Anti-CEA-antigen VHH structural domains and bispecific antibody comprising same
A domain and single-domain antibody technology, applied in the field of immunology, can solve the problems of lack of binding epitopes, restricting the application of antibodies, and mutation, and achieve the effects of long in vivo half-life, excellent antibody recognition ability, and broad application prospects.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
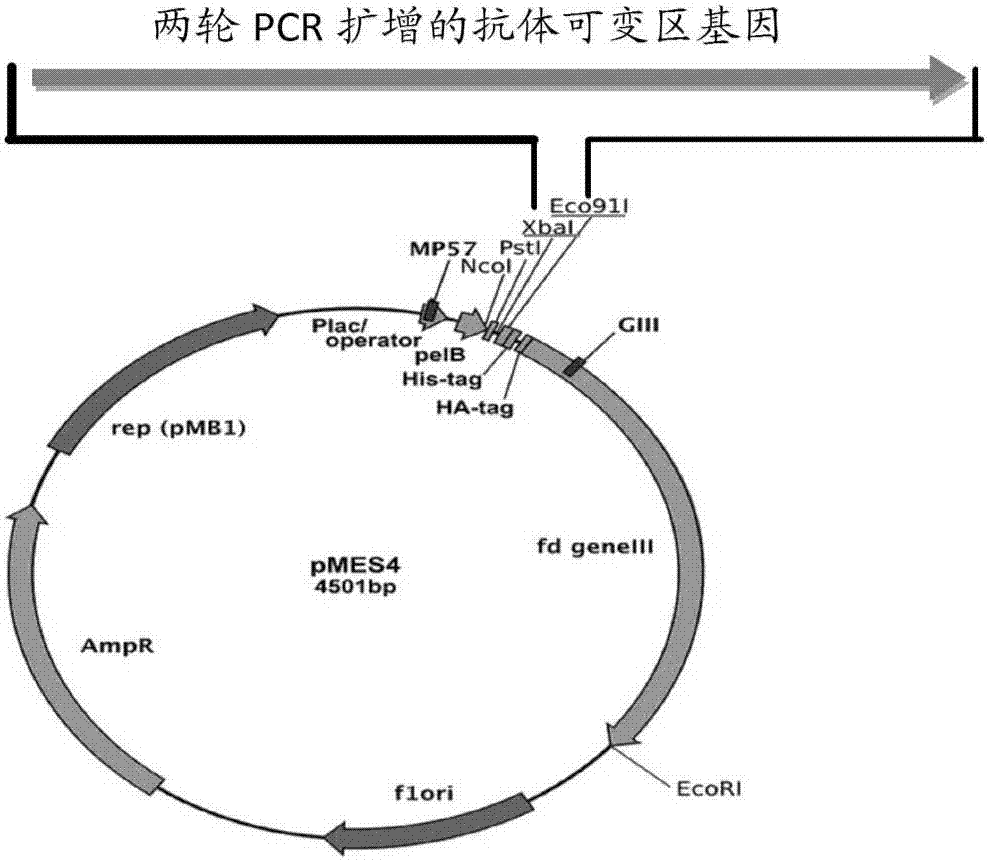
[0041] Example 1 Construction and Screening of Anti-CEA Nanobody Phage Display Library
[0042] 1.1 Immunization of alpaca: Select a healthy adult alpaca, mix the recombinant protein CEA and Freund's adjuvant at a ratio of 1:1, and immunize the alpaca by subcutaneous multi-point injection on the back at 6-7 μg / Kg. A total of four times of immunization, immunization interval of 2 weeks. Afterwards, the peripheral blood of the alpaca was collected for the construction of a phage display library.
[0043] 1.2 Separation of camel-derived lymphocytes: analyze lymphocytes from the collected camel-derived anticoagulated whole blood according to routine procedures in this technical field, every 2.5×10 7 Add 1mL RNA isolation reagent to each living cell, take 1mL for RNA extraction, and store the rest at -80°C.
[0044] 1.3 Extraction of total RNA: extract total RNA according to routine procedures in this technical field, and adjust the concentration to 1 μg / μL with RNase-free water ...
Embodiment 2
[0075] Example 2. Preparation of anti-CEA nanobody
[0076] 2.1 Amplification of original nanobody strain TG1 and transformation of nanobody recombinant plasmid into Escherichia coli BL21(DE3)
[0077] Inoculate the original strain TG1 glycerolbacterium containing Nanobody nucleic acid in 5 mL of fresh LB-A medium at a ratio of 1:1000, and culture overnight at 37°C and 200 rpm. The next day, plasmids were extracted using the Plasmid mini kit (OMEGA) according to the instructions. After verification, transform 1 ul of the above plasmid into 100 ul of competent cells, mix gently, place on ice for 30 min, heat shock in a water bath at 42°C for 90 s, and cool in an ice bath for 3 min. Add 600ul LB medium to the centrifuge tube, shake and culture at 37°C for 60min. Take 100ul of the supernatant, spread it on the LB-A plate with a triangle spreader, and culture it upside down at 37°C overnight.
[0078] 2.2 Induced expression and extraction of nanobodies
[0079] The above monoc...
Embodiment 3
[0082] Example 3 Affinity Activity of Anti-CEA Nanobody and CEA Antigen
[0083] 3.1 Chip antigen coupling
[0084] The antigen was prepared into a 20ug / mL working solution with different pH sodium acetate buffers (pH 5.5, pH 5.0, pH 4.5, pH4.0), and a 50mM NaOH regeneration solution was prepared at the same time, using the Biacore T100 protein interaction analysis system instrument The template method in the analysis of the electrostatic binding between the antigen and the surface of the chip (GE company) under different pH conditions, with the signal increase amount reaching 5 times RL as the standard, select the most appropriate neutral pH system and The antigen concentration was adjusted as the conjugation conditions. The chip was coupled according to the template method that comes with the instrument: select the blank coupling mode for channel 1, select the Target coupling mode for channel 2, and set the target to the designed theoretical coupling amount. The coupling p...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More