Patents
Literature
505results about How to "High product purity" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
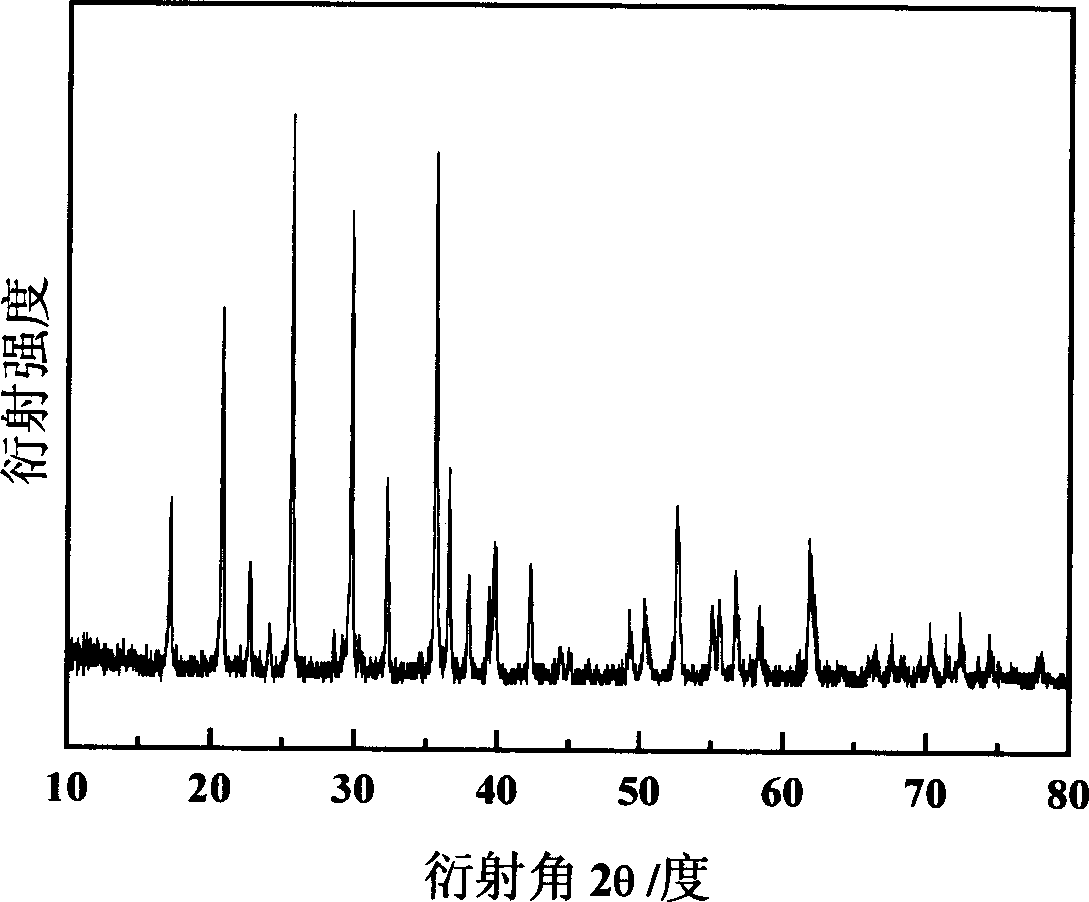
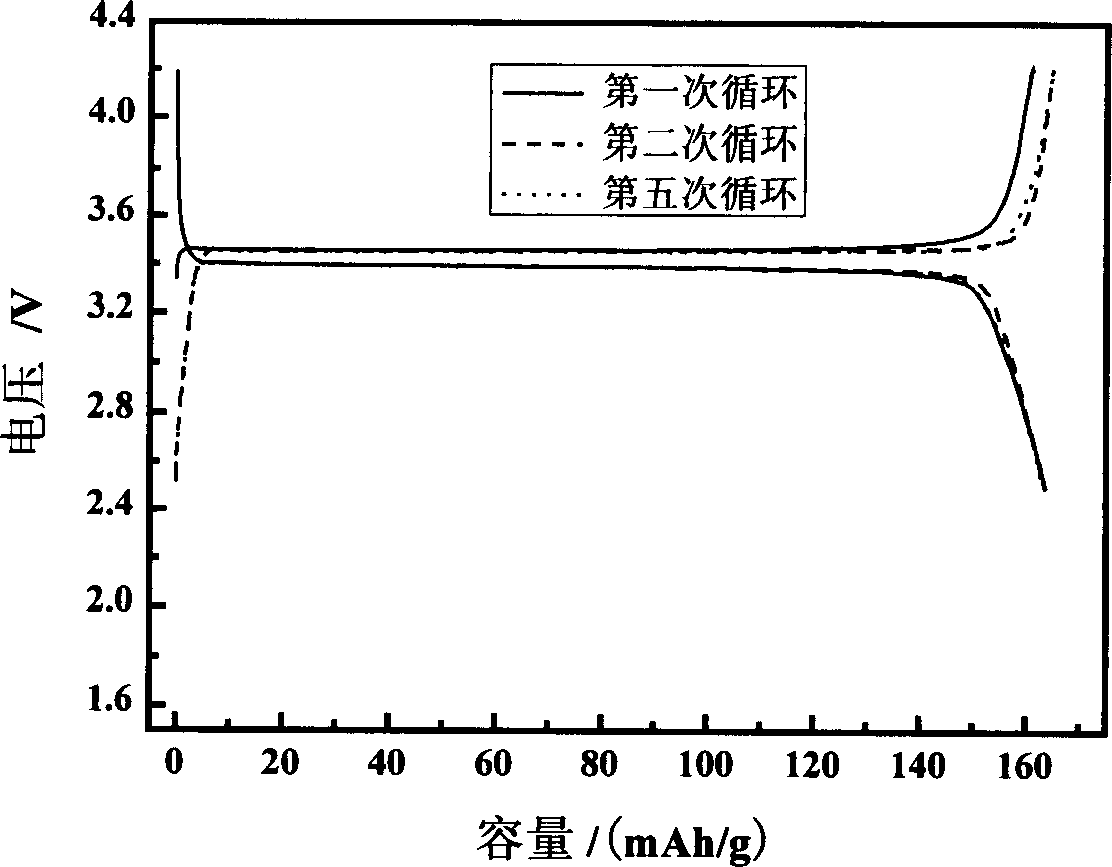
Method for preparing lithium ion battery anode material lithium ion phosphate
InactiveCN1803591ASimple preparation processHigh product purityCell electrodesPhosphorus compoundsSolventPhosphate
The invention discloses a preparation method for ferrous lithium phosphate as anode material of lithium cell. Wherein, dissolving the ferric iron salt, lithium salt, doped phosphorus salt and reducer into dissolvant to react for 0.5-30h and obtain the amorphous ferric lithium phosphate; heating with 5-30Deg / min speed at nitrogen, argon gas or mixed atmosphere to 450-800Deg; baking for 20-600min; cooling with 1-20Deg / min speed to room temperature, and obtaining the crystal-type product. This invention reduces the ferric iron into ferrous iron and generates the final product with high specific capacity and well cycle performance. Wherein, the first charge / discharge efficiency of the product can achieve 100%.
Owner:SHANGHAI JIAO TONG UNIV
Dabigatran etexilate intermediate, preparation method for same and method for preparing dabigatran etexilate
InactiveCN102633713AHigh product yieldHigh product purityOrganic chemistryReaction temperatureStructural formula
The invention relates to a dabigatran etexilate intermediate, a preparation method for the same and a method for preparing dabigatran etexilate, which belong to the technical field of pharmaceutical chemistry and pharmaceutical engineering. The dabigatran etexilate intermediate is represented as a structural formula (2), so that a preparation process for the dabigatran etexilate is simplified, yield and purity of the dabigatran etexilate are high, reaction temperature is low, expensive dehydrating agent is not needed, and good market prospect is provided.
Owner:NANJING UNIV OF TECH +1
New wild blueberry anthocyanin extraction and purification method
The invention belongs to the field of the natural organic chemistry, relates to a method for extracting and purifying anthocyanins from wild blueberries in Greater Khingan, and particularly relates to a new method adopting a biological enzyme process, ionic liquid microwave extraction, and ultrafiltration membrane concentration coupled high-speed countercurrent chromatography purification to finally obtain highly pure blueberry anthocyanins. The method has the following advantages: 1, an ionic liquid has a very strong microwave absorption ability, so the ionic liquid microwave extraction process is fast and efficient, has a simple and easy process, fully extracts the blueberry anthocyanins effective ingredients, and realizes a high content of the anthocyanins effective ingredients; and 2, compared with traditional solid-liquid chromatography technology, the high-speed countercurrent chromatography purification technology has the advantages of flexible operation, high efficiency, rapid separation speed, large preparation amount and low cost, and can be industrially used. The blueberry anthocyanins obtained by using the method have high content and high activity, and has a purity reaching 42.7%.
Owner:DAXINGANLING SHENZHOU NORTH POLE BLUEBERRY BIOLOGICAL ENG
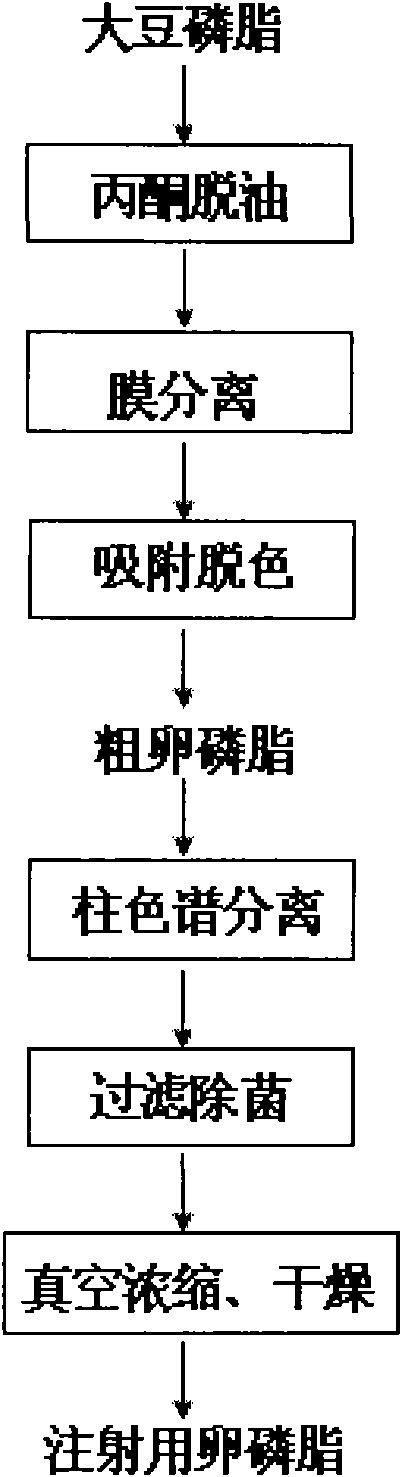
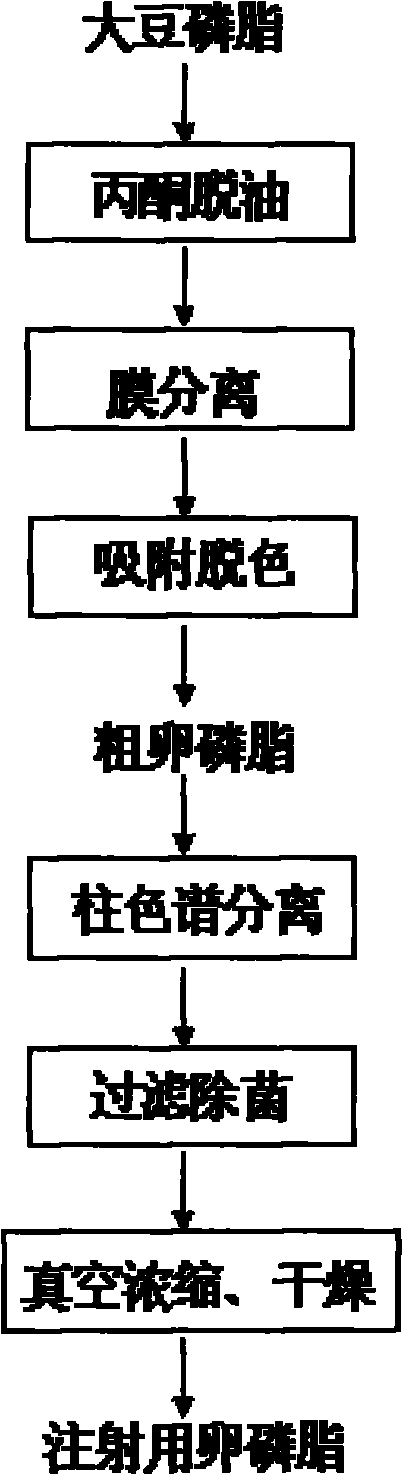
Preparation technology of soybean lecithin for injection
ActiveCN101792461AHigh product purityAbundant sources of raw materialsPhosphatide foodstuff compositionsSolventChemistry
The invention relates to a preparation technology of soybean lecithin for injection. Soybean lecithin is taken as a raw material, crude lecithin is firstly obtained after the treatment of acetone deoiling, membrane separation, adsorption decolorization and the like, then the crude lecithin is undergone column chromatographic fractionation, filtration sterilization, decompression concentration and vacuum drying, and finally the soybean lecithin for injection is obtained. The invention adopts membrane separation to replace the traditional solvent extraction, so the operation is easy. A polar compound with surface hydroxyl groups is taken as a stationary phase, mixed solvent composed of weak polar solvent and strong polar solvent is firstly used as eluent to carry out gradient elution, after all the cephalin flows out, the strong polar solvent is taken as the eluent, thereby not only the separation selectivity is high, but also the usage of the eluent is greatly reduced, and meanwhile, the extraction efficiency of lecithin is high, the product quality is stable, and the industrial mass production is benefited.
