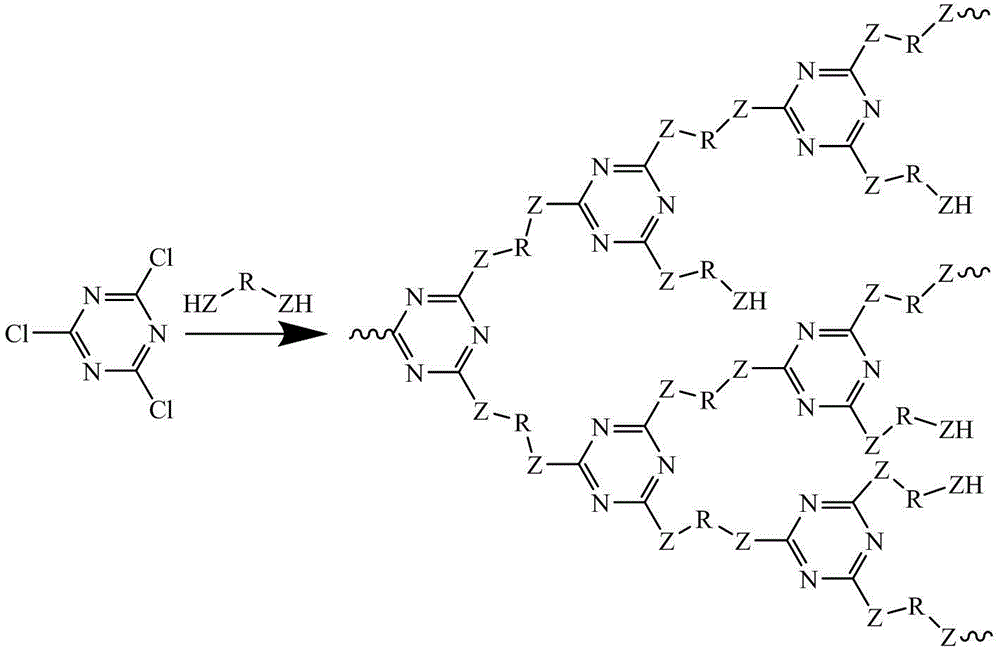
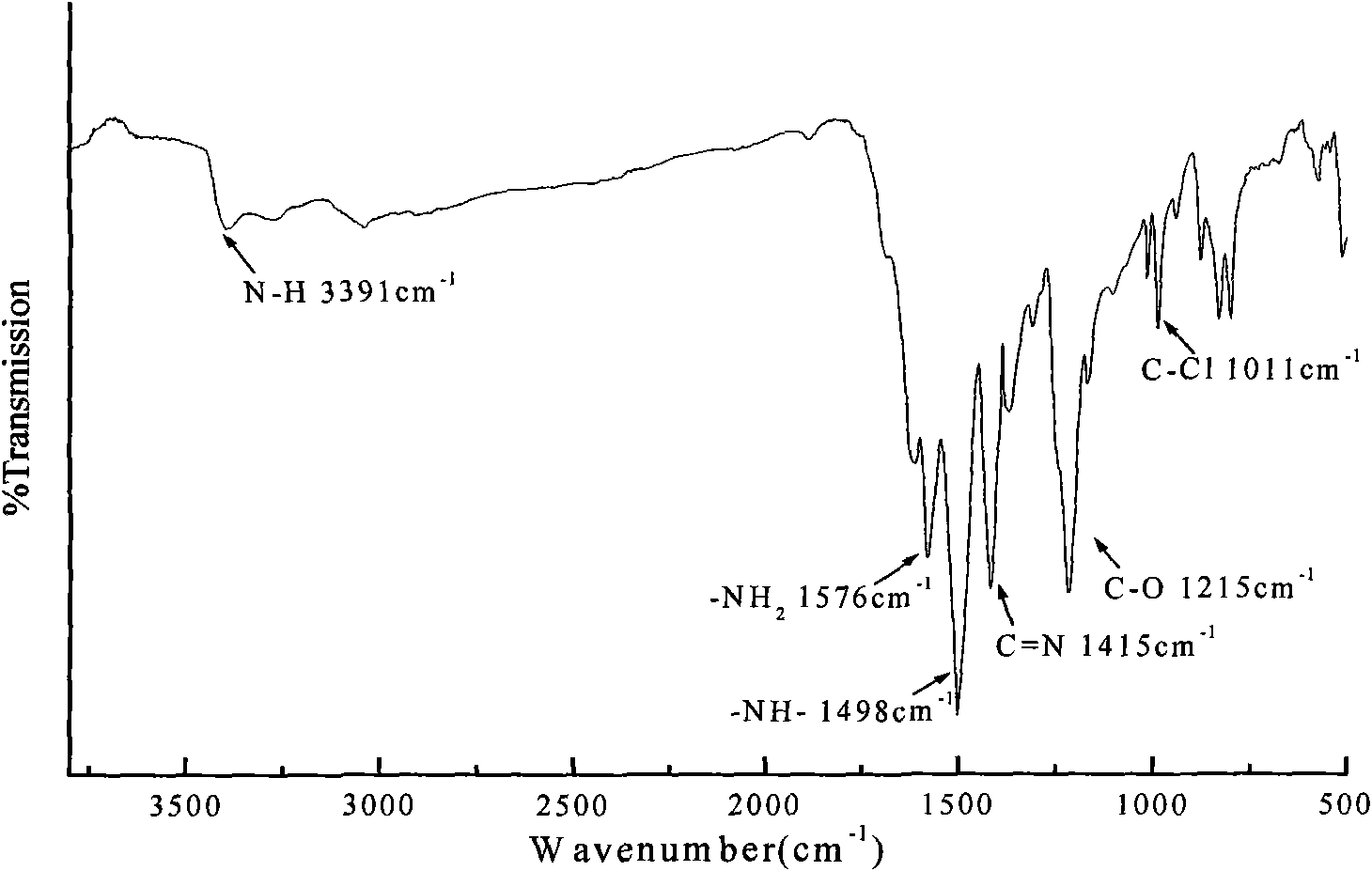
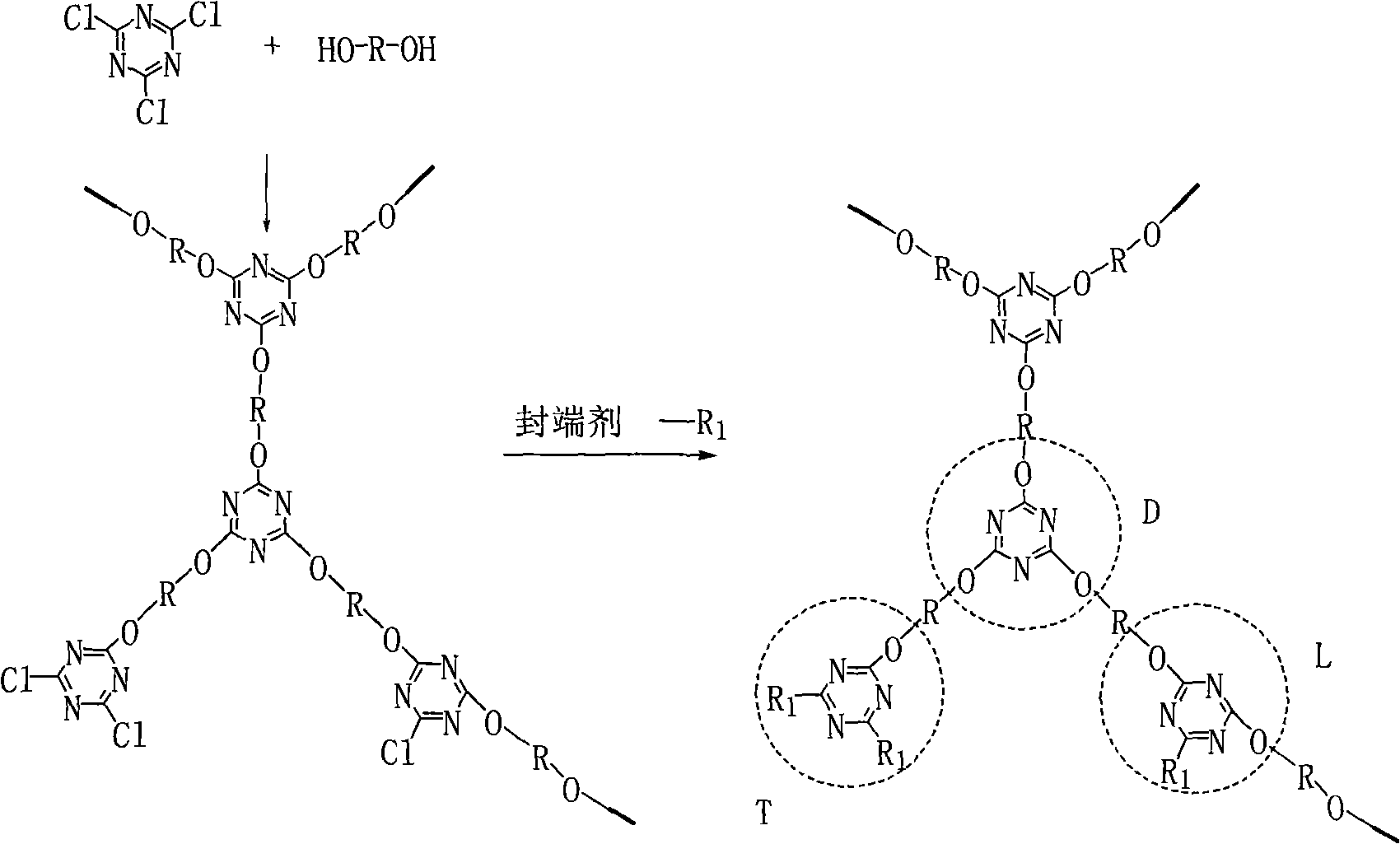
Preparation method of novel hyperbranched triazine char forming agent
The invention discloses a preparation method of a novel hyperbranched triazine char forming agent, which comprises the following steps: adding a dissolved diamine solution at 0-10 DEG C, gradually and dropwisely adding a dispersed cyanuric chloride solution, and reacting for 1-4 hours; heating to 30-70 DEG C, adding an acid-binding agent, and reacting for 3-8 hours; heating to 90-150 DEG C, and reacting for 6-10 hours; and stopping stirring, carrying out vacuum filtration, washing with water, and drying to obtain the triazine hyperbranched char forming agent. Compared with the prior art, the invention has the advantages of continuous reaction process, short reaction time, cheap raw materials, low solvent consumption, no addition of any blocking agent in the reaction process, simplified synthesis technique, high heat stability and high char forming ratio.
