Amorphous entecavir p-toluenesulfonate salt and preparation method and medicament application thereof
A technology of p-toluenesulfonate and entecavir, applied in the field of medicine, can solve problems such as differences in bioavailability, and achieve the effects of good stability, improved production quality and high purity
Inactive Publication Date: 2010-07-28
FUJIAN COSUNTER PHARMA
View PDF1 Cites 6 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Different crystalline forms of the same drug may have significantly different bioavailability
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0061] Put 2.95g of entecavir monohydrate in a reaction flask, add 30.0ml of methanol, stir, heat the reaction solution to 40°C, add a solution made of 2.0g of p-toluenesulfonic acid and 5.0ml of methanol in batches, after the addition, 65 React at ℃ for 3 hours, concentrate under reduced pressure to 12.5ml, cool to 0℃, add 130.0ml ethyl acetate, a viscous solid precipitates, pour off the solvent, wash with 4.0ml ethyl acetate, and dry the residue in vacuum to obtain a white foam Solid para-amorphous entecavir p-toluenesulfonate 4.45g.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
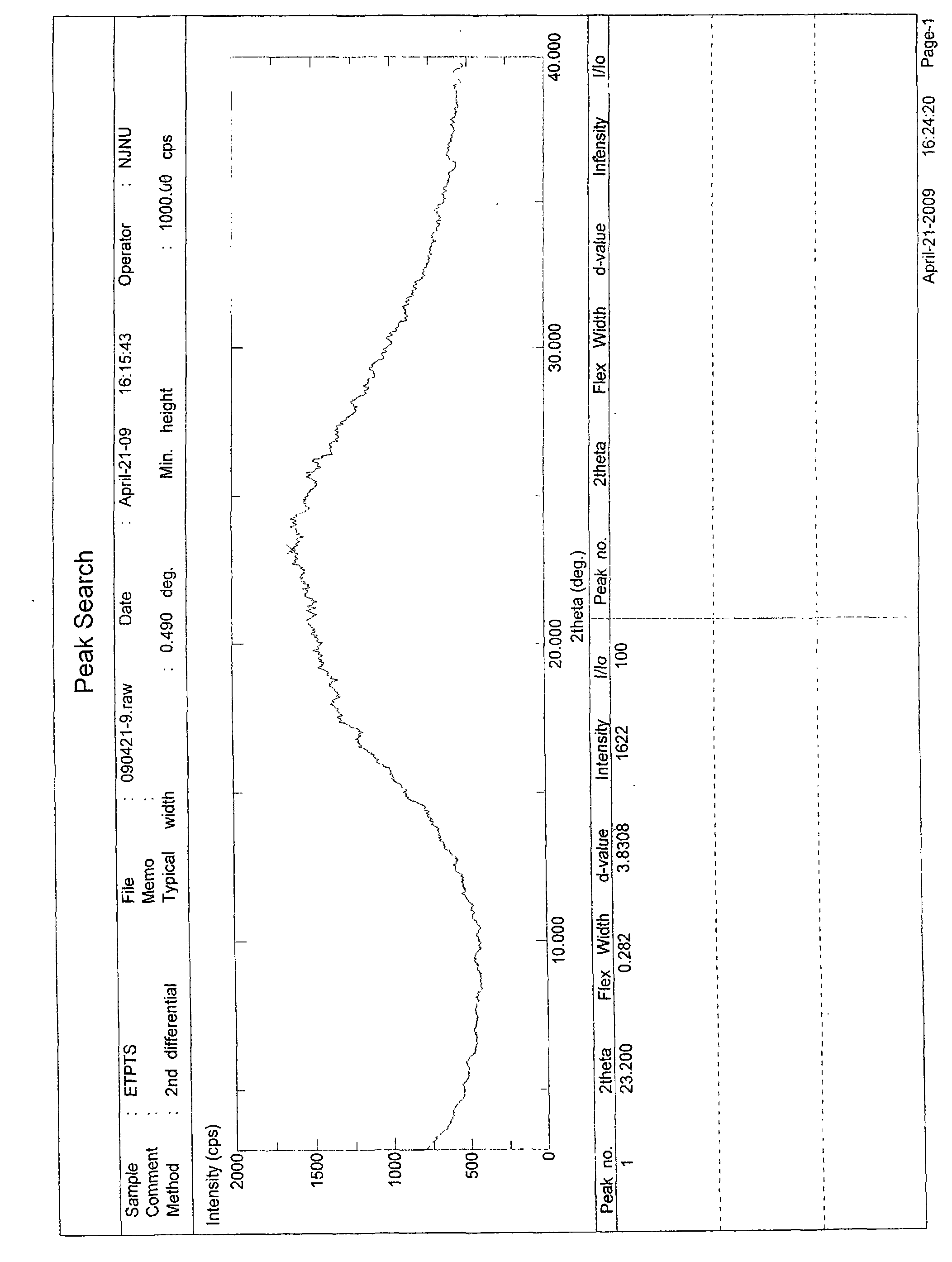
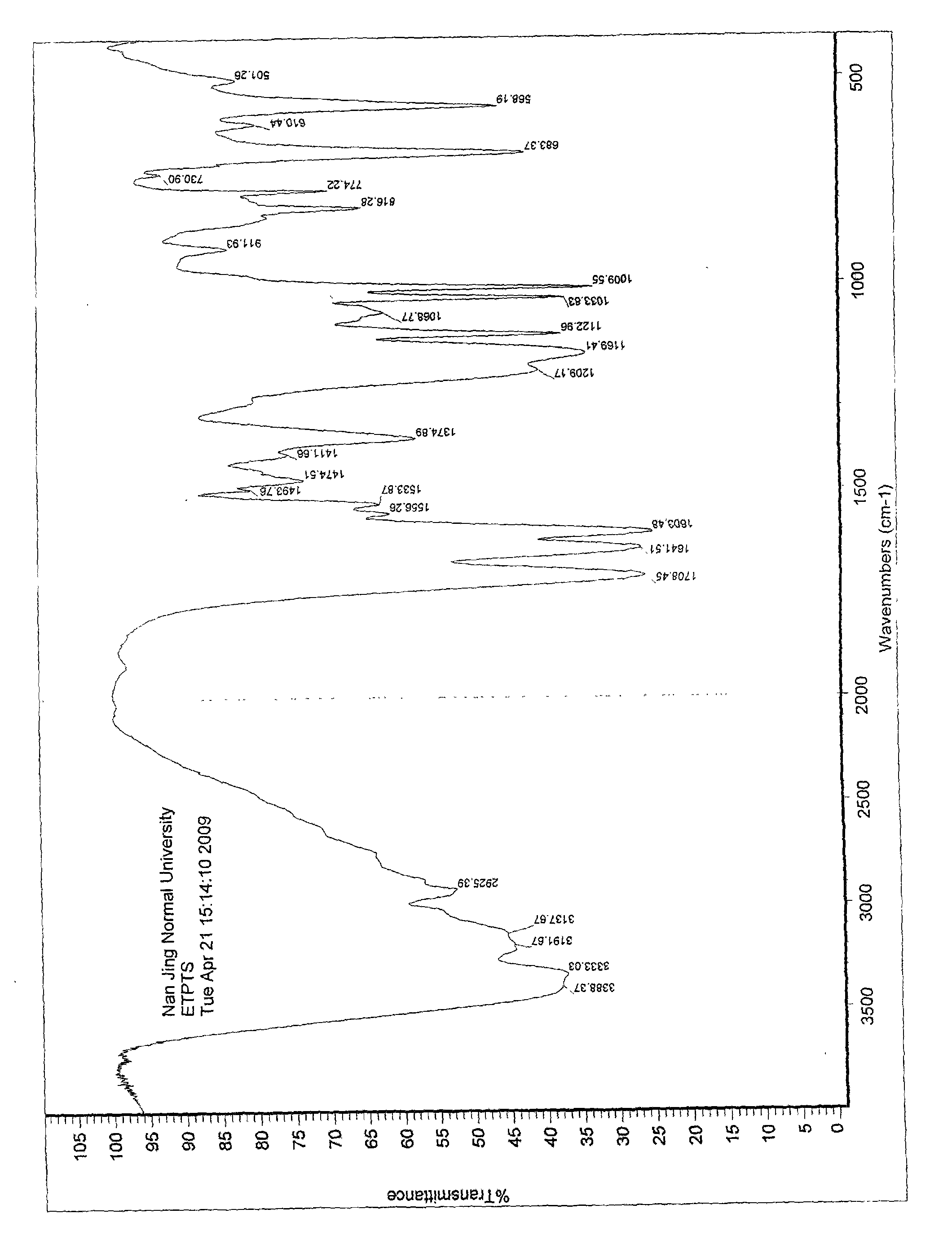
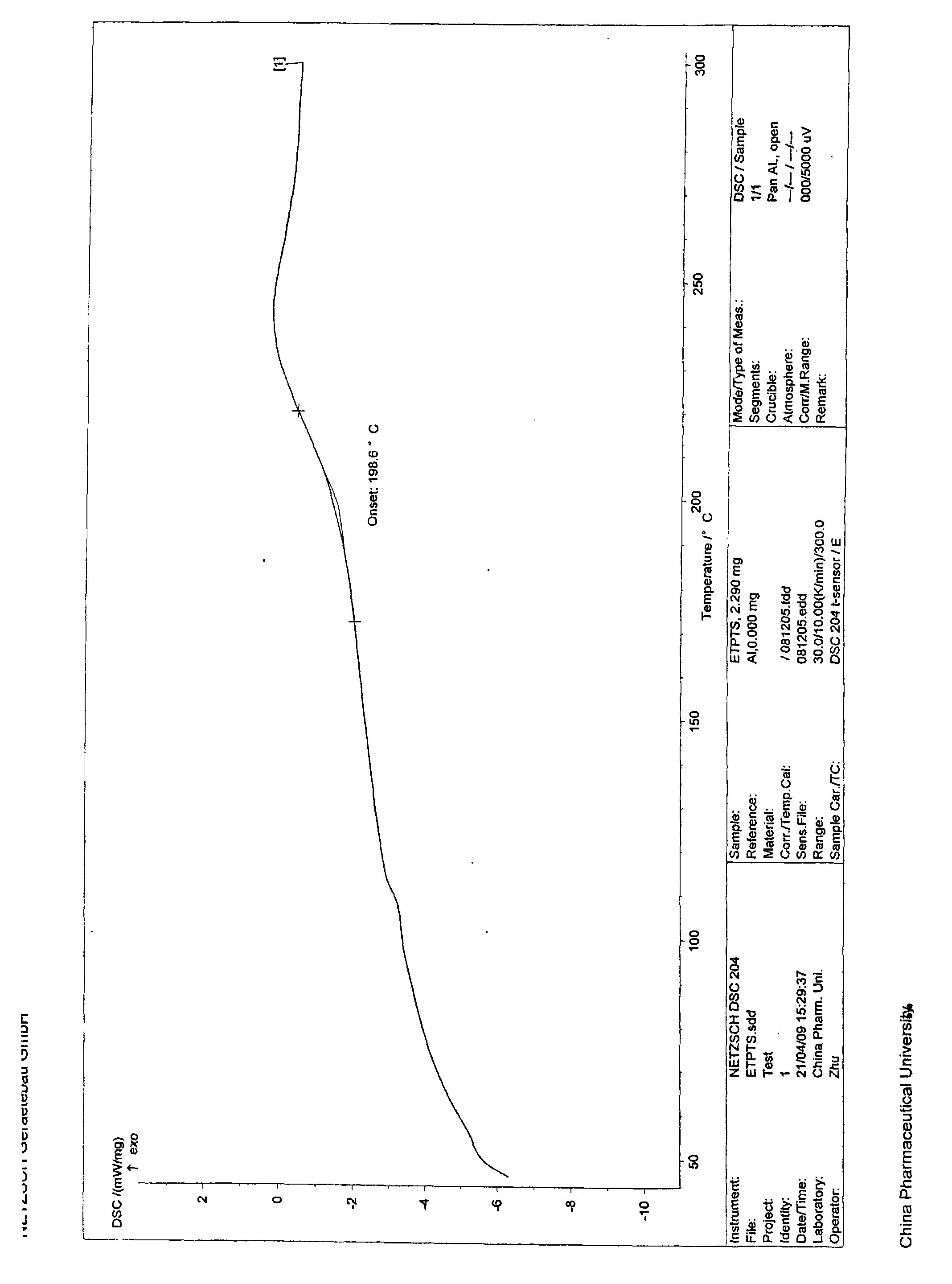
The invention provides amorphous entecavir p-toluenesulfonate salt with high bioavailability, high stability, high yield and high purity. The amorphous entecavir p-toluenesulfonate salt is determined as an amorphous state of entecavir p-toluenesulfonate salt through detections such as X-ray diffraction, infrared absorption spectrum determined by a KBr tablet method, differential scanning calorimetry (DSC) and the like. The invention also relates to a preparation method and medicament application of the amorphous salt.
Description
technical field [0001] The invention belongs to the field of medicine, and in particular relates to an antiviral drug entecavir tosylate, an amorphous crystal, a preparation method and a drug application thereof. technical background [0002] Entecavir (Entecavir) is a 2,-pentanecyclodeoxyguanosine analogue, the chemical name is [1S-(1α,3α,4β)]-2-amino-1,9-dihydro-9-[4 -Hydroxy-3-(hydroxymethyl)-2-methylenecyclopentyl]-6H-purin-6-one, usually monohydrate, the molecular structure is as follows: [0003] Molecular formula C 12 h 15 N 5 o 3 . [0004] Entecavir is a chiral compound, [1S-(1α, 3α, 4β)] optical isomer has a strong anti-hepatitis B (HBV) toxicity. [0005] Entecavir is a highly effective antiviral agent. Clinical studies have shown a good inhibitory effect on hepatitis B virus. Because entecavir has a very high anti-hepatitis B virus activity, a very low dose is sufficient to achieve the desired curative effect. Generally, adults daily Oral administration ...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): C07D473/18A61K31/522A61P31/20
Inventor 康惠燕陈国华
Owner FUJIAN COSUNTER PHARMA