Celecoxib and irbesartan coamorphous substance
An amorphous substance, celecoxib technology, applied in the field of medicine, can solve problems such as difficulty in preparing ideal solid dosage forms, low bulk density, and poor compressibility
Inactive Publication Date: 2016-06-08
CHINA PHARM UNIV
View PDF4 Cites 8 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Celecoxib is in the form of long needle-like crystals, resulting in low bulk density and poor compressibility, making it difficult to prepare ideal solid dosage forms
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0062] Example 1: Preparation of co-amorphous celecoxib irbesartan
[0063] Add 0.300g celecoxib and 0.339g irbesartan into 50ml of acetonitrile, stir at room temperature (20±5°C) to obtain a clear solution, then evaporate the solvent under reduced pressure at 55°C, and dry in vacuum at 25°C for 24h , to obtain 0.541 g of white powder.
Embodiment 2
[0064] Example 2: Preparation of co-amorphous celecoxib irbesartan
[0065] Add 0.300g celecoxib and 0.339g irbesartan into 50ml of methanol, stir at room temperature (20±5°C) to obtain a clear solution, then evaporate the solvent under reduced pressure at 55°C, and dry in vacuum at 25°C for 24h , to obtain 0.585 g of white powder.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
| Property | Measurement | Unit |
|---|---|---|
| glass transition temperature | aaaaa | aaaaa |
| wavelength | aaaaa | aaaaa |
Login to View More
Abstract
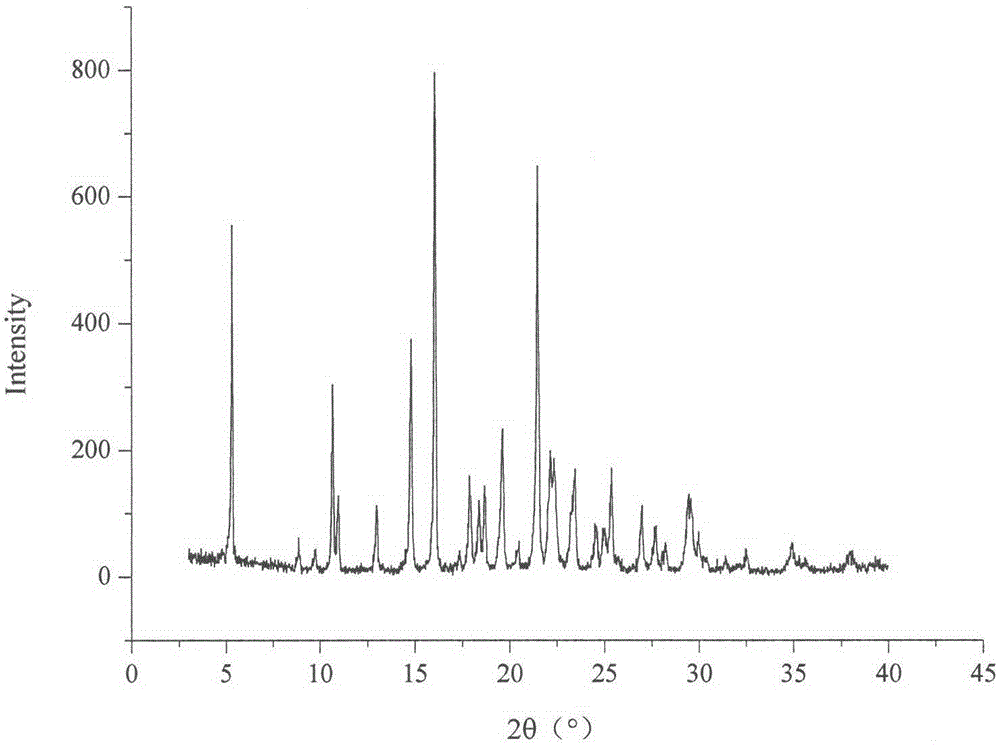
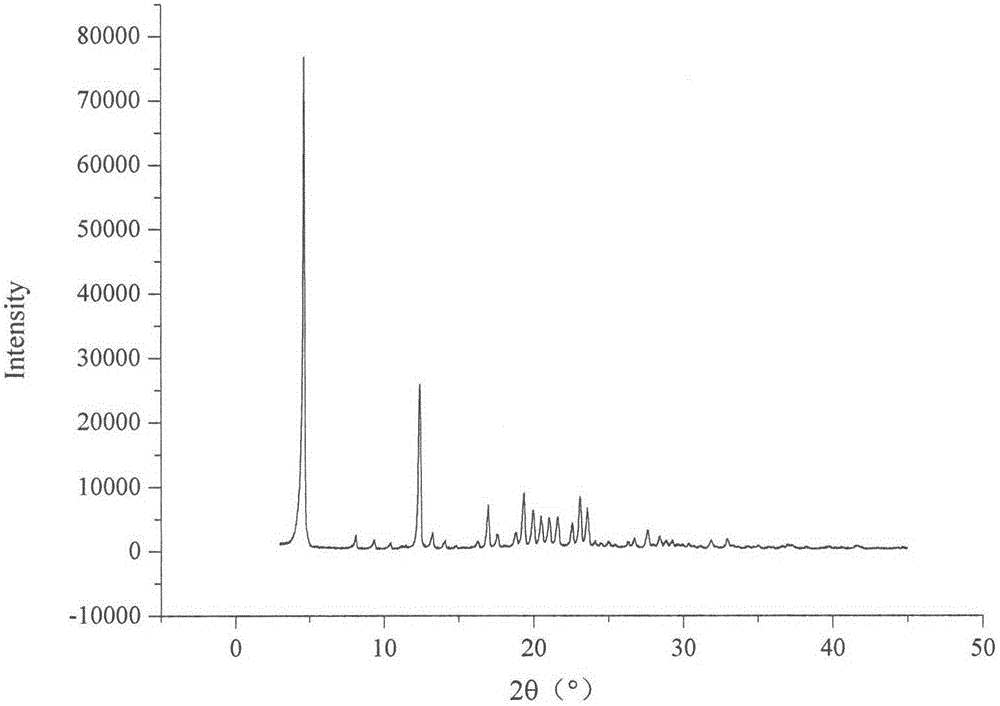
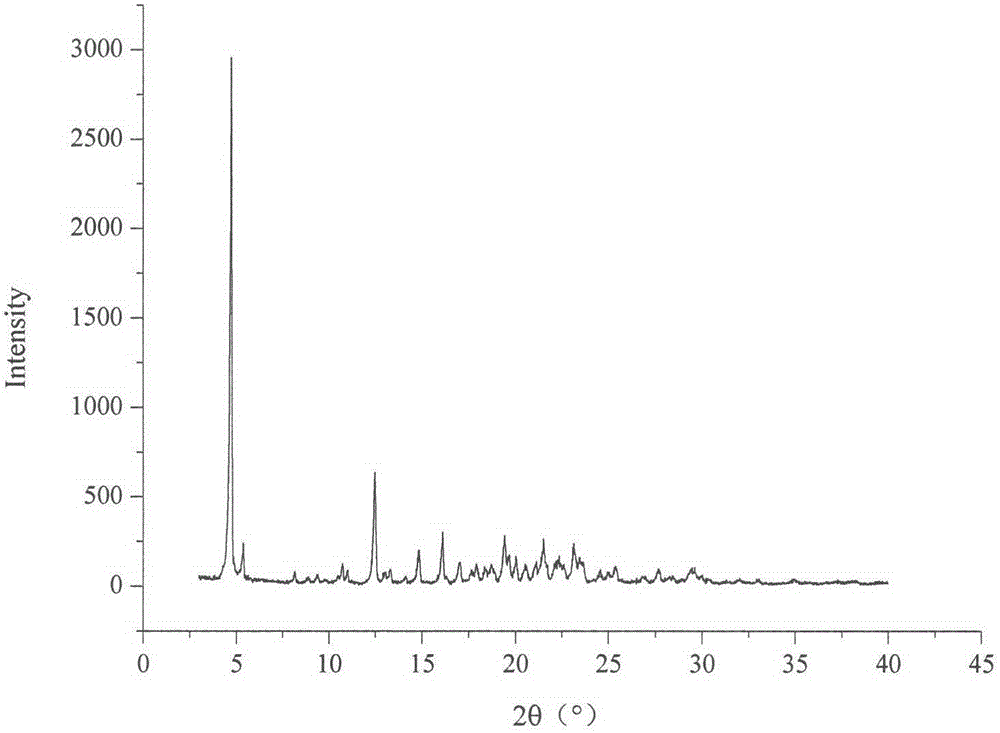