Owner:合肥安聚达新材料科技有限公司
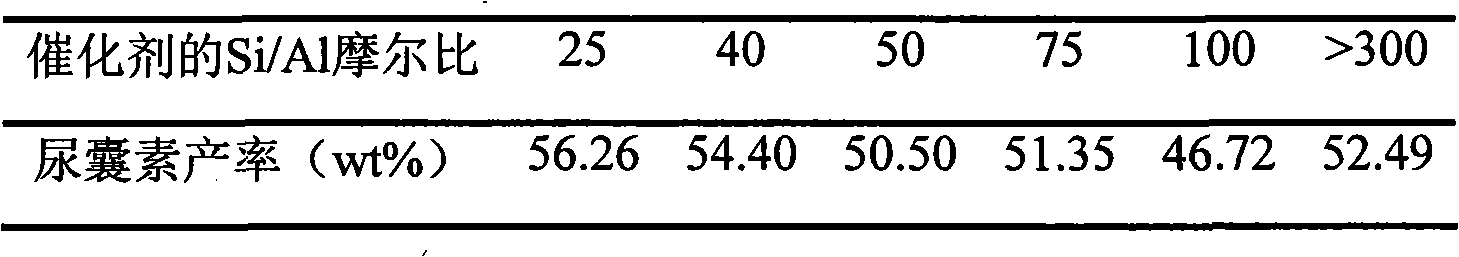
Solid acid catalyst and reaction technique for synthesis of allantoin
InactiveCN101347739AHigh activityImprove stabilityOrganic chemistryMolecular sieve catalystsRare-earth elementSolid acid
The invention relates to a catalyst for synthesis of allantoin by the reaction of glyoxylic acid and urea and a corresponding reaction process. The catalyst is the solid acid catalyst and takes one or the combination of more of oxides and molecular sieves of IVB to VIB elements as a carrier, or a complex which is composed of one or the combination of more of the oxides and the molecular sieves of IVB to VIB elements and one or the combination of more of the oxides of IB, IIB, VIIB, VIIIB and rare earth element as the carrier, and the carrier is carried out the impregnation supporting, the drying and the calcination by using inorganic strong acid or ammonium salt solution thereof for the preparation. The use of the catalyst can synthesize an allantoin product by one-step reaction of the glyoxylic acid and the urea in an atmospheric pressure tank reactor. The catalyst of the invention has the advantages of high activity and stability, recyclable property, no corrosion of the device and environmental pollution, etc. The synthesis reaction process of the allantoin has the advantages of simple process, less using amount of the catalyst, short reaction time and high yield of the allantoin, etc.
Owner:HUNAN UNIV
Preparation method of temperature sensitivity chitosan derivate-hydroxybutyl chitosan
The invention relates to a method for making a chitosan derivative. The method is characterized in that: firstly, the purified chitosan is activated by a strong base to form a hydroxyl-K type chitosan; secondly, a 1, 2-butyl oxide linkage is taken as an etherifying agent, an isopropanol is taken as a disperse phase, the reaction lasts 2 to 4 days moderately at room temperature; after the reaction is finished, 8 to 10 times volume of acetone is precipitated and washed to be neutral as well as is dried in vacuum at a temperature of 50 DEG C to obtain the white powder. The hydroxybutyl chitosan with different degrees of substitution is obtained by controlling the proportion of the chitosan and the 1, 2-butyl oxide linkage and the reaction time. The method has the advantages of good water-solubility and the existence of the temperature sensitivity at a physiological temperature.
Owner:SHANGHAI QISHENG BIOLOGICAL PREPARATION CO LTD
Covalent organic framework material and preparation method thereof
ActiveCN108219158AReduce energy consumptionEasy to operateGas treatmentOther chemical processesRoom temperatureBoric acid
The invention discloses a covalent organic framework material and a preparation method thereof and belongs to the technical field of preparation of covalent organic framework materials. Multiple covalent organic framework materials are quickly synthesized under a room temperature and an open system by utilizing ionothermal reaction, and include a Schiff-base type covalent organic framework material and a boric-acid type covalent organic framework material, and also include covalent organic framework materials with two-dimensional and three-dimensional structures. The synthetic method has the advantages of short reaction time, low energy consumption, no pollution of volatile organic matters, no additive, simple operation and the like. Ironic liquid can be reused for at least three times after being simply filtered and separated. The covalent organic framework material synthesized by the method has potential application in the aspects of gas storage and separation and the like because ofloading the ironic liquid.
