Solid preparation
A technology for solid preparations and drugs, which is applied in the directions of adhesives, pill delivery, medical preparations with inactive ingredients, etc., can solve the problems of decreased drug dissolution, easy decomposition and deterioration of drugs, etc. Highly disintegrating effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1)
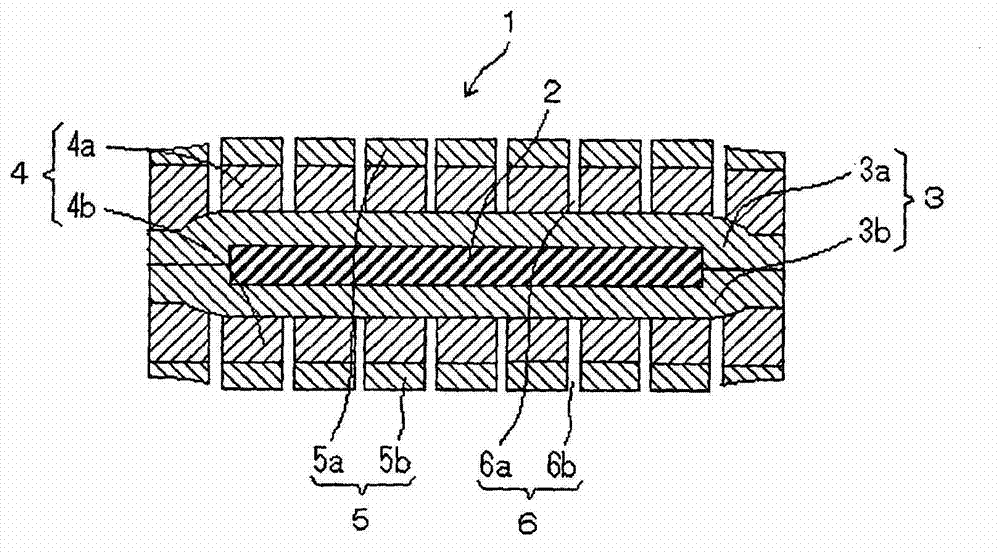
[0169] (a) Anti-adhesion layer production process
[0170] The coating solution A containing the constituent components of the antiadhesive layer was prepared as follows.
[0171] Calcium chloride (Calcium chloride H, manufactured by Tomita Pharmaceutical Co., Ltd.): 0.27 parts by mass was added as a viscosity reducer to purified water: 380 parts by mass, and stirred for 5 minutes to dissolve it. Polyacrylic acid (Carbopol 974P, manufactured by Nobeon Co., Ltd., 0.2% by mass aqueous solution viscosity (20°C): 12100 mPa·s): 10 parts by mass was slowly added to the solution, and after the addition, stirred for 1 hour, and each The mixture of ingredients was heated to 80°C. Slowly add hydroxypropyl methylcellulose (TC-5E, manufactured by Shin-Etsu Chemical Co., Ltd., 2% by mass aqueous solution viscosity (20°C): 3mPa·s) as an antiadhesive agent to the mixed liquid while stirring: 80 After adding parts by mass, stir for 15 minutes, lower the liquid temperature to 30° C., and sti...
Embodiment 2)
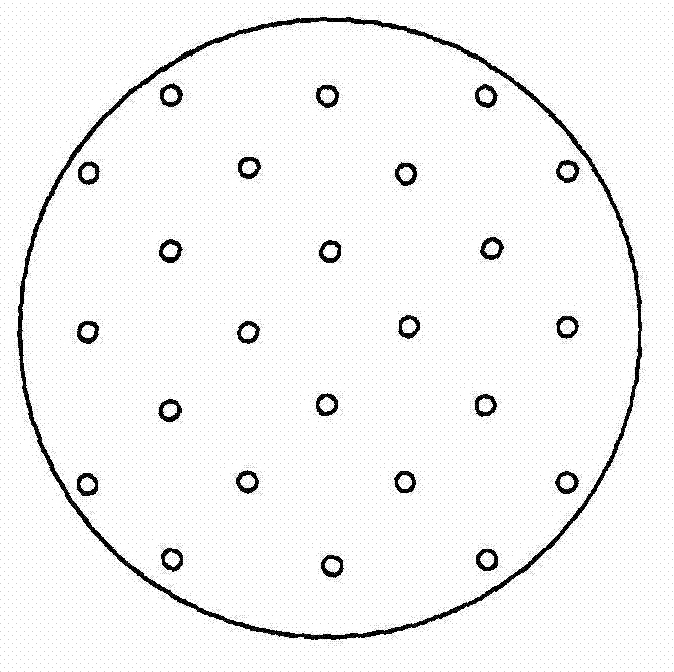
[0186] Except in the through-hole forming step (c), laser (carbon dioxide laser, pulse wave irradiation) is used to form a center-to-center layer in the laminated intermediate 2b (laminate of gel-forming layer / anti-adhesion layer / peelable base material). A solid preparation (orally administered drug) was produced in the same manner as in Example 1 above, except that pores with an average pore diameter of 70 μmφ were formed at a distance of 2.0 mm.
PUM
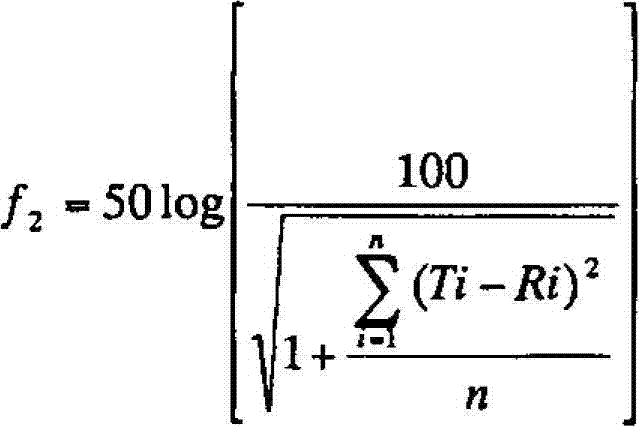
| Property | Measurement | Unit |
|---|---|---|
| diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More