Owner:NANJING UNIV OF TECH +1
Method for preparing scutellarin
ActiveCN101941999AHigh yieldHigh product puritySugar derivativesSugar derivatives preparationBasic hydrolysisPhenols
Owner:KPC PHARM INC +1
Purification method of silicon carbide superfine micropowder
The invention discloses a purification method of silicon carbide superfine micropowder, which comprises the following steps: mechanically crushing and shaping the selected silicon carbide raw material, washing the shaped raw material with initial water to remove carbon and magnetically separating the washed raw material to remove partial impurities on the surface; airing and drying the raw material washed with initial water, and carrying out crushing by a raymond mill and air flow classification to produce a product the grain diameter of which is between 1 and 15mu m; adding the product into a purification kettle, adding water to produce slurry, adding a flotation agent for floating to remove carbon, wherein the floating agent is kerosene and terpineol; respectively adding hydrochloric acid, sulfuric acid and hydrofluoric acid into the purification kettle; stirring the mixture and then washing with water until the pH value reaches about 7; adding solid sodium hydroxide and solid calcium hydroxide into the purification kettle then washing with water after stirring; and feeding the washed mixture to a classification kettle for classification after the pH value reaches 7. Through the purification treatment by using silicon carbide, the superfine micropowder is easy to separate. The product has the advantages of high purity, small grain diameter distribution, uniform grains, fewer impurities, high quality and wider application range.
Owner:连云港市沃鑫高新材料有限公司
Porous ferric oxide/carbon nanometer plate composite material as well as preparation method thereof and application in preparing lithium ion battery
ActiveCN103682251AThe process is simple and easy to controlHigh product purityMaterial nanotechnologyCell electrodesAmmonium sulfateChemistry
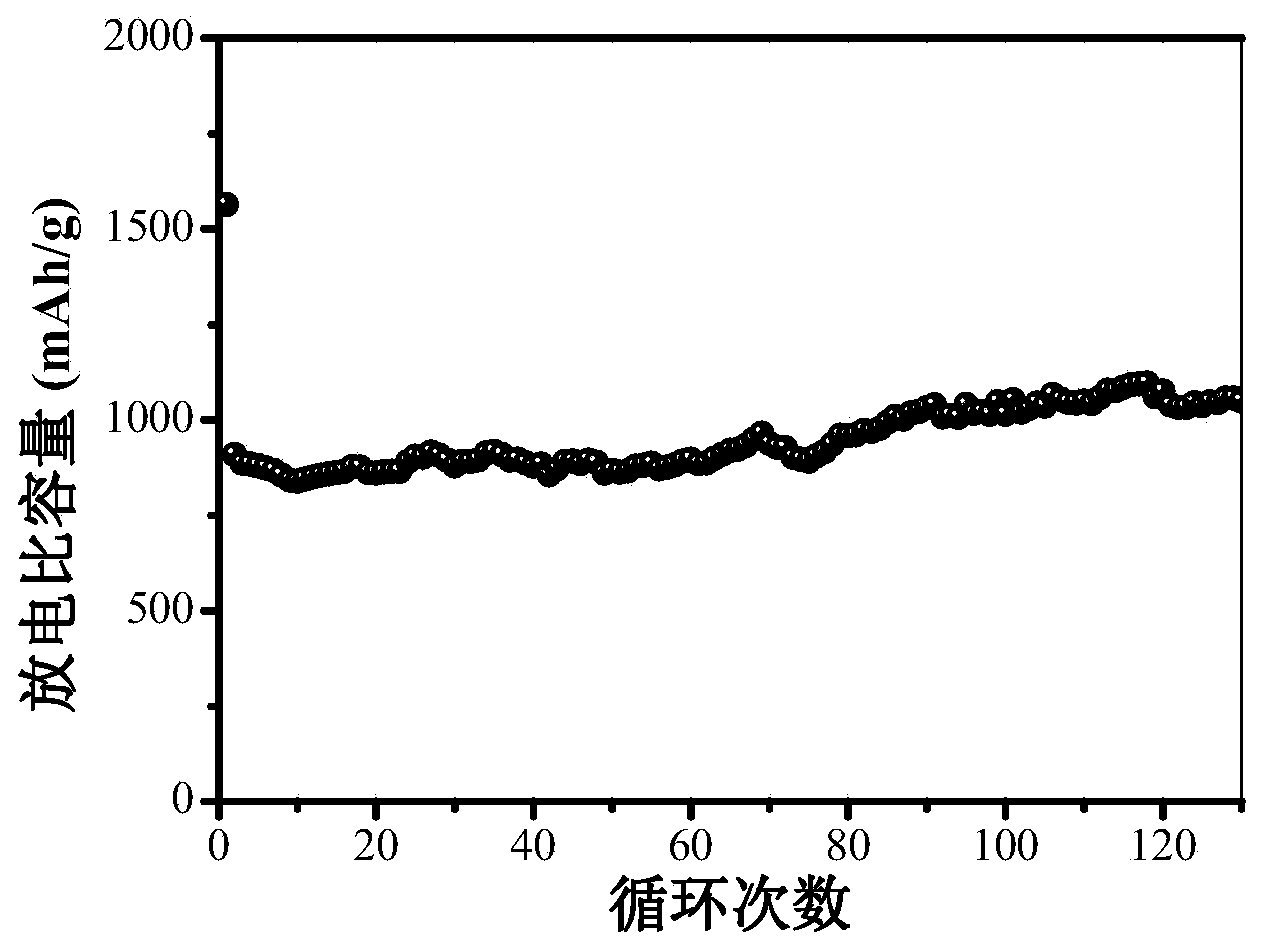
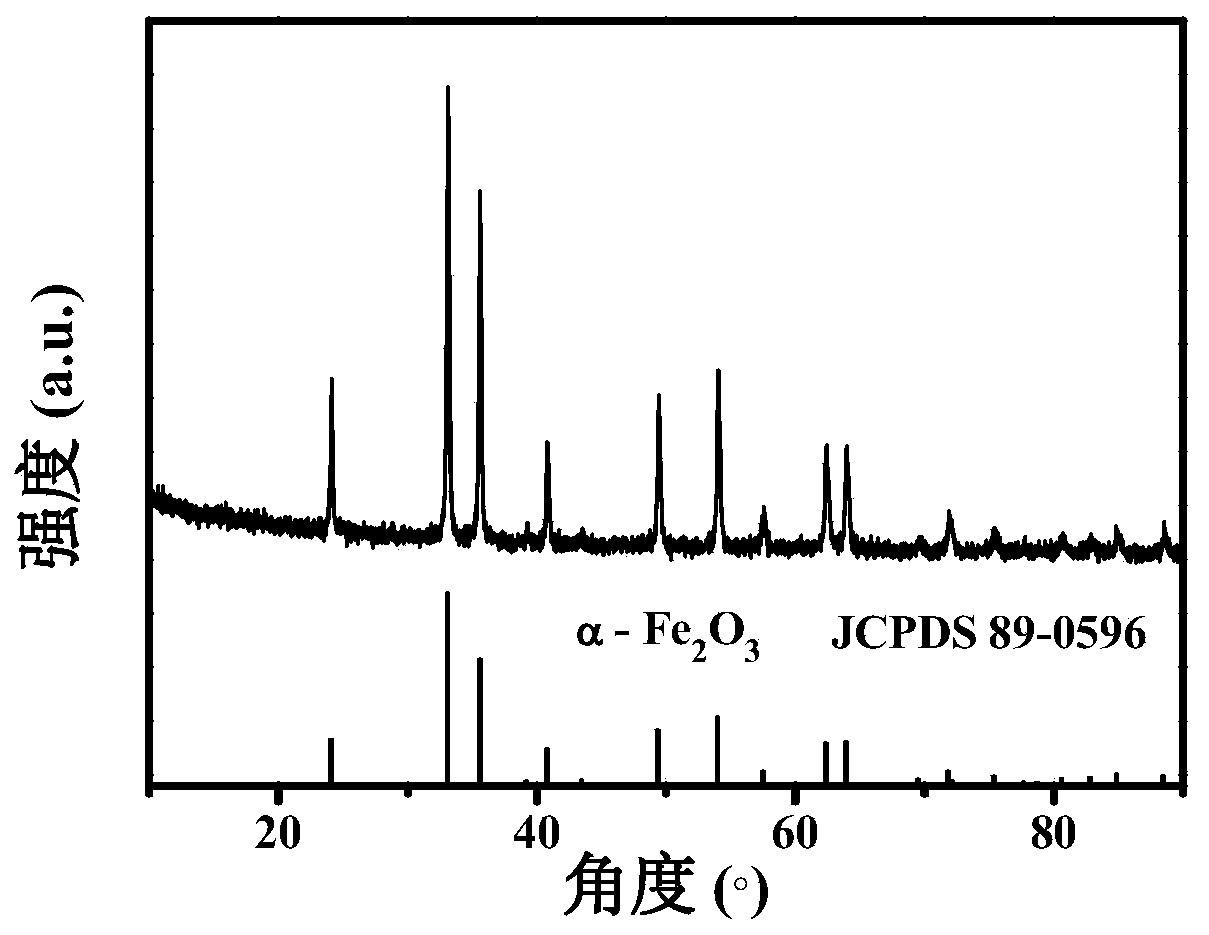
The invention relates to a porous ferric oxide / carbon nanometer plate composite material as well as a preparation method of the porous ferric oxide / carbon nanometer plate composite material and application in preparing a lithium ion battery. The method comprises the following steps of uniformly mixing raw materials including ferrocene and ammonium sulfate in a given mass ratio; calcining the mixture in an inert gas high-pressure reaction kettle, then calcining the mixture in the air at a low temperature to obtain the porous ferric oxide / carbon nanometer plate composite material. The preparation method is simple in process, easy to operate, high in yield and applicable to the mass production. When being used as a lithium ion battery cathode material, the prepared porous ferric oxide / carbon nanometer plate composite material is high in specific capacity and excellent in circulation stability, and the capacity of the material is more than 1000mAh / g after being circulated for 100 times.
Owner:江苏海四达电源有限公司
Preparation method of mannose
InactiveCN102807593AReduce viscosityOvercome the disadvantage of easy moisture absorptionSugar derivativesSugar derivatives preparationSimulated moving bedD-Glucose
The invention discloses a preparation method of D-mannose, which comprises the following steps that glucose is epimerized under a normal pressure or pressurized condition by using ammonium molybdate as catalyst to obtain mixed solution of glucose and mannose; the mixed solution is discolored, desalted and refined and then enters a simulated moving bed 1 for separation and purification to obtain a component A rich in mannose and a component B rich in glucose; the component A enters a simulated moving bed 2 for separation to obtain a component C rich in mannose and a component D rich in glucose; the component B is filtered through a membrane and then enters the step of epimerization; the component D is filtered through a membrane and then enters the simulated moving bed 1 for cyclic separation; and the component C is centrifugally spray-dried to obtain D-mannose finished products. The preparation method of the D-mannose has the advantages of simple steps and easiness in operation. The purity of the D-mannose finished products prepared by adopting the method reaches more than 99 percent and the total yield reaches 53-60.4 percent.