Owner:JILIN UNIV
Catalyst for wet oxidation technique and preparation method thereof
InactiveCN101185887AHigh catalytic activityImprove stabilityMetal/metal-oxides/metal-hydroxide catalystsWater/sewage treatment by oxidationActive componentSalt solution
The invention relates to a catalyst which is used for wet air oxidation technique and the preparation method in the field of environmental protection technology. The catalyst uses Gamma-Al2O3 as a vector and the oxide of transition metal Mn or Mn and rear earth element M or the oxide of the M as active components, wherein, M is one or two kinds in La and Ce and the loading of each metal relative to Gamma-Al2O3 vector is 5wt to 8wt percent. The catalyst takes Gamma-Al2O3 as the vector, is cleaned and roasted to be a consistent weight; the obtained Gamma-Al2O3 vector is immersed into the metallic salt solution of the rear earth element M and the transition metal Mn; the loading of each metal relative to the vector is 5wt to 8wt percent; the mixture after being immersed and dried is roasted in air to get the catalyst used in the wet air oxidation. The invention does not use expensive precious metal materials and greatly reduces the cost of the catalyst; at the same time, the catalytic activity of the catalyst is enhanced; the operation condition is improved; and the reaction time is shortened.
Owner:SHANGHAI JIAO TONG UNIV
Catalytic combustion system and method
InactiveUS7096671B2Emission reductionShorten flame lengthContinuous combustion chamberTurbine/propulsion fuel supply systemsNitrogenNitrogen oxide
Aspects according to the invention relate to a catalytic combustor system for a turbine engine and an associated method. Catalytic combustors are used in connection with turbine engines because they can minimize the formation of oxides of nitrogen during combustion. Despite this emissions advantage, catalytic combustion systems can increase the level of CO in the turbine exhaust. According to aspects of the invention, vortex formation devices includes vortex generators, swirlers and mixers can be placed downstream of each catalytic module surrounding the pilot nozzle so as to form one or more vortices in the otherwise substantially laminar flow exiting the modules. The vortices can create a suction so that a portion of the flow exiting the pilot nozzles is mixed with the flow exiting the catalyst modules. The introduction of the higher temperature pilot flow can accelerate the catalytic reaction time, promoting burnout of the CO formed during combustion.
Owner:SIEMENS ENERGY INC
Three-phase ultrasound light-catalyzed reaction device and method for CO2 reduction thereof
InactiveCN101138700AImprove photocatalytic efficiencyShort reaction timeDispersed particle separationEnergy based chemical/physical/physico-chemical processesWater bathsUltrasound - action
The present invention discloses a three-phase ultrasonic photo-catalyzed reaction device and a CO2 reducing method with the device. With the device, a photo-catalyst is placed in a photo-reactor, and the reactor is placed in a water bath of an ultrasonic generator. A light source is arranged above the photo-reactor. The method is that the CO2 gas to be reduced is mixed evenly with the photo-catalyst and a sodium hydroxide solution in the photo-reactor under the ultrasonic action, and reacts for 3-20 hours with radiation of the light source. Then the CO2 gas is reduced. The photo-catalyst is made from titanium dioxide or silicon dioxide and metal phthalocyanine. Metal phthalocyanine is supported on the surface of titanium dioxide in the way of in-situ synthesis, sol-gel, and immersion. The present invention promotes the photo-catalysis efficiency, reduces the reaction time, and saves the energy. Visible light can be used for reducing. Moreover, the phot-catalyst can be recycled, which reduces the pollution and the cost, sot the present invention is quite applicable for the three-phase ultrasonic photo-catalyzed reaction device and photo-catalyzed CO2 reducing.
Owner:TAIYUAN UNIV OF TECH
Method for hydrolyzing lignocellulose
The invention discloses a method for hydrolyzing lignocellulose. Lignocellulose is pulverized into particles, and the particles are mixed with water to form even seriflux; the seriflux is preheated to60-150 DEG C by using ultrasonic waves or microwaves; the preheated even seriflux is quickly heated to 120-300 DEG C, solid acid is added and used as a catalyst, and stirring and catalytic reaction are simultaneously carried out for 15-300 minutes. The use of the ultrasonic waves or microwaves radiation in synergism with solid alkali for catalyzing and hydrolyzing the lignocellulose can greatly reduce the reaction time as well as energy consumption. The solid acid catalyst can be repeatedly used, is easily separated, and has the advantages of high mechanical strength, high durability, certainselectivity, relative singularity of catalytic hydrolyzate, high hydrolyzing efficiency and good market application prospect.
