Ciprofluoroquinolone C3/C7 dithizone Schiff base, preparation method and application thereof
A technology of ciprofluoroquinolone and Schiff base, which is applied in the field of medicine, can solve the problems that ciprofluoroquinolone has not yet been seen, and achieve the effect of strong anti-tumor activity and strong in vitro cytotoxic activity
Inactive Publication Date: 2012-06-27
HENAN UNIVERSITY
View PDF0 Cites 3 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
In addition, although retaining the 7-piperazinyl group is a common structural modification method for antibacterial fluoroquinolones, we know little about whether the piperazinyl group can be replaced by other groups for antitumor
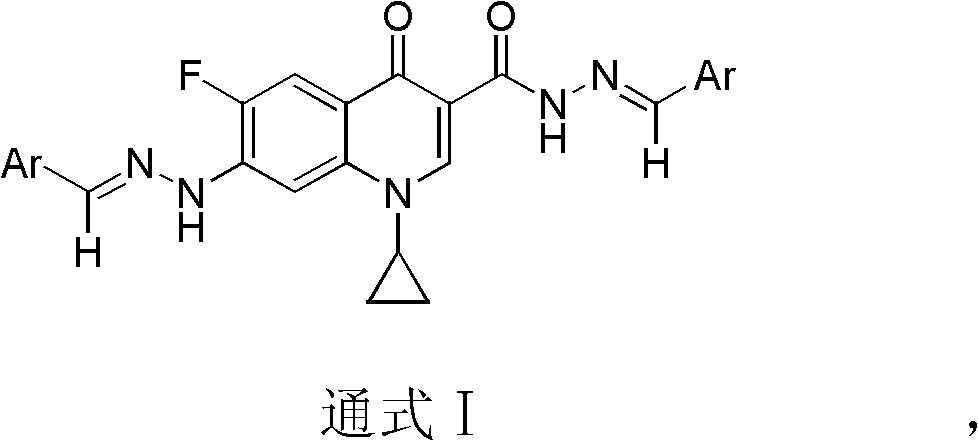
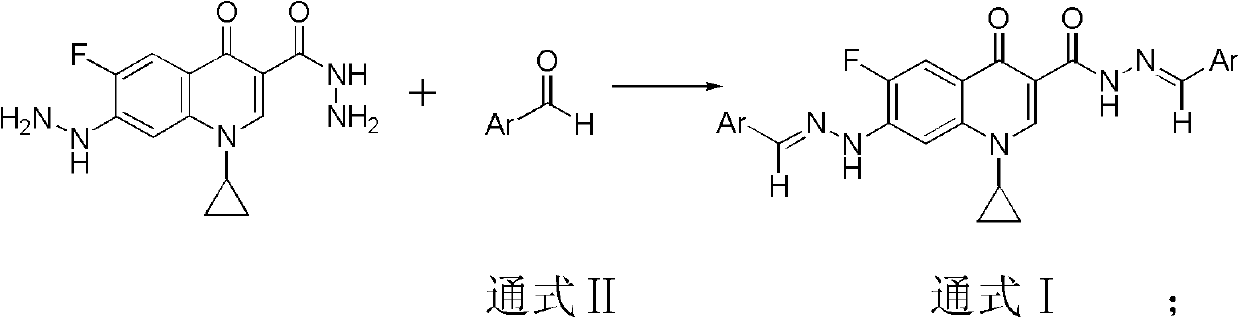
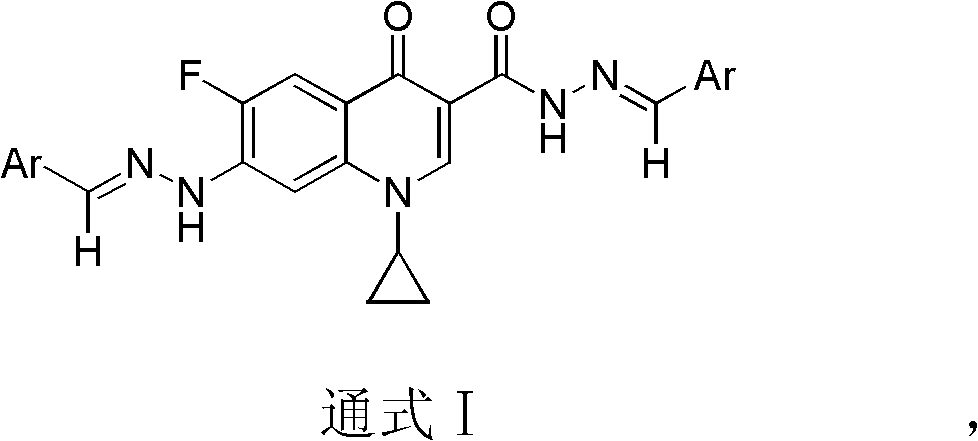
In view of the wide application of acylhydrazone and hydrazone compounds in antibacterial, antiviral, antitumor, etc., acylhydrazone and hydrazone were used to replace the carboxyl and piperazine groups at the C-3 and C-7 positions of the antibacterial fluoroquinolone skeleton, respectively, to obtain a new structure The dihydrazone Schiff base antitumor fluoroquinolone lead compound of the present invention is the main purpose of the present invention, and there is no relevant report on the cyprofluoroquinolone C3 / C7 dihydrazone Schiff base
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1-15
[0018] Examples 1-15 are specific examples of ciprofluoroquinolone C3 / C7 dihydrazone Schiff base.
Embodiment 1
[0020] The Cyprofluoroquinolone C3 / C7 dihydrazone Schiff base provided in this example is benzaldehyde acetal (1-cyclopropyl-6-fluoro-7-hydrazino-quinolin-4(1H)-one-3-methanol Hydrazide) dihydrazone, its structural formula is:
[0021]
Embodiment 2
[0023] The Cyprofluoroquinolone C3 / C7 dihydrazone Schiff base provided in this example is p-tolualdehyde acetal (1-cyclopropyl-6-fluoro-7-hydrazino-quinolin-4(1H)-one- 3-formylhydrazide) dihydrazone, its structural formula is:
[0024]