Owner:白心亮
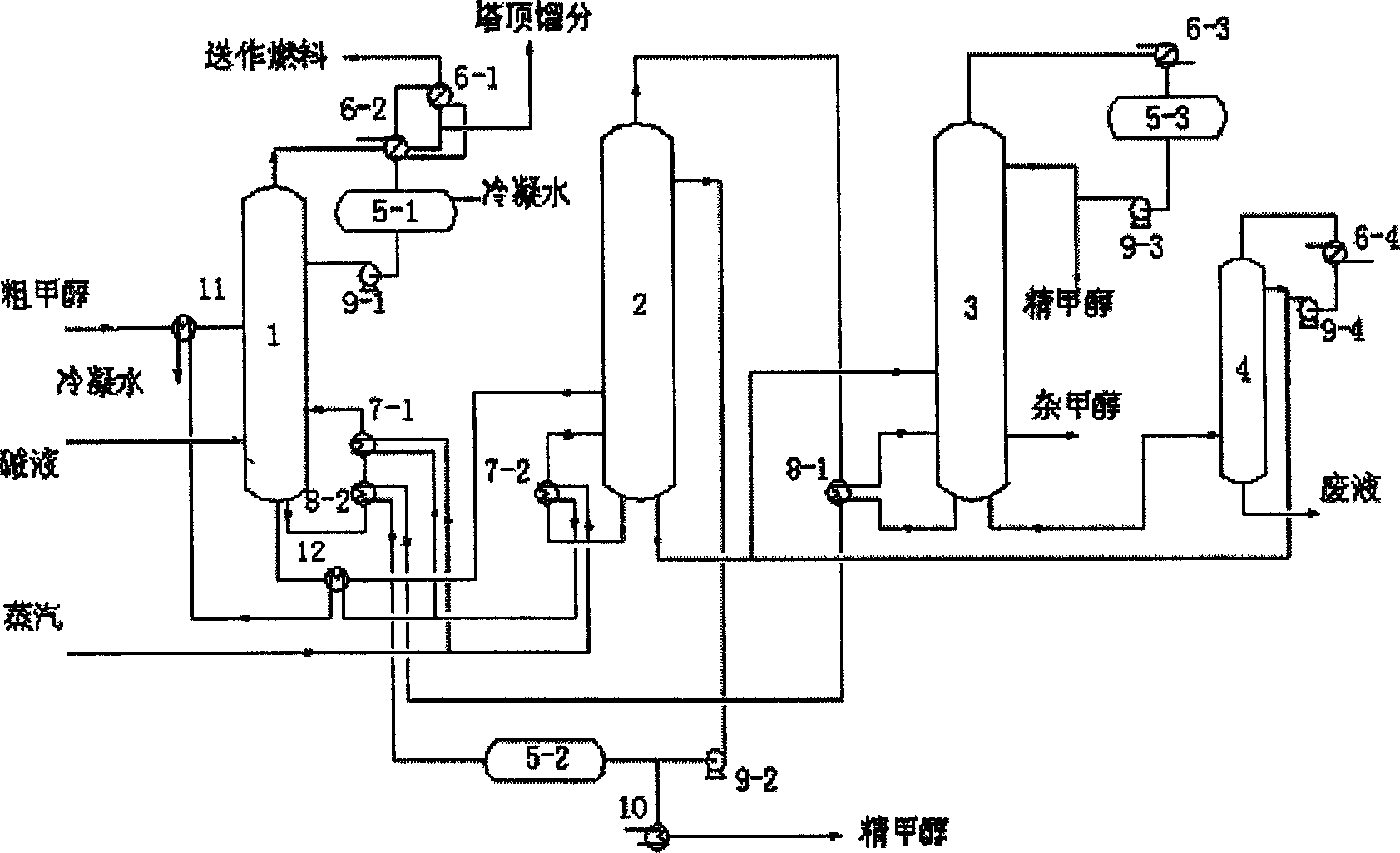
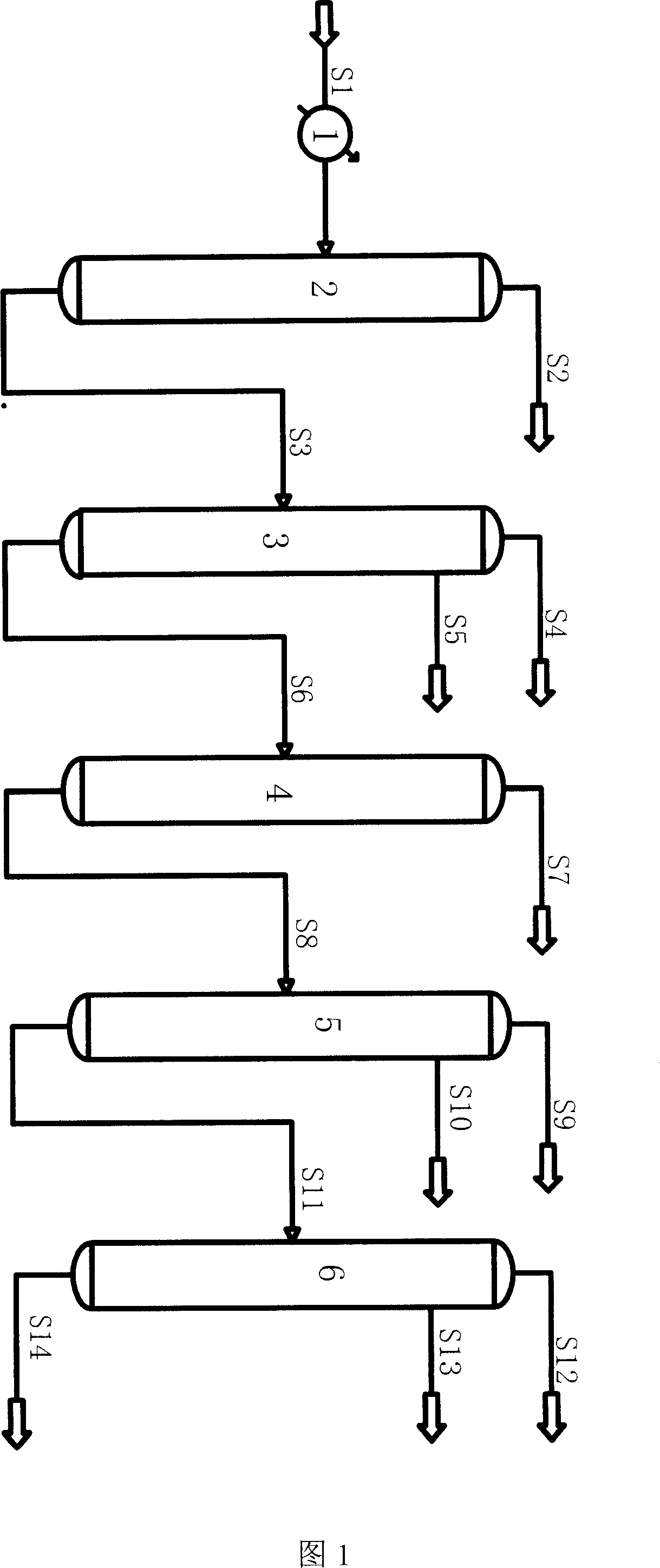
Energy saving equipment of double effect rectification for methanol, and method
InactiveCN1760164AReduce energy consumptionHigh product purityOrganic compound preparationHydroxy compound preparationPipingReboiler
An energy-saving dual-effect methanol rectifying apparatus is composed of preheater for raw methanol, pre-rectifying tower, preheater for pre-rectified methanol, pressurizing tower, ordinary-pressure tower, re-boiling unit, condensing-reboiling unit and methanol recovering tower. The connections between said all units are also diseclosed.
Owner:TIANJIN UNIV
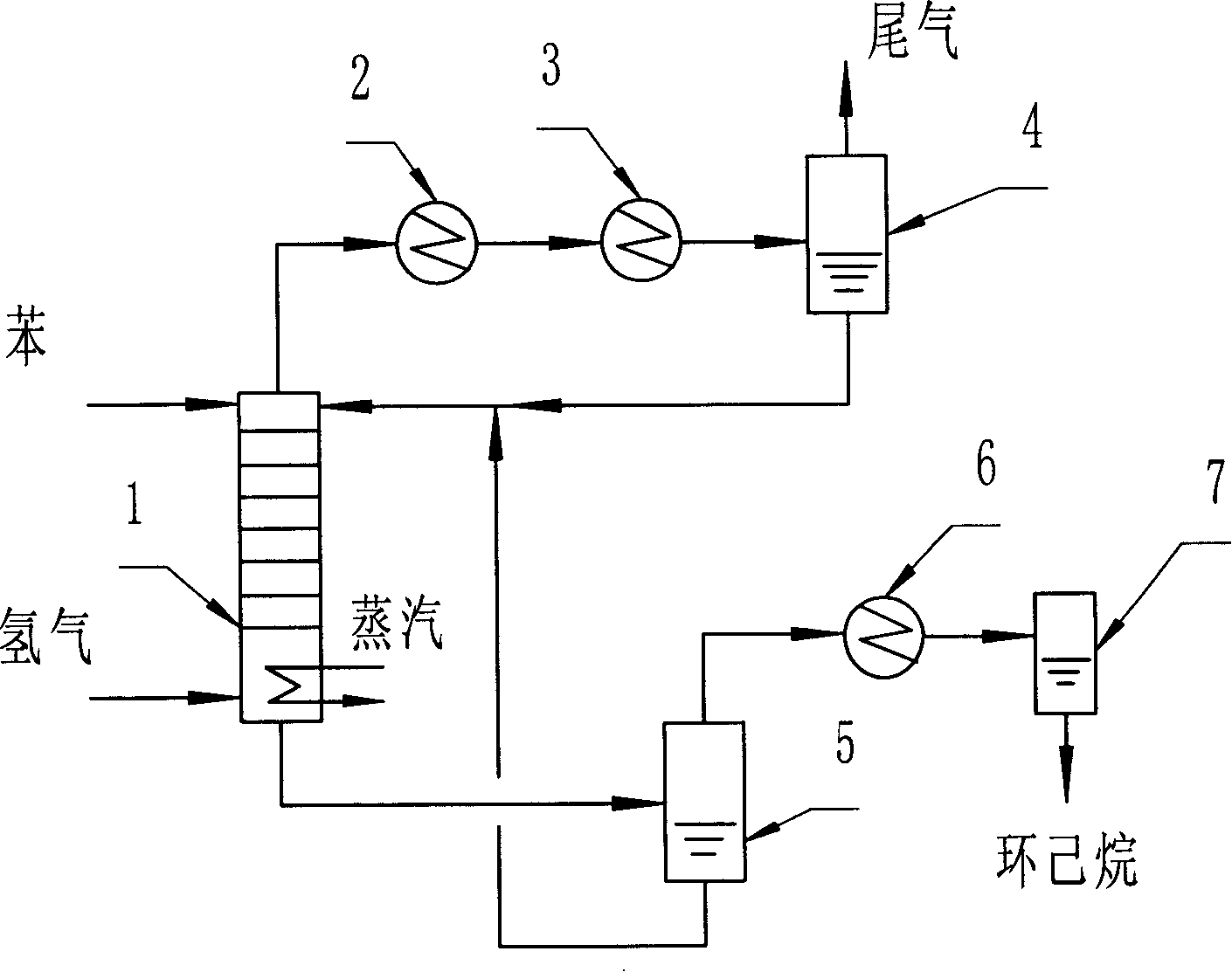
Preparation method of cyclohexane
ActiveCN101205163ASolve the problem of heat transferStable reaction temperatureHydrocarbon by hydrogenationBenzeneHydrogen
A method for preparing a cyclohexane comprises the following steps: a benzene and a hydrogenation catalyst are driven to enter a distillation tower from the tower top and the hydrogen goes into the distillation tower from the tower bottom; the tower bottom is heated by the steam and a reaction in which the benzene hydrogenation is used for generating the cyclohexane is conducted in the tower; the condensate at the tower top refluxes and the discharged materials from the tower bottom are subject to the flash separation so as to obtain the cyclohexane. The method of the invention can lower the content of the methyl cyclopentane in the products and is beneficial to the stabilized operation and the enhancement of the hydrogen utilization ratio and the effective reaction volume of the tower.
Owner:CHINA PETROLEUM & CHEM CORP +1
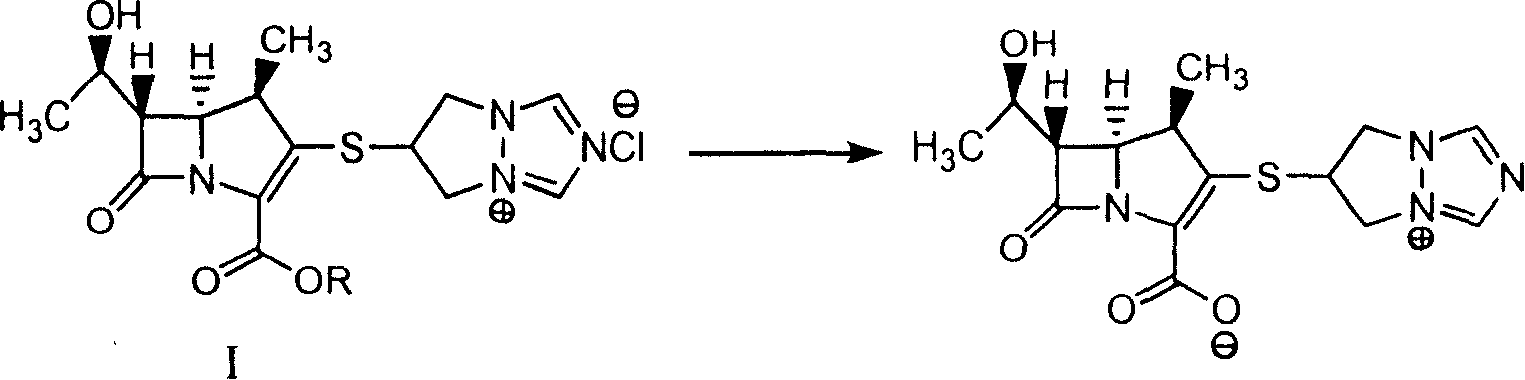
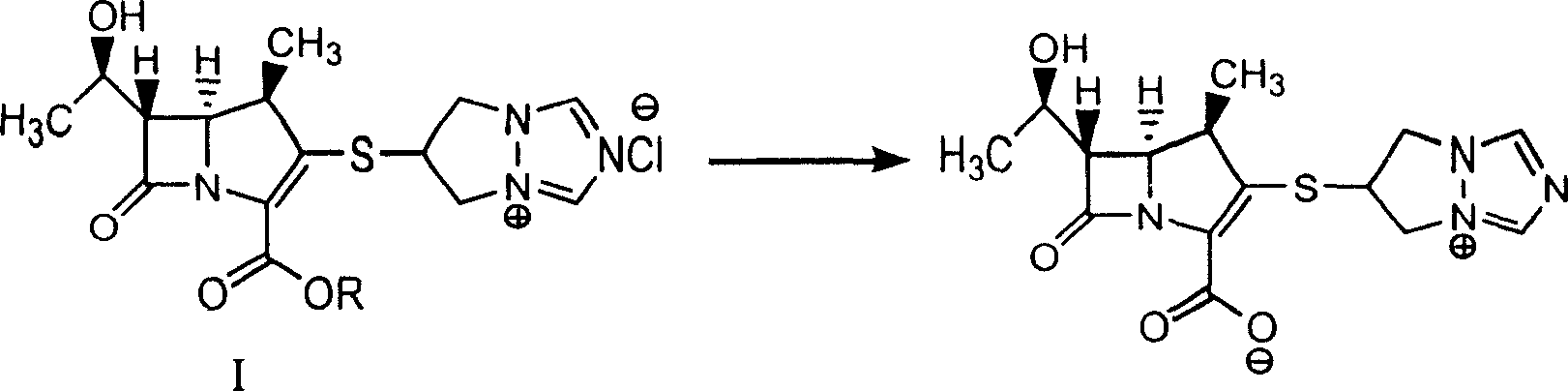
Synthesis method of biapenem
InactiveCN1927867AEasy to operateHigh product purityOrganic chemistryBulk chemical productionBiapenemOrganic solvent
The present invention discloses process of synthesizing Biapenem. The synthesis process includes the following steps: the hydrogenation reaction of the compound shown in expression I to eliminate protecting group inside buffer solution or the mixed liquid of buffer solution and organic solvent in the presence of catalyst of Pd or Pt containing compound; and the subsequent collection of Biapenem from the reaction product. The synthesis process is simple, high in product purity and suitable for industrial production.