Owner:XISHUANGBANNA TROPICAL BOTANICAL GARDEN CHINESE ACAD OF SCI
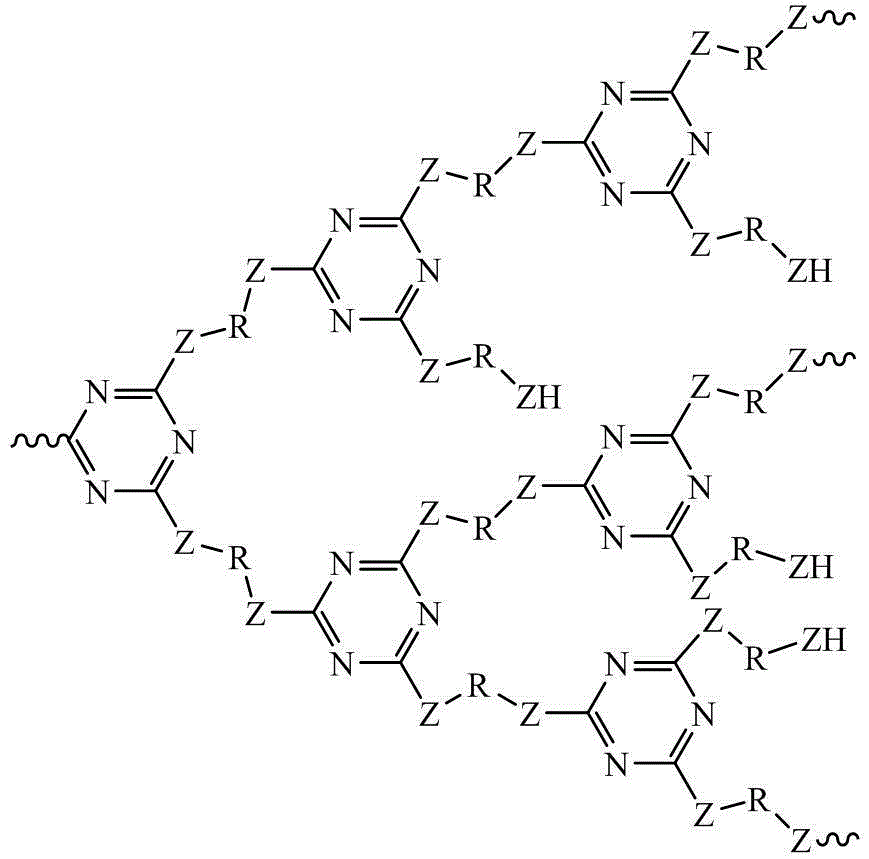
Preparation method for triazine hyperbranched macromolecular carbon forming agent
The invention discloses a preparation method for a triazine hyperbranched macromolecular carbon forming agent. The method is characterized by comprising the following steps: 1), mixing diamino or hydroxyl compound with non-protonic organic solvent, and adding catalyst in the atmosphere of inert gases; 2), dropwise adding cyanuric chloride non-protonic organic solution, and continuing the reaction at the temperature after finishing dropwise adding the cyanuric chloride non-protonic organic solution; 3), adding blocking agent, and carrying out reaction again; and 4), precipitating, filtering, cleaning the organic solvent, separating, and drying to obtain the triazine hyperbranched macromolecular carbon forming agent. Compared with the prior art, the invention has the advantages that the 'one-pot' method is adopted, the reaction process is continuous, the reaction time is short, the product has favorable heat stability, the carbon forming rate is high, and the processability is good.