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention belongs to the technical field of medicine, particularly discloses a ciprofluoroquinolone C3 / C7 dithizone Schiff base, and meanwhile discloses a preparation method of the ciprofluoroquinolone C3 / C7 dithizone Schiff base and application of the ciprofluoroquinolone C3 / C7 dithizone Schiff base in pharmaceuticals. The chemical structural formula of the ciprofluoroquinolone C3 / C7 dithizone Schiff base is shown in a general formula I, wherein Ar is selected from phenyl, substituted phenyl, heterocyclic aromatic aryl alkyl and substituted heterocyclic aromatic aryl alkyl. Tests show that the ciprofluoroquinolone C3 / C7 dithizone Schiff base provided by the invention has strong in vitro cytotoxic activity to L1210, CHO (Chinese hamster ovary) and HL60 cancer cell strains, has strong antitumor activity, can be used for preparing antitumor drugs with human body acceptable pharmaceutical carrier, and can be further made into human body acceptable pharmaceutical salts for preparing antitumor drugs.
Description
technical field [0001] The invention belongs to the technical field of medicine, and in particular relates to a ciprofluoroquinolone C3 / C7 dihydrazone Schiff base, and also relates to its preparation method and its application in pharmacy. Background technique [0002] Fluoroquinolones (FQ) are clinical antibacterial drugs developed in the 1980s, represented by norfloxacin. Based on the similar function of its target topoisomerase (TOPO) and mammalian TOPO, it is a new research direction to transform antibacterial FQ into antitumor FQ. Although anti-tumor FQ compounds including bicyclic quinolones, tricyclic quinolones, tetracyclic quinolones, chiral quinolones, flavonoids and other anti-tumor FQ compounds have been designed and synthesized, at present, these compounds have toxicity and activity in parallel to varying degrees, and poor solubility in vivo Leading to common problems such as low bioavailability, easy to be metabolized and inactivated in vivo. Therefore, findi...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): C07D215/56C07D405/14C07D401/14A61P35/00A61P35/02
Inventor 胡国强敬永升陈寅生张东娣张亚红毋小魁王国强段楠楠温晓漪曹铁耀
Owner HENAN UNIVERSITY