Owner:SHANGHAI INST OF PHARMA IND
Processing method for barley 20-30cm seedling green juice powder
InactiveCN105685982AProtect Extraction ColorAvoid layeringNatural extract food ingredientsFiberCentrifugation
The invention relates to a processing method for barley 20-30cm seedling green juice powder. The processing method includes the following steps of 1, pretreatment, wherein sorting and washing are conducted; 2, preparation of barley 20-30cm seedling green juice, wherein shredding, color protecting, freezing, unfreezing, primary juicing, secondary juicing and centrifugation are conducted; 3, preparation of the barley 20-30cm seedling green juice powder, wherein homogenizing and spray drying milling are conducted. Residual fibers in the product can be effectively reduced, the powder can be easily absorbed by the human body, the process difficulty and component damage caused by high temperature are conveniently lowered, and the nutritional value of the product is improved.
Owner:JIANGSU JIANGDAYUAN ECOLOGICAL BIOLOGICAL TECH
Production method for preparing high-purity oligosaccharides by hemicellulose enzymolysis
The invention provides a method for preparing high-purity oligosaccharides by the enzymolysis of hemicellulose containing mannosan, and a product which is a high-effect beta-mannase BM-II produced by fermenting bacillus subtilis TQBm with the preservation No. of CCTCC No: M 211147. Konjac glucomannan, guar gum, sophora bean gum, sesbania gum and other hemicelluloses containing the mannosan are hydrolyzed into relatively small molecule materials, and then are desalted, decolored and deodorized through centrifugal separation, flocculation clarification and ion exchange techniques, large molecule materials in sugar solution are separated by the combination of an ultrafiltration technique and a molecular sieve chromatography technique, and finally the high-purity oligosaccharides is prepared through concentration and drying. The method for preparing the high-purity oligosaccharides disclosed by the invention has the advantages of simple operation, good separation effect, low cost and no chemical pollution, and industrialized production can be realized. The oligosaccharides product disclosed by the invention can obviously proliferate bifidobacteria in the human intestinal tract and otherbeneficial bacteria and obviously reduce enterobacter and other harmful bacteria, has the functions of blood sugar and blood fat reduction and immunoregulation, and is a novel raw material and assistant for gastrointestinal health, blood sugar and fat reducing, immunoregulation and disease resisting and preventing medicines.
Owner:深圳绿天琪生物医药有限公司
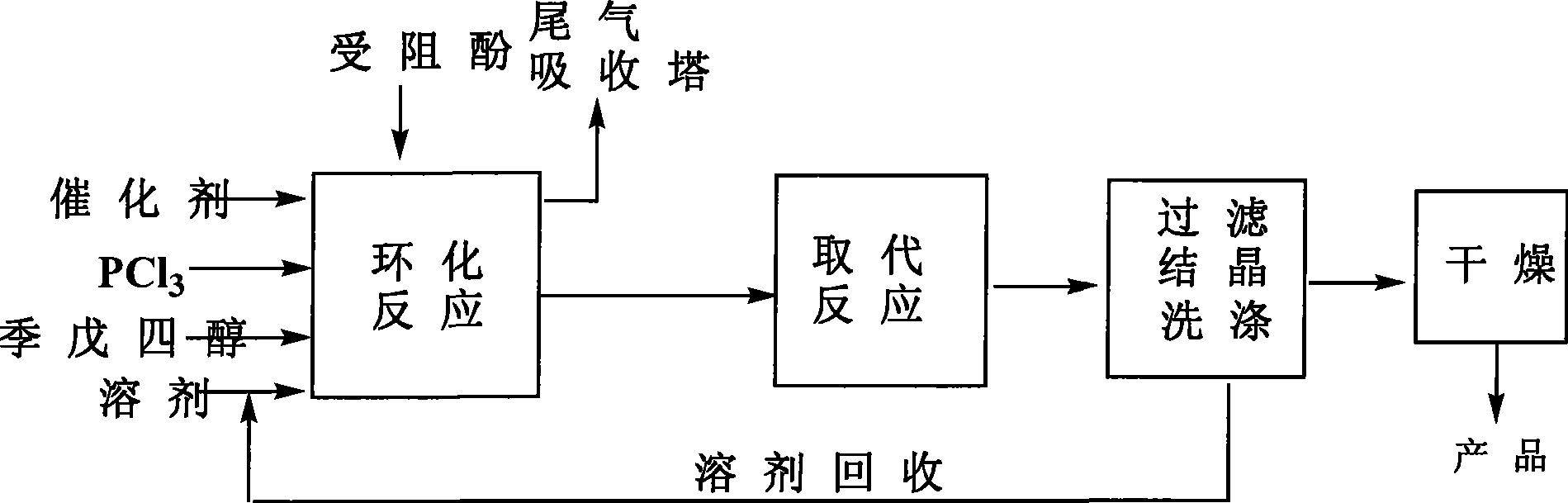
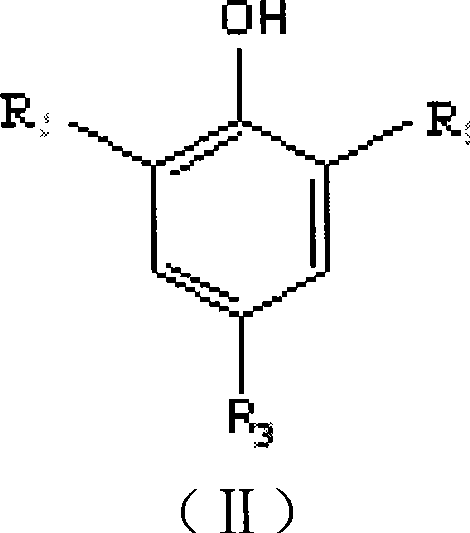
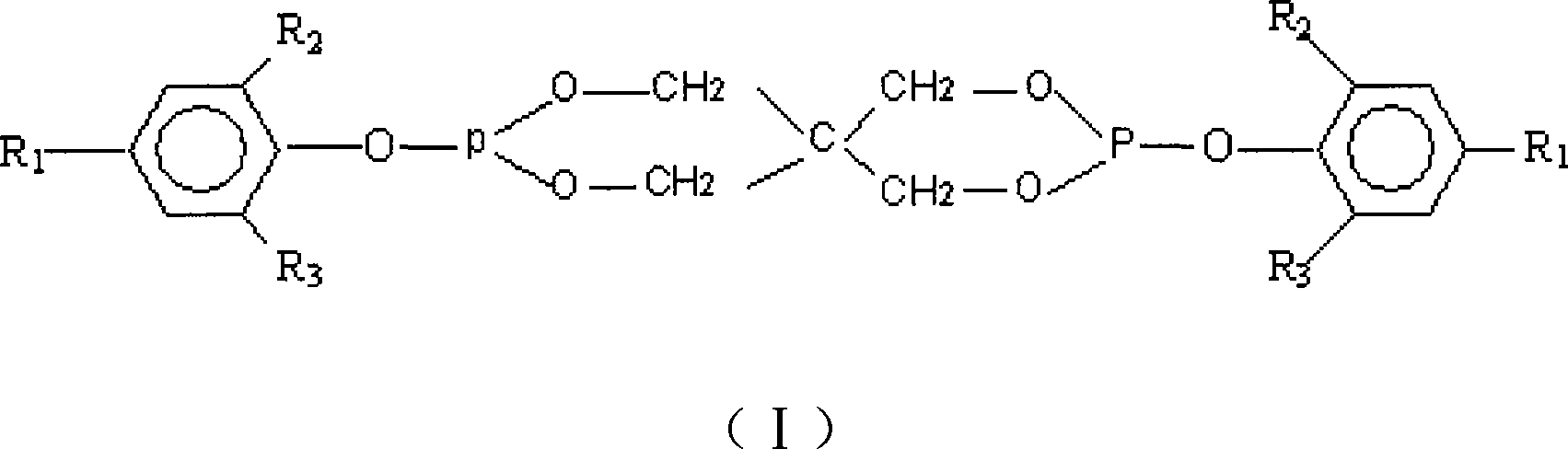
Method for preparing pentaerythritol bis-phosphite antioxidant
ActiveCN101418018AReduce manufacturing costHigh product purityGroup 5/15 element organic compoundsChemical inhibitorsPolypropylenePentaerythritol
The invention relates to a method for preparing an antioxidant. Pentaerythritol and phosphorus trichloride are taken as raw materials and subjected to cyclization to give dichloropentaerythritol diphosphite, and dichloropentaerythritol diphosphite is then subjected to substitution reaction with hindered phenol to give pentaerythritol diphosphite which is subjected to separation, washing, drying, and crushing to obtain the antioxidant. The method adopts a one-pot process to prepare the antioxidant, controls the mixing ratio of raw materials of the cylization, analyzes and supervises the reaction process through 31P NMR, saves separation and purification of intermediates, and reduces production costs. The obtained antioxidant is high in purity, used in polypropylene, and has good high temperature antioxygen property and color protecting capacity.
Owner:CHINA PETROLEUM & CHEM CORP +1
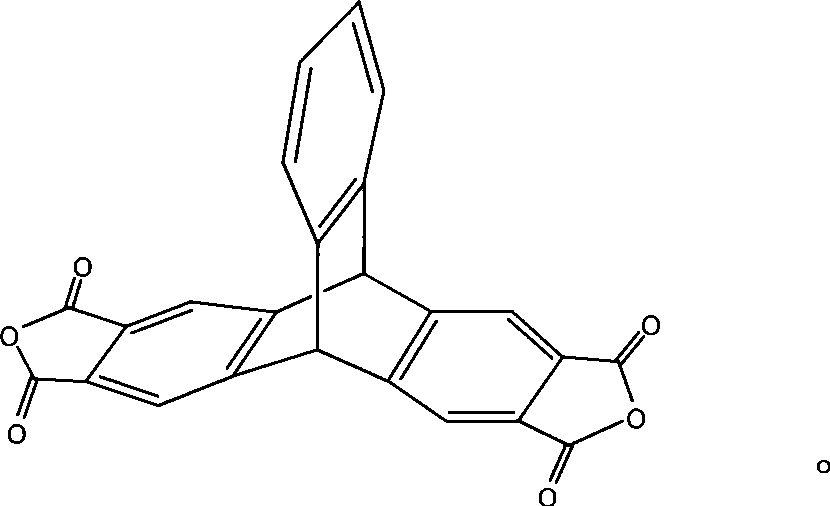
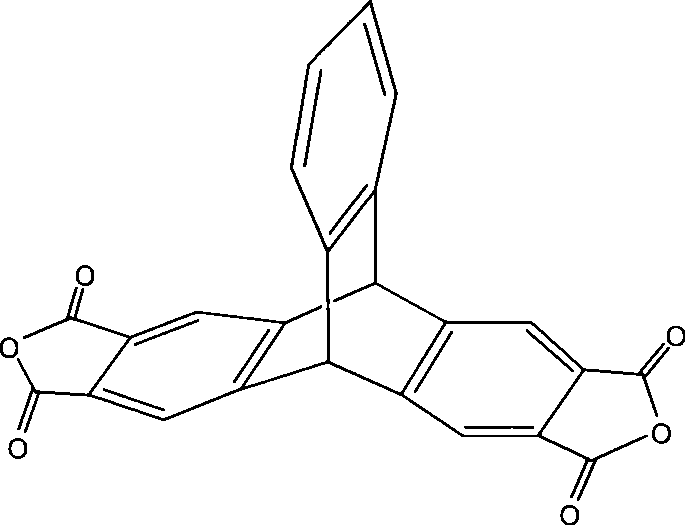
2,3,6,7-tetracarboxylic dianhydride triptycene and method for synthesizing the same
The invention discloses 2,3,6,7-tetracarboxylic dianhydride triptycene and a synthetic method thereof. The synthetic method comprises the following steps: preparing 2,3-dimethyl anthracene by a series of reaction of oxylene and phthalic anhydride which are cheap and easily obtained, and further reacting with 2-amino-4,5-hemellitic acid to obtain 2,3,6,7-tetramethyl triptycene; obtaining high-purity 2,3,6,7-tetramethyl triptycene by simple and rapid separation on columns, further oxidizing by potassium permanganate to obtain 2,3,6,7-tetracarboxylic acid triptycene, then obtaining the 2,3,6,7-tetracarboxylic dianhydride triptycene by dehydration. The method has the advantages of cheap and easily-obtained raw material, simple preparation method, high product purity which is up to 98% and the like, and the obtained product dianhydride monomer is fit for preparation of polyimide materials with good heat resistance and dissolubility.