Owner:NINGBO INST OF MATERIALS TECH & ENG CHINESE ACADEMY OF SCI
Method for synthesizing methylethyl carbonate
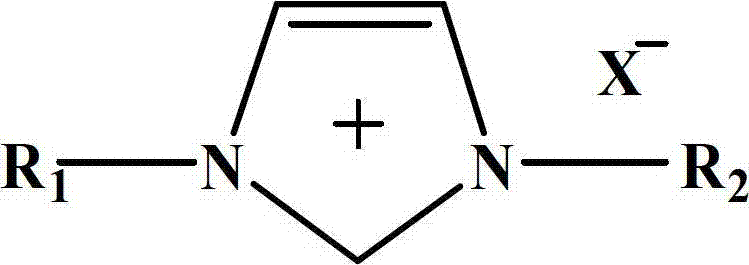
ActiveCN102850223AHigh catalytic activityLow costOrganic-compounds/hydrides/coordination-complexes catalystsChemical recyclingMethyl carbonateReaction temperature
The invention relates to a method for synthesizing methylethyl carbonate. According to the method, under ordinary pressure, dimethyl carbonate and ethanol which are used as raw materials are subjected to synthetic reaction of methylethyl carbonate at certain reaction temperature for some time by using an imidazole ionic liquid as a catalyst. The invention has the following advantages: the catalyst consumption is only 0.5-5 wt% of the dimethyl carbonate; the ionic liquid catalyst has high catalytic activity in the reaction process, the maximum yield of methylethyl carbonate is up to 71.22%, and the selectivity is up to 88.52%; and the catalyst can be recycled for cyclic utilization after being subjected to simple treatment after reaction, has the advantages of long service life and no pollution, and greatly lowers the preparation cost of methylethyl carbonate.
Owner:辽阳东昌化工股份有限公司
Acid corrosion solution for preparing multicrystal silicon pile surface and its using method
ActiveCN1821446AEliminate hazardsSuitable for mass productionSemiconductor/solid-state device manufacturingPhotovoltaic energy generationCorrosion reactionHydrofluoric acid
The present invention discloses acid corrosion solution for preparing polycrystalline silicon pile surface and its usage. The solution is compounded with oxidant and hydrofluoric acid and through mixing, and the oxidant is nitrate or nitrite. During use, the cut polycrystalline silicon chip is set inside the corrosion solution for corrosion at the temperature of -10 to +25 deg.c for 0.5-20 min to eliminate damage surface caused by wire electrode cutting, with the acid corrosion time and temperature being dependent on the solution concentration. The present invention is suitable for both intermittent production and continuous production, and has easy treatment of the produced waste acid.
Owner:WUXI SUNTECH POWER CO LTD
Nano-copper conductive ink preparation method
InactiveCN101608077ASimple processMild reaction conditionsInksNon-conductive material with dispersed conductive materialDistillationSolvent
The invention belongs to the nanometer material technical field, in particular to a nano-copper conductive ink preparation method, comprising the following steps: dissolving copper salt and organic protective agents in a solvent, heating the solution to 30-100 DEG C, simultaneously adding reducing agent in the reaction system to react while stirring for 20-30min, then cooling gradually; finally obtaining stable nano-copper conductive ink through electrodialysis separation and reduced pressure distillation. The invention has simple process, mild reaction conditions and short reaction time; the conductibility of the product is good, the particle size of nano-copper is 20-70nm, the nano-copper can not be oxidized for one month in the air; the production cost is low and no hazardous waste can be generated, thus meeting the demand of 'green production'.
Owner:FUDAN UNIV
Preparation method of spinelle lithium titanate for lithium secondary battery negative electrode material
InactiveCN1919736AWide variety of sourcesFast preparationCell electrodesLithium compoundsDielectricReaction timing
The invention discloses a preparing method of spinel lithium titanate as Li-secondary battery cathode material, which adopts titanium dioxide and inorganic Li salt as raw material to low-temperature fusing salt as synthetic dielectrics to generate the product through solid-phase reaction.