The invention relates to a celecoxib and irbesartan coamorphous substance. Solubility of the celecoxib and irbesartan coamorphous substance in various buffer solutions is remarkably increased. The celecoxib and irbesartan coamorphous substance is an amorphous state totally different from celecoxib and irbesartan crystals and is different from the celecoxib and irbesartan crystals in melting point, x-ray powder diffraction spectrogram, DSC (differential scanning calorimetry) diagram and infrared spectrum. Under Cu-Kalpha radiation, the x-ray powder diffraction spectrogram shown by degree 2theta has no sharp diffraction peak. The glass-transition temperature of the celecoxib and irbesartan coamorphous substance is about 74.5 DEG C.
Description
technical field [0001] The invention belongs to the technical field of medicines, and in particular relates to celecoxib-irbesartan co-amorphous formed by combining celecoxib and irbesartan at a molar ratio of 1:1 and a preparation method thereof. Background technique [0002] Celecoxib (CX), the chemical name is 4-[5-(4-benzyl)-3-(trifluoromethyl)-1 hydrogen-1-pyrazol-1-yl]sulfonamide, belongs to Non-steroidal anti-inflammatory drugs (NSAIDs), used for the treatment of rheumatoid arthritis, osteoarthritis, relief of various fever and various pain symptoms, its inhibitory effect on cyclooxygenase 2 (cox-2) is 400 times that of cox-1. Celecoxib is in the form of long needle-like crystals, resulting in low bulk density and poor compressibility, making it difficult to prepare ideal solid dosage forms. Moreover, Cx is a weakly acidic drug of BCSII, with a pKa of 11.1, almost insoluble in water, and low bioavailability after oral administration, and its absorption can be improv...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): C07D231/12C07D403/10
CPCC07D231/12C07D403/10
Inventor 张建军苗娜娜高缘
Owner CHINA PHARM UNIV