Owner:HUAQIAO UNIVERSITY
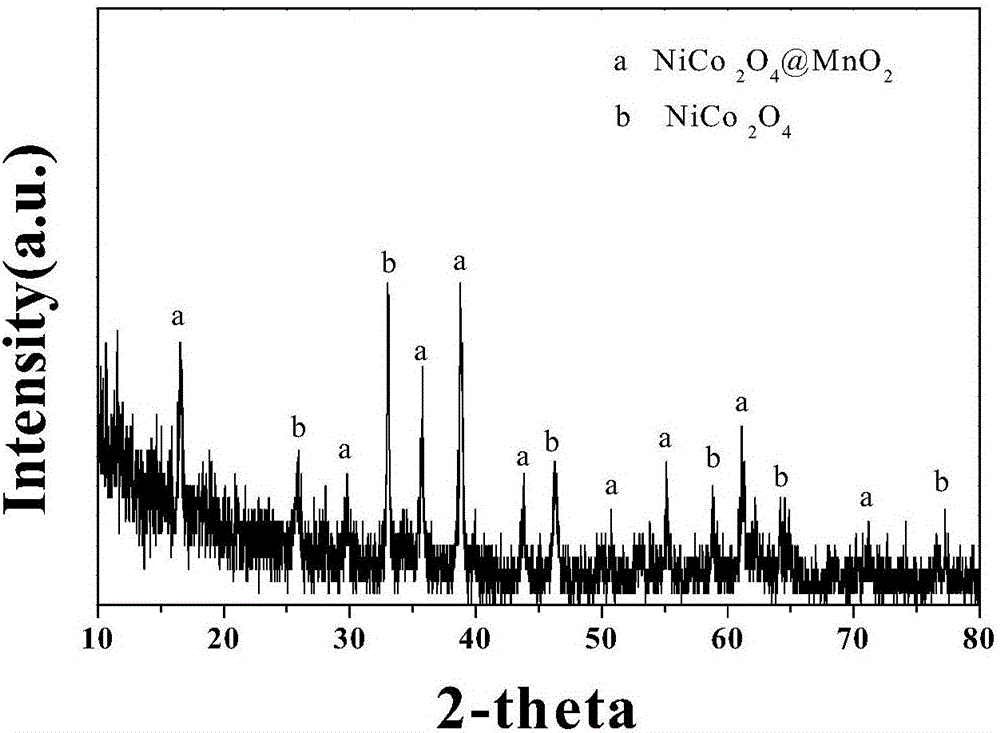
Nickel cobalt oxide<@>manganese dioxide nuclear shell heterostructure nanowire array grown on carbon cloth and preparation method and application thereof
InactiveCN106373785AHigh product purityGood dispersionHybrid capacitor electrodesHybrid/EDL manufactureCobalt oxideCapacitance
The invention provides a nickel cobalt oxide<@>manganese dioxide nuclear shell heterostructure nanowire array grown on carbon cloth and a preparation method and application thereof. Compared with the prior art, the product of the preparation method provided by the invention is high in purity, good in dispersibility, good and controllable in crystalline form, low in production cost and good in repeatability. The prepared nickel cobalt oxide<@>manganese dioxide nuclear shell heterostructure nanowire array grown on the carbon cloth can serve as an electrode material of a supercapacitor directly, long cycling stability, large specific capacitance, high energy density and power density are realized, and the nanowire array has a potential application value in the aspect of energy storage.
Owner:ANHUI NORMAL UNIV
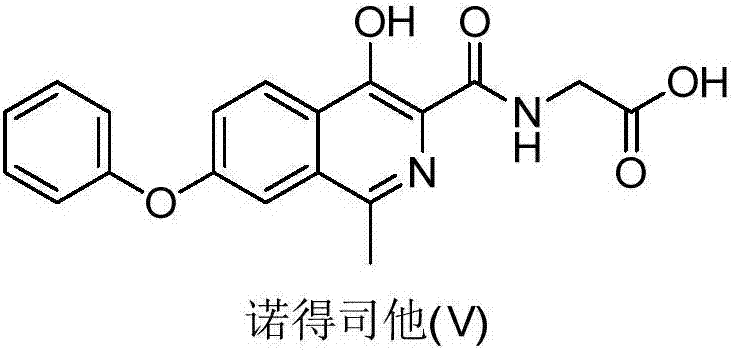
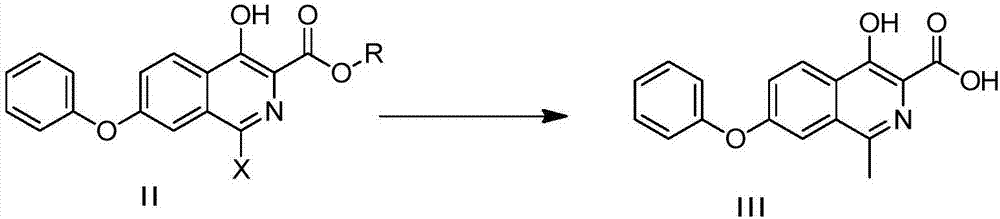
Preparation method of roxadustat
ActiveCN107954931ARaw materials are easy to getHigh product purityOrganic chemistryBulk chemical productionMedicinal chemistryRoxadustat
The invention relates to preparation method of roxadustat, which is [(4-hydroxy-1-methyl-7-phenoxyisoquinoline-3-formyl) amino] acetic acid. The preparation method has low cost, simple method and goodsafety and is suitable for industrial production.
Owner:SHANGHAI PHARMA GRP QINGDAO GROWFUL PHARMA CO LTD
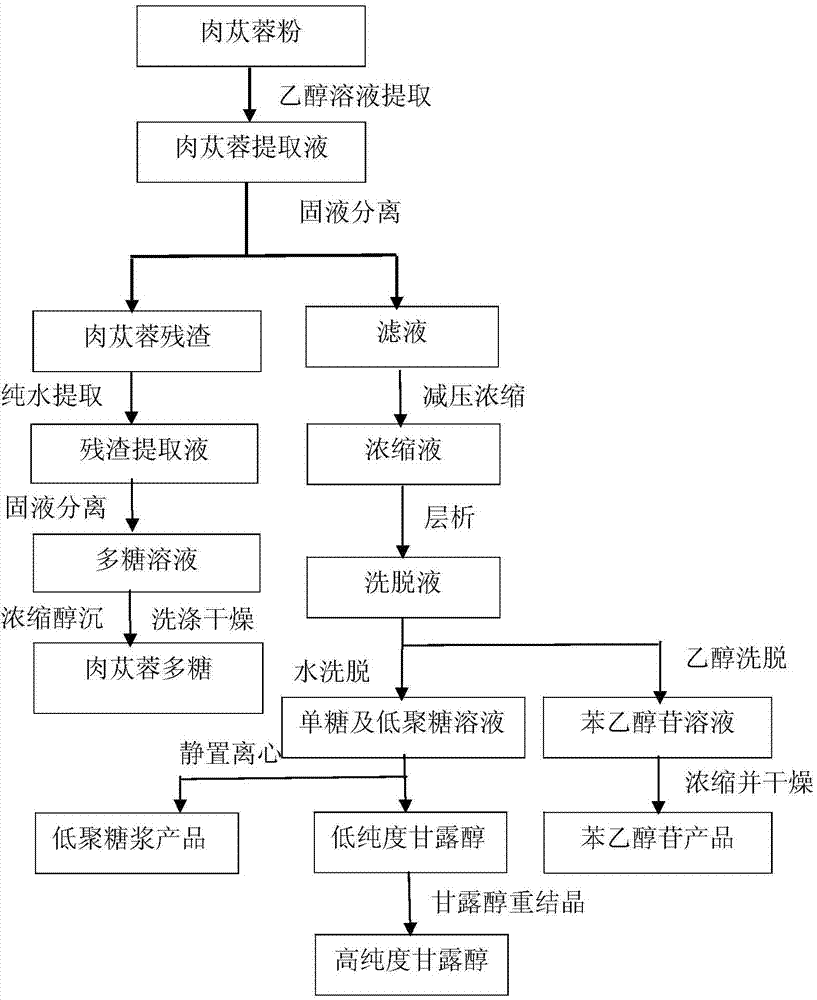
Method for preparing phenylethanoid glycoside, mannitol, oligosaccharide syrup and polysaccharide from broomrape
The invention relates to a method for simultaneously preparing phenylethanoid glycoside, mannitol, oligosaccharide syrup and polysaccharide from broomrape, which comprises the following steps: extracting monosaccharide, oligosaccharide and phenylethanoid glycoside in broomrape by using an ethanol water solution, separating the phenylethanoid glycoside and saccharide with a macroporous adsorbent resin column, carrying out concentration and drying on the phenylethanoid glycoside solution obtained by elution to obtain the product, concentrating the saccharide under reduced pressure, crystallizing at normal temperature, separating the oligosaccharide and mannitol, recrystallizing the mannitol to obtain the high-purity mannitol, centrifugating the clear solution to obtain the oligosaccharide syrup, extracting the polysaccharide from residues by using pure water, and carrying out alcohol precipitation to collect the polysaccharide. The method enhances the comprehensive utilization ratio of the broomrape resources, simplifies the production technique, develops the phenylethanoid glycoside, mannitol, oligosaccharide syrup and polysaccharide products simultaneously, increases the economic added value of the broomrape product, and has favorable industrial production and market development prospects.
Owner:INST OF PROCESS ENG CHINESE ACAD OF SCI
Synthesis method for 2,3,6,7-triptycene tetracarboxylic dianhydride
InactiveCN102617587AHigh yieldHigh product purityOrganic chemistryChromatographic separationAcetic anhydride
The invention discloses a synthesis method for 2,3,6,7-triptycene tetracarboxylic dianhydride. The method comprises the following steps of: 1) preparing 2,3,6,7-tetramethylanthracene from o-xylene and benzyl alcohol or methylene chloride under the catalysis of anhydrous aluminum trichloride; 2) diazotizing the 2,3,6,7-tetramethylanthracene, anthranilic acid and amyl nitrite to generate a crude product 2,3,6,7-tetramethyl triptycene; 3) performing chromatographic separation on the crude product 2,3,6,7-tetramethyl triptycene, and flushing the separated 2,3,6,7-tetramethyl triptycene by using a mixed eluting agent to obtain 2,3,6,7-tetramethyl triptycene; 4) oxidizing the 2,3,6,7-tetramethyl triptycene in a mixed solvent consisting of potassium permanganate, pyridine and water under a reflux condition to generate 2,3,6,7-triptycene tetracarboxylic acid; and 5) refluxing and dehydrating the 2,3,6,7-triptycene tetracarboxylic acid in acetic anhydride to generate the 2,3,6,7-triptycene tetracarboxylic dianhydride. The method has the characteristics of readily available and low-cost raw materials, high yield and high product purity.
Owner:HUAQIAO UNIVERSITY
Process for preparing spherical high purity alumina
InactiveCN1631788AUniform particle size distributionHigh product purityAluminium oxide/hydroxide preparationEnergy efficient lightingChemistrySapphire
A method for preparing a spherical alumina of high purity, relates to the preparation of alumina of high purity which is especially suitable to the used in the fields such as compact fluorescent lamp, illuminating material for PDP and LED and transparent ceramics, monocrystal of sapphire, monocrystal of ruby and polishing of paint of car. Its characteristics are that aluminum ammonium sulfate and ammonium hydrogen carbonate are used as the raw material to react into nanometer structure spherical aluminum ammonium carbonate AACH in the container with agitation, and the nanometer structure spherical ª†-Al2O3 and ª‡- Al2O3 are obtained by filtration, washing and roasting. The spherical alumina of high purity has remarkable liquidity, high mechanic strength, infragile and keeping its shape in the course. The invention has high purity, remarkable quality, few requirements for technical condition, without pollution and has low cost.