Owner:BEIJING INSTITUTE OF TECHNOLOGYGY +1
Reaction system and preparation method for nitrous alkyl ester
InactiveCN101314569AHigh yieldImprove efficiencyLiquid-gas reaction processesNitrous acid preparation ester preparationRetention timeGas phase
The invention relates to a reaction system for alkyl nitrite and a preparation method for the alkyl nitrite. The preparation method comprises the following steps that: a trickle-bed reactor is adopted and porcelain ring filler is filled into the trickle-bed reactor; liquid added into the reactor forms liquid membranes on the filler; alkyl alcohol is taken as a liquid-phase raw material and nitric monoxide, oxygen and nitrogen are taken as gas-phase raw materials; and gas and liquid phases are reacted in the trickle-bed reactor by means of co-current flow or countercurrent flow under the condition of nitrogen protection so as to generate the alkyl nitrite. The mol ratio of the alkyl alcohol to NO is between 2 to 1 and 7 to 1, and the mol ratio of the NO to O2 is between 4 to 1 and 8 to 1; the reaction temperature is between 40 and 100 DEG C, and the reaction pressure is between 0.1 and 1.0 MPa; and the retention time of continuous feeding of liquid-phase alkyl alcohol in the trickle-bed reactor is between 10 and 500 minutes, and the retention time of continuous feeding of gas phase in the trickle-bed reactor is between 0.02 and 1 minute. The reaction system and the preparation method have the advantages that: the operating condition is mild; the reaction time is short; the conversion rate of the raw materials, and the selectivity and the yield of products are high; and the whole reaction system is convenient for realizing continuous production, etc.
Owner:TIANJIN UNIV
Rutile type titanium dioxide nanowire film and preparation method and applications thereof
InactiveCN102774883ASolve the difficult preparation of rutile phaseSolve conductivity problemsPhysical/chemical process catalystsElectrolytic capacitorsSulfate radicalsTithonia longiradiata
The invention relates to a rutile type titanium dioxide nanowire film and a preparation method and applications thereof. The preparation method of the film comprises the steps of directly growing rutile phase titanium dioxide nanowire array on a conductive glass substrate, and evenly distributing titanium dioxide nanowire clusters on the surface of the top end of the array; and regulating the proportion of various reactants, reaction time, temperature and other factors by taking sulfate radical-containing titanium slat as precursor for preparing the film. The invention further provides applications of the film, and the film can be used as anode materials to assemble a solar cell, or as photocatalyst to carry out water photolysis or organic matter degradation treatment. The method is simple, and has strong controllability, the problems that therutile phase is difficult to be prepared by using the sulfate radical-containing titanium slat and the titanium dioxide nanowire array grows on the conductive substrate in situ are solved, and the change on the diameter and length of the nanowire can be realized while the no change on crystallinity of a sample can be ensured through a calcination method.
Owner:QINGDAO UNIV OF SCI & TECH
Preparation method of biodegradable polyester with low carboxyl end group content
The invention relates to a preparation method of biodegradable polyester, and specifically relates to a preparation method of biodegradable polyester with a low carboxyl end group content. The method comprises esterification reactions and condensation and polymerization reactions: after the esterification reaction, polycyclic oxy-compounds are directly added into the reaction products to carry out reactions, and the reaction products are subjected to carry out condensation and polymerization reactions so as to obtain the biodegradable polyester with a low carboxyl end group content, wherein the carboxyl end group content is 5 to 20 mmol / kg. The invention aims to solve the problem of high carboxyl end group content in biodegradable polyesters, namely aliphatic polyester / copolymer and aliphatic-aromatic copolyester materials. The method provided by the invention reduces the carboxyl end group content in the materials, improves the flux strength, shortens the reaction time, obtains a polyester material with a good color (low b value), improves the anti-aging ability of the biodegradable polyester material, and prolongs the service life of the biodegradable polyester material. The preparation method has the advantages of controllable reaction conditions, low cost, and suitability for industrial production.
Owner:山东悦泰生物新材料有限公司