Nutritional type traditional Chinese medicine granules for enhancing disease resistance of livestock and poultry and preparation method thereof
A technology of disease resistance and traditional Chinese medicine granules, which is applied in the field of traditional Chinese veterinary medicine, can solve problems such as limited feed formula space, influence of feed palatability, and unstable effect, so as to reduce breeding losses, prevent and treat livestock and poultry diseases, and improve product purity. Improved effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
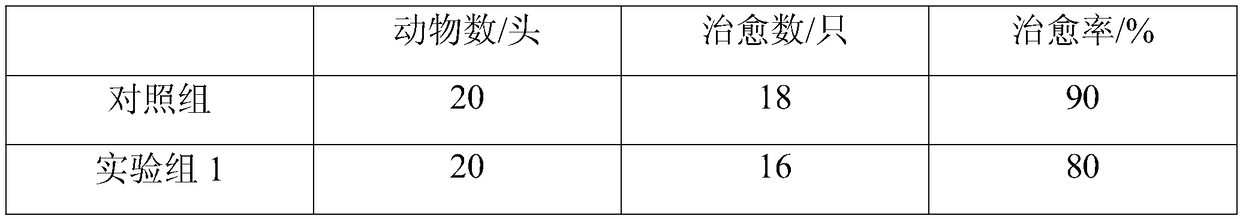
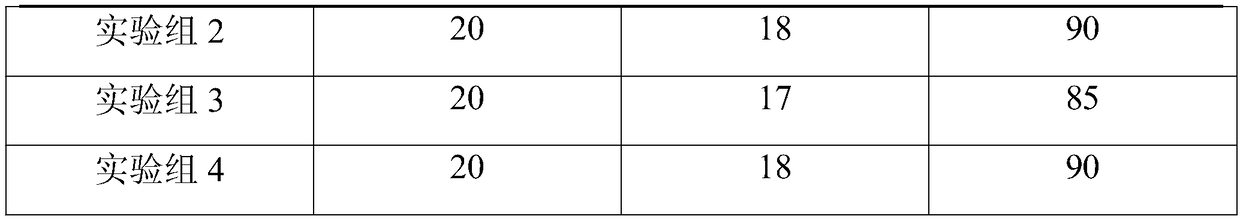
[0027] A nutritional traditional Chinese medicine granule for enhancing the disease resistance of livestock and poultry, comprising the following raw materials in parts by weight: 20 parts of Qinpi, 20 parts of Pulsatilla, 20 parts of Eucommia, 15 parts of persimmon leaves, 15 parts of Jinbuhuan, 12 parts of hawthorn, and 12 parts of Baibu 10 parts of flavescens, 10 parts of seaweed, 10 parts of cassia, 8 parts of wolfberry, 8 parts of rhodiola, 8 parts of daylilies, 8 parts of impatiens, 5 parts of jasmine, 5 parts of osmanthus, 3 parts of sweet orange leaves, 3 parts lemon juice.
[0028] A preparation method of nutritional traditional Chinese medicine granules for enhancing the disease resistance of livestock and poultry as described above, comprising the following steps:
[0029] (1) Pulverize Qinpi, Pulsatilla, Eucommia, Persimmon Leaf, Jinbuhuan, Hawthorn, Poria, Sophora, Laver, Cassia, Lycium, Rhodiola, and mix them evenly according to the weight ratio;
[0030] (2) Pu...
Embodiment 2
[0035] A nutritional traditional Chinese medicine granule for enhancing the disease resistance of livestock and poultry, comprising the following raw materials in parts by weight: 30 parts of Qinpi, 28 parts of Pulsatilla ulmoides, 23 parts of Eucommia ulmoides, 22 parts of persimmon leaves, 20 parts of Jinbuhuan, 18 parts of hawthorn, and 13 parts of Baitou 12 parts of flavescens, 12 parts of seaweed, 12 parts of cassia, 10 parts of wolfberry, 10 parts of rhodiola, 9 parts of daylilies, 8 parts of impatiens, 8 parts of jasmine, 7 parts of osmanthus, 6 parts of sweet orange leaves, 5 parts lemon juice.
[0036] A preparation method of nutritional traditional Chinese medicine granules for enhancing the disease resistance of livestock and poultry as described above, comprising the following steps:
[0037] (1) Pulverize Qinpi, Pulsatilla, Eucommia, Persimmon Leaf, Jinbuhuan, Hawthorn, Poria, Sophora, Laver, Cassia, Lycium, Rhodiola, and mix them evenly according to the weight ra...
Embodiment 3
[0043] A nutritional traditional Chinese medicine granule for enhancing disease resistance of livestock and poultry, comprising the following raw materials in parts by weight: 25 parts of Qinpi, 25 parts of Pulsatilla, 25 parts of Eucommia, 20 parts of persimmon leaves, 20 parts of Jinbuhuan, 18 parts of Hawthorn, and 15 parts of Baibu 12 parts of flavescens, 12 parts of seaweed, 12 parts of cassia, 12 parts of wolfberry, 12 parts of rhodiola, 10 parts of daylilies, 10 parts of impatiens, 8 parts of jasmine, 8 parts of osmanthus, 6 parts of sweet orange leaves, 5 parts lemon juice.
[0044] A preparation method of nutritional traditional Chinese medicine granules for enhancing the disease resistance of livestock and poultry as described above, comprising the following steps:
[0045] (1) Pulverize Qinpi, Pulsatilla, Eucommia, Persimmon Leaf, Jinbuhuan, Hawthorn, Poria, Sophora, Laver, Cassia, Lycium, Rhodiola, and mix them evenly according to the weight ratio;
[0046] (2) Pu...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More