Owner:GUIZHOU BRANCH CHINA ALUMINUM IND
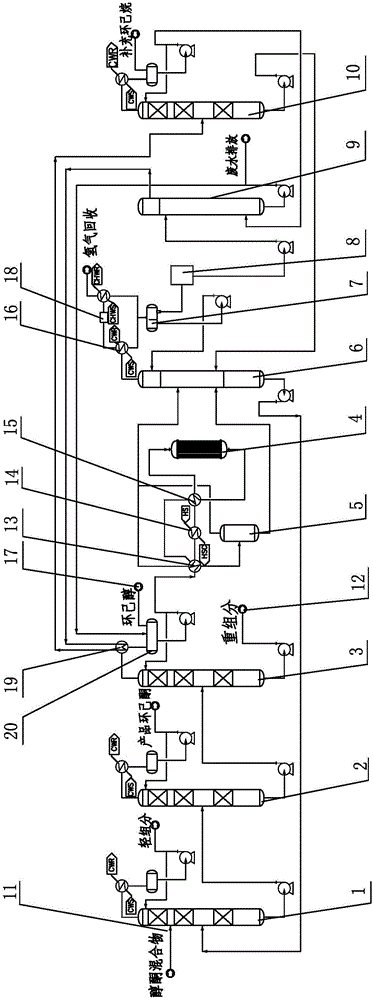
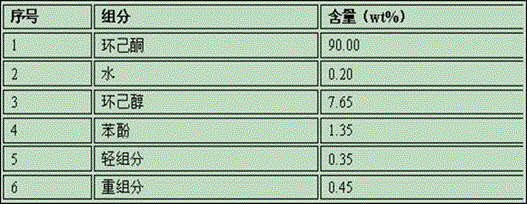
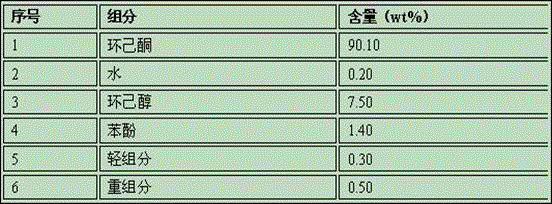
Method and system for refining cyclohexanone and recycling cyclohexanol
ActiveCN106083544AHigh product purityLess impuritiesOrganic compound preparationHydroxy compound separation/purificationKetoneCyclohexanone
The invention discloses a method and a system for refining cyclohexanone and recycling cyclohexanol. The method is characterized by comprising the following steps: sending an alcohol ketone mixture into a light-component removal tower for removing light components, sending the material discharged from the tower bottom into a cyclohexanone product tower, acquiring a high-purity cyclohexanone product from the tower top and a crude product mainly comprising cyclohexanol from the tower bottom, then sending the crude product into a cyclohexanol recovery tower, and acquiring cyclohexanol from the tower top; mixing the acquired cyclohexanol with proper amount of water, heating and sending into a cyclohexanol dehydrogenation reactor for dehydrogenation, so as to obtain a mixed product containing cyclohexanol, cyclohexanone and water, performing two-stage cooling on the dehydrogenated product, then performing gas-liquid separation, sending the gas phase and the liquid phase into a dewatering tower for dewatering processing, sending oil-containing wastewater removed at the tower top to a cyclohexane extraction tower, sending an obtained upper-layer oil phase to an alcohol-ketone recovery tower, and sending an oil phase containing cyclohexanone and cyclohexanol and obtained at the bottom of alcohol-ketone recovery tower to a dewatering tower for cycling. The invention comprises the advantages that the product cyclohexanone is high in purity, cyclohexnoal is efficiently converted into cyclohexanone at a high yield, and the system is high in energy integration degree and low in energy consumption.
Owner:SEDIN NINGBO ENG
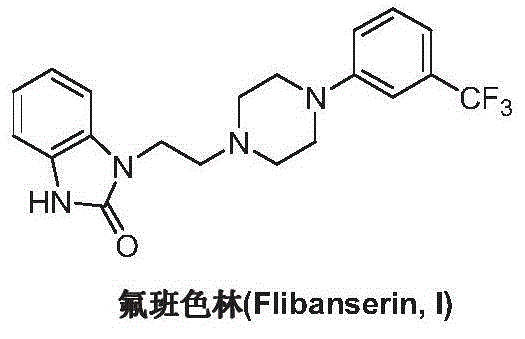
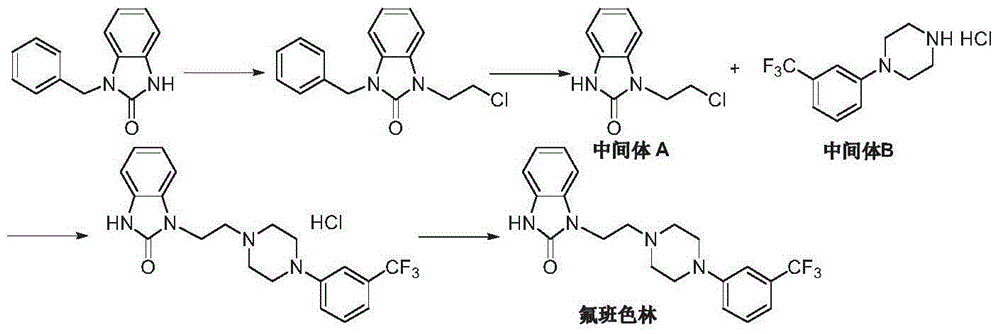
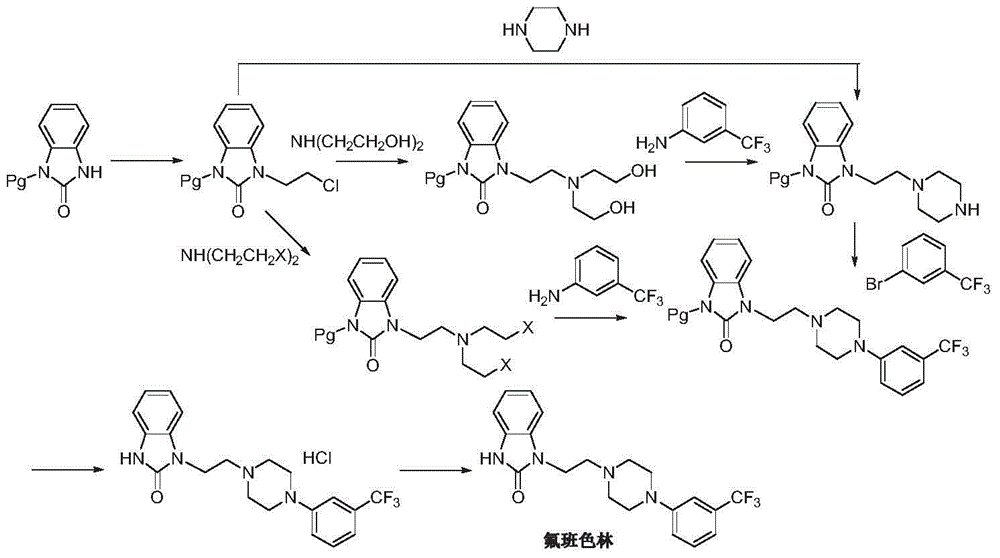
Preparation method of flibanserin
ActiveCN104926734AThe preparation process is convenientHigh yieldOrganic chemistryOrtho-nitroanilineHalogen
The invention discloses a preparation method of flibanserin. The preparation method uses trifluoromethylbenzene, triamine (2-halogen ethyl) and ortho-nitroaniline which are easy to obtain as raw materials and adopts classical elementary reactions such as cyclization, substitution, reduction and condensation, so that the flibanserin is prepared. The raw materials of the preparation method are easy to obtain, the technology is succinct, the yield is high, the preparation method is economical and environment-friendly, and a new preparation way is provided for the industrial production of the flibanserin.
Owner:SUZHOU LIXIN PHARMA
Energy-saving technique for producing high-purity pentane and pentane foaming agent
InactiveCN1919809AHigh product purityHigh purityDistillation purification/separationSource materialDirect production
Owner:天津天大天海化工新技术有限公司
Method for preparing nano NaY molecular sieve
InactiveCN101177281AHigh product purityWell dispersedFaujasite aluminosilicate zeoliteSodalitesChemistryMolecular sieve
The invention relates to a preparation method of nanometer NaY molecular sieve, which is as follow: under the hypergravity condition of the revolving bed, performing guiding agent preparation, mother solution preparation, and mixing of guiding agent and mother solution, and the sequent crystallization of the reaction product. The invention enables to shorten the crystallization reaction time, simplify the technological process and prepare the high-quality nanometer NaY molecular sieves. The nanometer NaY molecular sieve catalyst prepared through the method can be used for the catalytic cracking of the heavy oil. Compared with the normal industrial molecular sieve, the nanometer NaY molecular sieve has the advantages of great increase of gasoline productivity, diesel fuel productivity, clean oil yield and total-liquid yield, improvement of the product selectivity, and obvious improvement of the product distribution.
Owner:BEIJING UNIV OF CHEM TECH
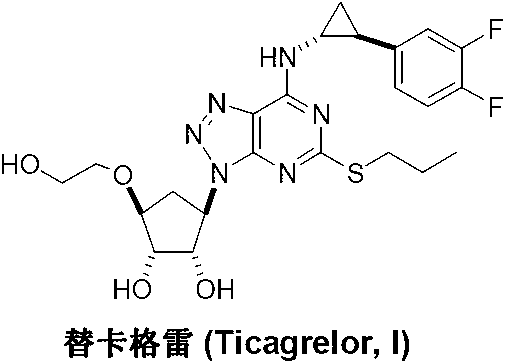
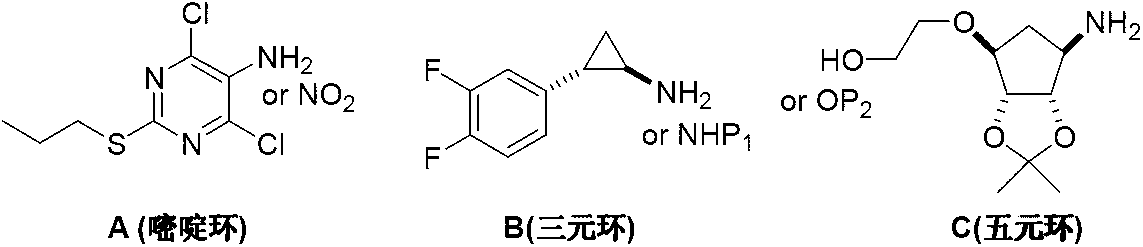
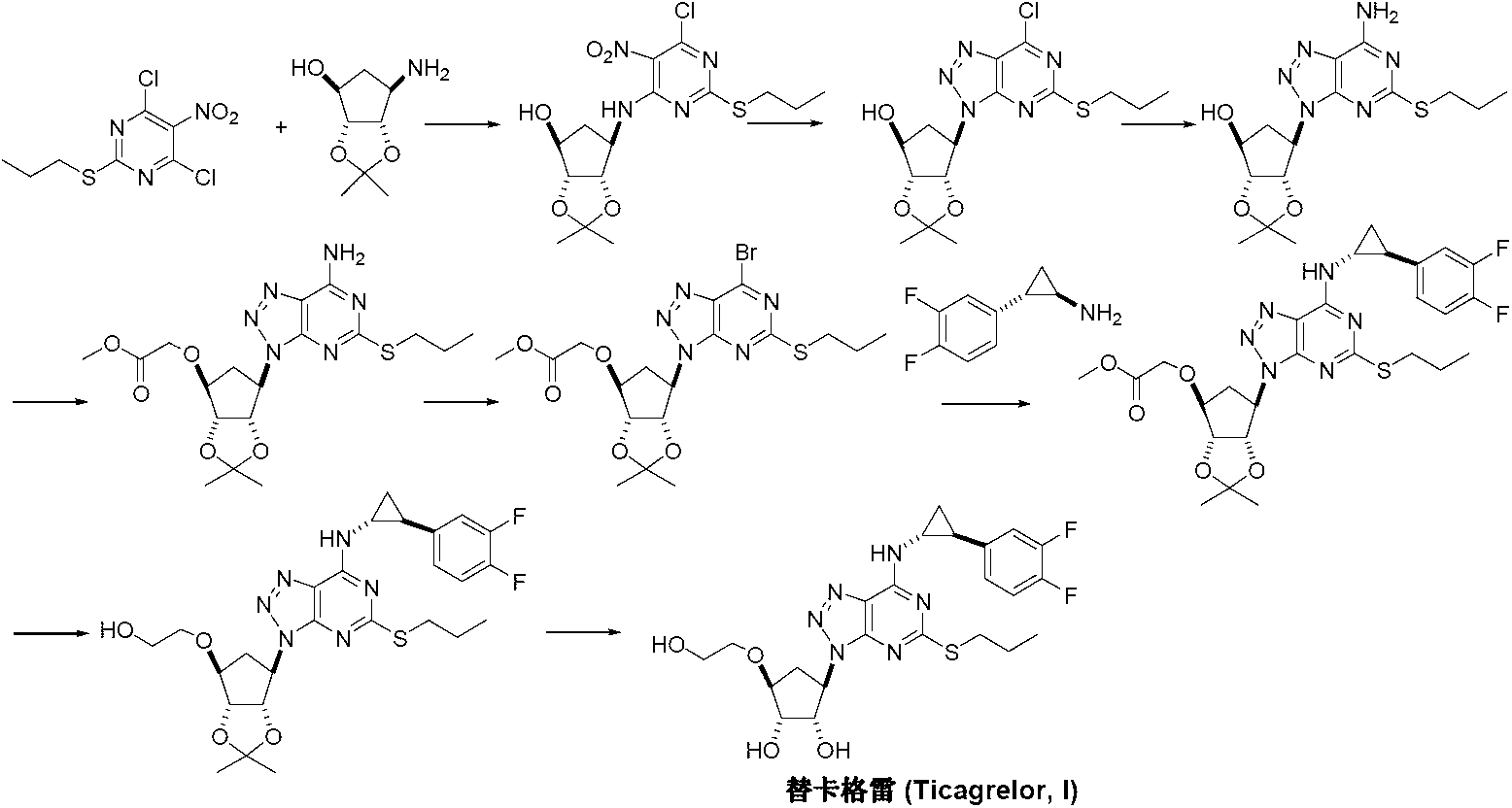
Preparation method of ticagrelor
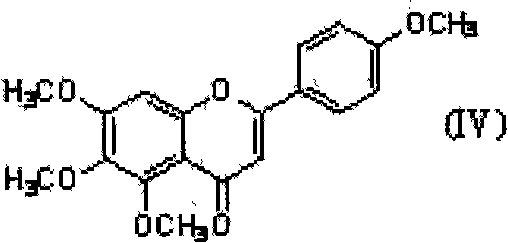
The invention discloses a preparation method of ticagrelor. The preparation method comprises the following steps of: carrying out cyclization reaction between 5-amido-1,4-bis-substituent group-1,2,3-triazole (II) and a sulfur-containing cyclizing agent (III) to obtain 9-substitution-2-sulfo-6-oxo-8-azapurine (IV); carrying out substitution reaction between an intermediate (IV) and halogenated propane (V) to generate 9-substitution-2-propyl sulfydryl-6-oxo-8-azapurine (VI); chloridizing an intermediate (VI), and carrying out amination reaction between the intermediate (VI) and trans-(1R, 2S)-2-(3,4-difluorophenyl) rolicyprine (VII) to generate 9-substitution-6-amido substituendum-2-propyl sulfydryl-8-azapurine (VIII); and removing the propylidene acetonyl out of an intermediate (VIII) to obtain ticagrelor (I). The preparation method has the advantages of simple process and high chemical and chiral purity and provides a new preparation way for the industrialization of the ticagrelor.
