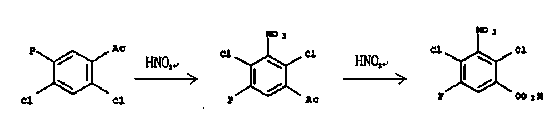
Synthesis method for 2,4-dichloro-3-nitro-5-fluorobenzoic acid
A technology for the synthesis of fluorobenzoic acid and methods, applied in chemical instruments and methods, preparation of organic compounds, organic chemistry, etc., can solve the problems of cumbersome operation, high price, and difficult availability of starting materials, etc., and achieve simple process and high product quality The effect of high yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0017] Add 280 grams of 90% nitric acid into the reactor, control the temperature from 0°C to 10°C, add 207 grams of 2,4-dichloro-5-fluoroacetophenone in batches, and maintain the temperature from 0°C to 10°C after adding React in a range of 4 hours, then slowly raise the temperature to 40°C and keep it for 2 hours, then slowly increase the temperature at a controlled rate, up to 110°C, and keep it at this temperature for 1 hour. After the reaction, cool down to room temperature, then add ice water, filter , collect the solid, and refine the product to obtain the product. After the nitration reaction is completed, the temperature is raised to 50°C to 150°C for oxidation reaction until the reaction is complete, and 221 grams of the product are obtained in the latter process, M.P.192~196°C.
Embodiment 2
[0019] Add 250 grams of 98% nitric acid to the reactor, control the temperature from 0°C to 10°C, add 207 grams of 2,4-dichloro-5-fluoroacetophenone in batches, and maintain the temperature from 0°C to 10°C after adding React in a range of 4 hours, then slowly raise the temperature to 40°C and keep it for 2 hours, then slowly increase the temperature at a controlled rate, up to 110°C, and keep it at this temperature for 1 hour. After the reaction, cool down to room temperature, then add ice water, filter , collect the solid, and refine the product to obtain the product. After the nitration reaction is completed, the temperature is raised to 50°C to 150°C for oxidation reaction until the reaction is complete, and 233 grams of the product are obtained in the latter process, M.P.192~195°C.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More