Owner:鄄城县人民医院
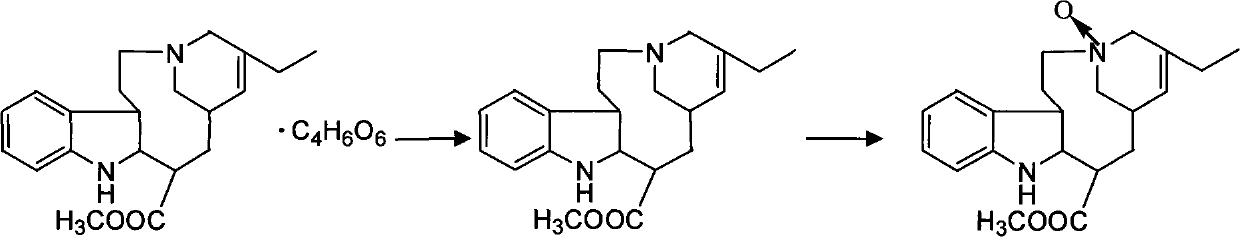
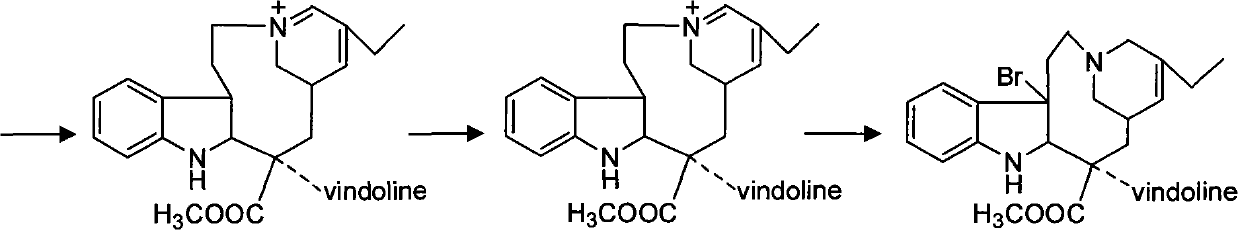
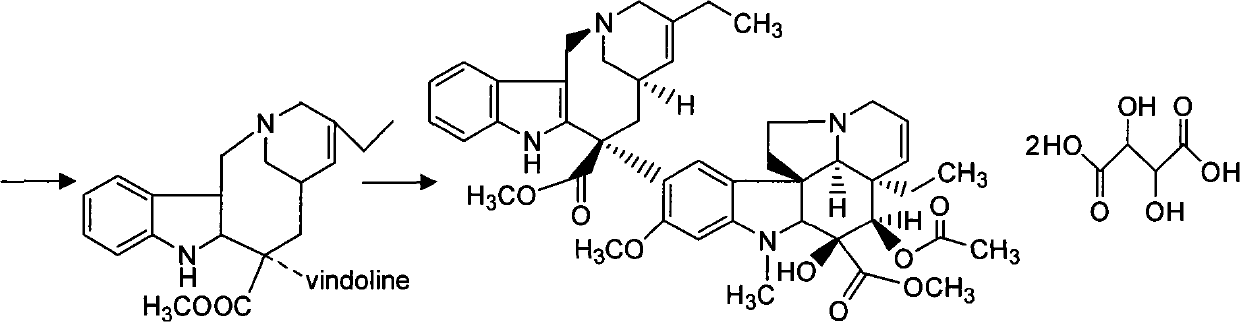
Preparation method for vinorelbine tartrate
ActiveCN102199165AReduce manufacturing costLow impurity contentCarboxylic acid salt preparationTrifluoroacetic anhydrideSodium borohydride
The invention discloses a preparation method for vinorelbine tartrate; the preparation method comprises the following steps of: taking tartrate vinblastine as a starting material; using ammonia water for dissociating the starting material, reacting the dissociated starting material with the metachloroperbenzoic acid so as to obtain vinblastine-N oxide, then reacting the vinblastine-N oxide with vindoline and trifluoroacetic anhydride, carrying out the reduction through sodium borohydride so as to obtain dehydrated vinorelbine, treating the dehydrated vinorelbine so as to obtain vinorelbine, and finally salifying the vinorelbine with tartaric acid so as to obtain the vinorelbine tartrate. The preparation method has the advantages of low cost, few side reaction, easily controlled operation process, higher yield and product purity and wide industrialized application prospect.
Owner:HUBEI HONCH PHARMA
Method for preparing silica through coal gangue aluminum extraction waste slag
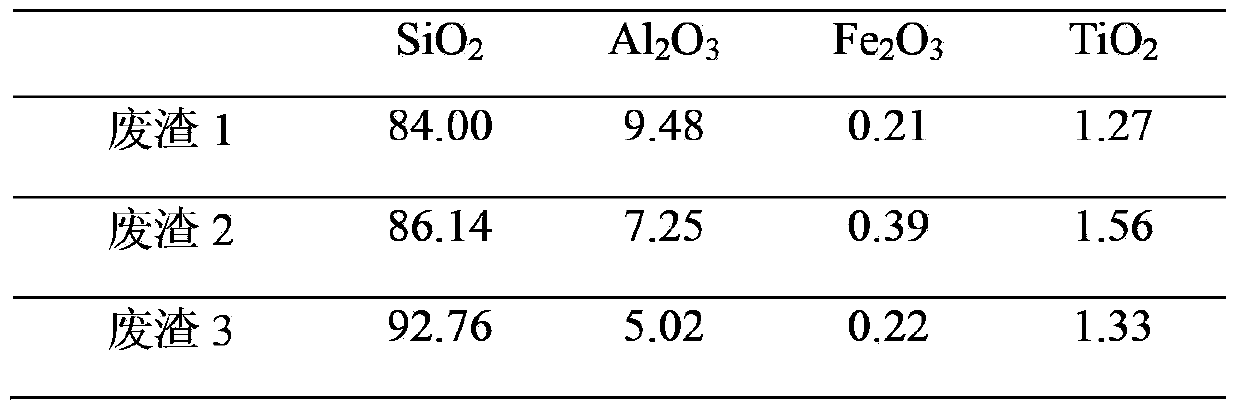
InactiveCN103420386AReaction conditions are easy to controlHigh product puritySilicaSolid waste disposalCarbonationCoal gangue
The invention relates to a method for preparing silica through coal gangue aluminum extraction waste slag. The method comprises the following steps: firstly, mixing the coal gangue aluminum extraction waste slag with quartz sand while enabling the SiO2 content of the mixture to be larger than or equal to 90% and the Al2O3 content of the mixture to be smaller than 6%; then according to the molar ratio of 1: (0.3-1) of SiO2 to sodium carbonate, adding the sodium carbonate to the mixture; grinding the mixture into particles with diameters of 50-200 [mu]m; placing the particles in a converter at 750-830 DEG C for fusing reaction for 1-5 hours, so as to obtain coarse sodium silicate particles; dissolving the coarse sodium silicate particles in a high-pressure reaction kettle to obtain a sodium silicate solution; diluting the sodium silicate solution to 5-15 baume degrees; leading CO2 produced during the process of the fusing reaction to the diluted sodium silicate solution for carbonation, so as to obtain the silica. The solubility of the solid sodium silicate prepared according to the method can reach up to 90%. The silica conforms to the national standard; the oil absorption rate of the silica is larger than 3.1 ml / g. Through the adoption of the method, the comprehensive utilization rate of coal gangue is improved, CO2 produced during the process of the fusing reaction is eliminated by dissolving, and emission of three wastes is reduced.
Owner:SHANXI UNIV
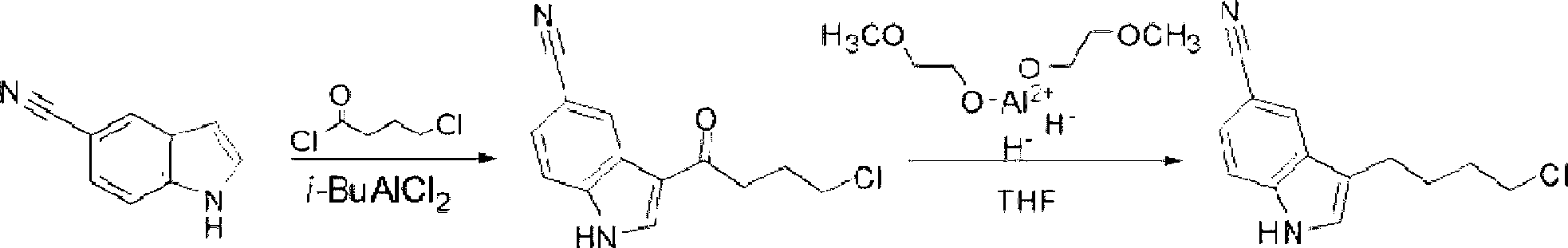
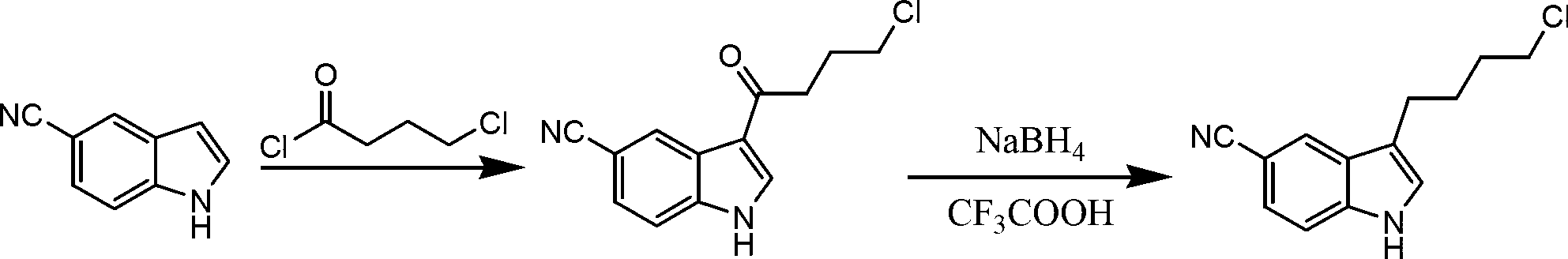
Preparation method of 3-(4-chlorobutyl)indole-5-formonitrile
InactiveCN103058912AHigh yieldHigh product purityOrganic chemistryTrifluoroacetic acidEconomic benefits
The invention relates to a preparation method of 3-(4-chlorobutyl)indole-5-formonitrile. 3-(4-chlorobutyl)indole-5-formonitrile is an important intermediate for synthesis of vilazodone hydrochloride. 5-cyanoindole and 4-chlorobutyryl chloride as raw materials undergo a Friedel-Crafts acylation reaction and then the product is reduced by sodium borohydride / trifluoroacetic acid into 3-(4-chlorobutyl)indole-5-formonitrile. The preparation method utilizes the cheap and easily acquired reagents, has a high yield and high product purity, is simple and reliable, and has a low production cost, a large implement value and social and economic benefits.
Owner:SHANDONG ZOUPING DAZHAN NEW MATERIALS
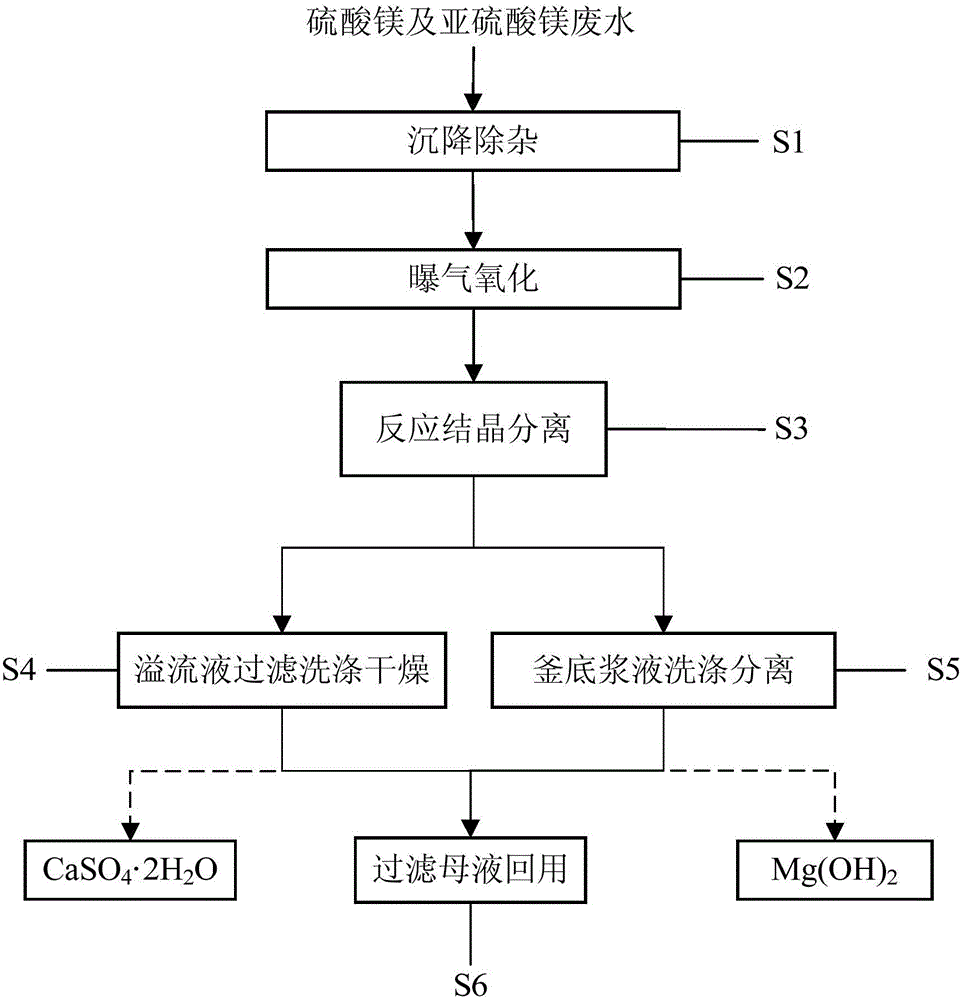
Method for treating magnesium sulfate and magnesium sulfite wastewater by virtue of lime method
ActiveCN105858692AHigh calcium and magnesium separation coefficientHigh product purityCalcium/strontium/barium sulfatesSolution crystallizationMagnesium carbonate hydroxideChemistry
The invention discloses a method for treating magnesium sulfate and magnesium sulfite wastewater by virtue of a lime method. Technical steps comprise settling purification, aeration oxidization, reactive crystallization separation, overflow liquid filtering, washing and drying, kettle-bottom slurry washing and separation and filtered stock recycling. Purified and oxidized wastewater and lime slurry are continuously added into a reaction crystallizer respectively, and a reactive crystallization condition is controlled to bond sulfate ions and calcium ions to form and settle calcium sulfate dihydrate crystals with larger particle sizes at the bottom of the reactive crystallizer and bond magnesium ions and hydroxide ions to form magnesium hydroxide with smaller particle sizes to continuously overflow from an overflow opening of the reactive crystallizer. The method has the characteristics of simple procedures, high separation efficiency, low equipment investment and running cost and the like, and can be used for treating magnesium sulfate and magnesium sulfite wastewater in large batches. Reaction products can be used for preparing high-quality magnesium hydroxide and calcium sulfate dihydrate for selling as chemical products; reaction mother liquor can be used as process water instead of fresh water, so that zero emission of magnesium sulfate and magnesium sulfite wastewater can be implemented.
Owner:TIANJIN SEA WATER DESALINATION & COMPLEX UTILIZATION INST STATE OCEANOGRAPHI
Method for degrading dyeing waste water by using cerium oxide nano particle as catalyst
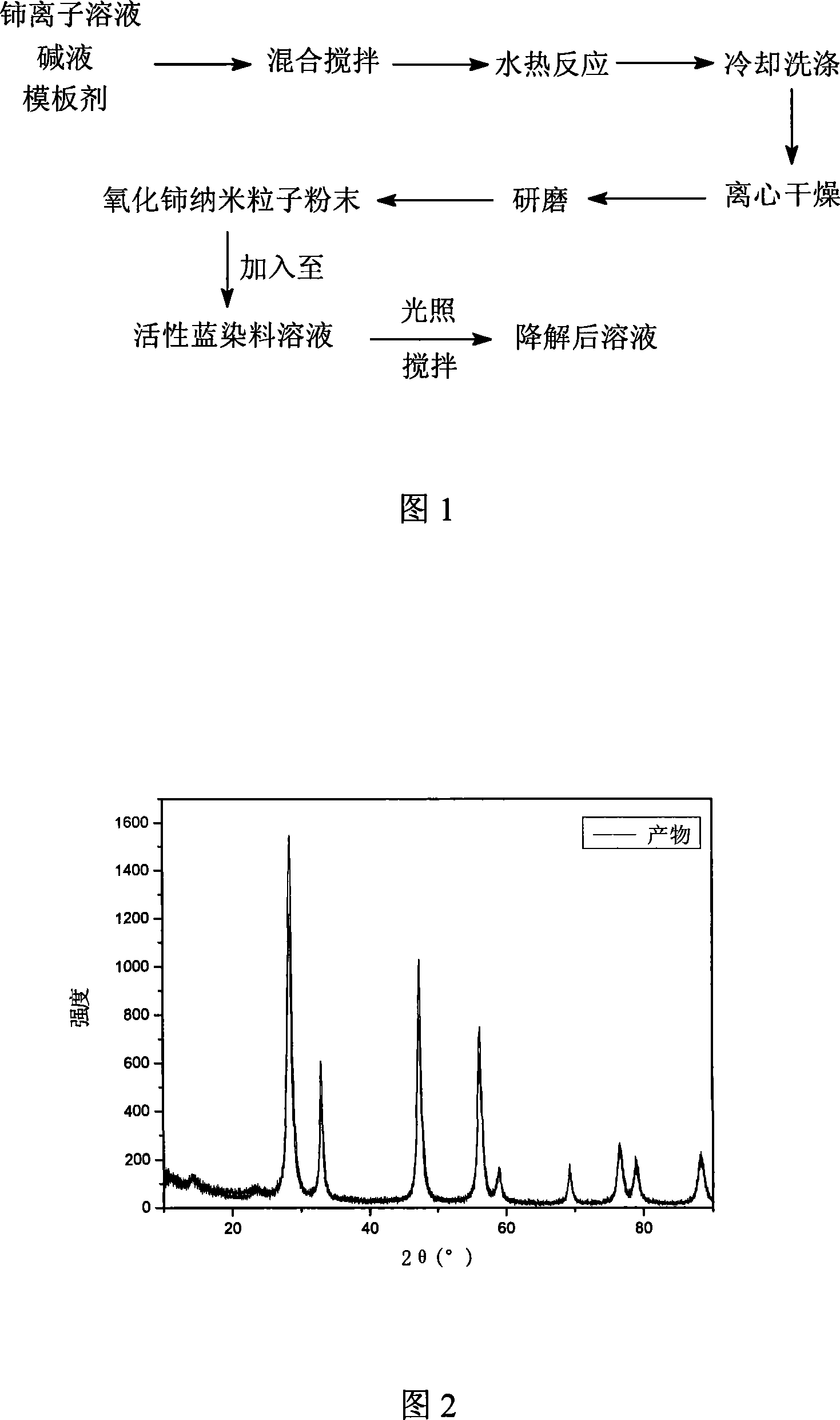
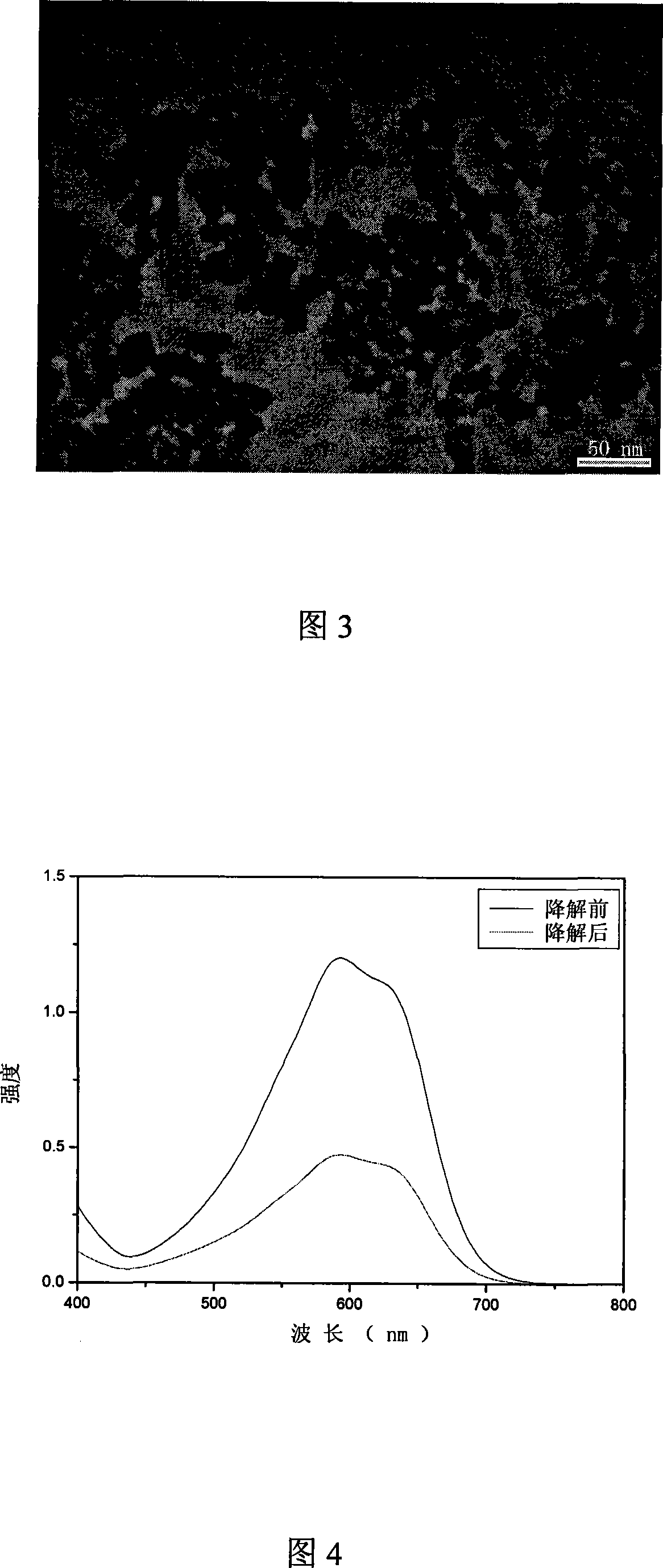
InactiveCN101125704ASimple preparation processHigh product purityWater/sewage treatment by irradiationRare earth metal compoundsChemistryNanometre
A method for degrading dye waste water with ceria nano particles as catalyst pertains to the application technical field of ceria nano particles. The invention is characterized in that: firstly, powder of ceria nano particles is produced with a hydro-thermal method; secondly, the powder of ceria nano particles is added into activated blue dye waste water to degrade the activated blue dye waster water by illumination. The preparation technique of the catalyst of the invention is simple without calcinations or load, thereby, the purity of the product is high; the operation technique of the degradation is simple with easily controlled conditions and rapid degradation speed and the reaction can be taken placed by illumination under normal temperature and atmosphere, thereby being energy conservation.
Owner:BEIJING UNIV OF CHEM TECH