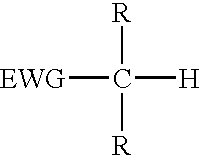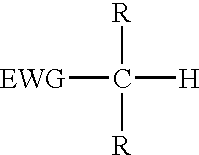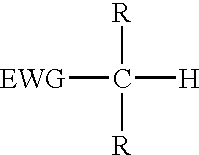Patents
Literature
7407 results about "Side reaction" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
A side reaction is a chemical reaction that occurs at the same time as the actual main reaction, but to a lesser extent. It leads to the formation of by-product, so that the yield of main product is reduced: A+B->[k₁]P₁ A+C->[k₂]P₂ P₁ is the main product if k₁> k₂. The by-product P₂ is generally undesirable and must be separated from the actual main product (usually in a costly process).
Implantable biomedical devices including biocompatible polyurethanes
Disclosed are implantable devices that include biocompatible polyurethane materials. In particular, the disclosed polyurethane materials can maintain desired elastomeric characteristics while exhibiting thermoset-like behavior and can exhibit improved characteristics so as to be suitable in load-bearing applications. For example, the disclosed polyurethane materials can be suitable for use in artificial joints, including total joint replacement applications. The disclosed polyurethane materials include biocompatible cross-linking agents as chain extenders, more particularly chain extenders comprising a terminal group capable of side reactions and further comprising an electron withdrawing group immediately adjacent the terminal group. In addition, the reaction materials and conditions can be selected to encourage intermediate levels of cross-linking without the use of traditional cross-linking trifunctional reagents. In addition, the chain extenders can also include substantially inflexible moieties so as to increase the rigidity of the product polyurethanes.
Owner:CLEMSON UNIVERSITY +1
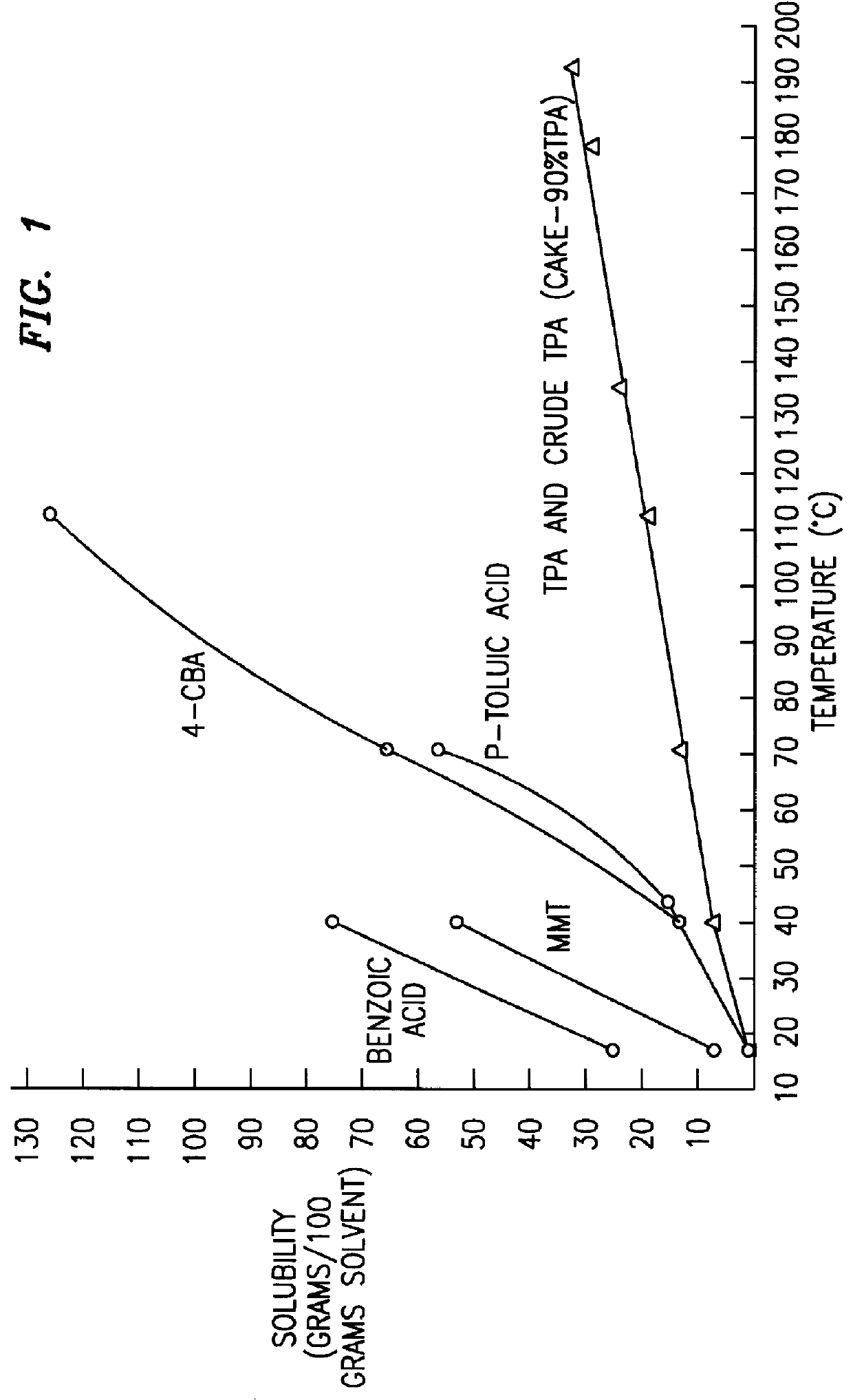
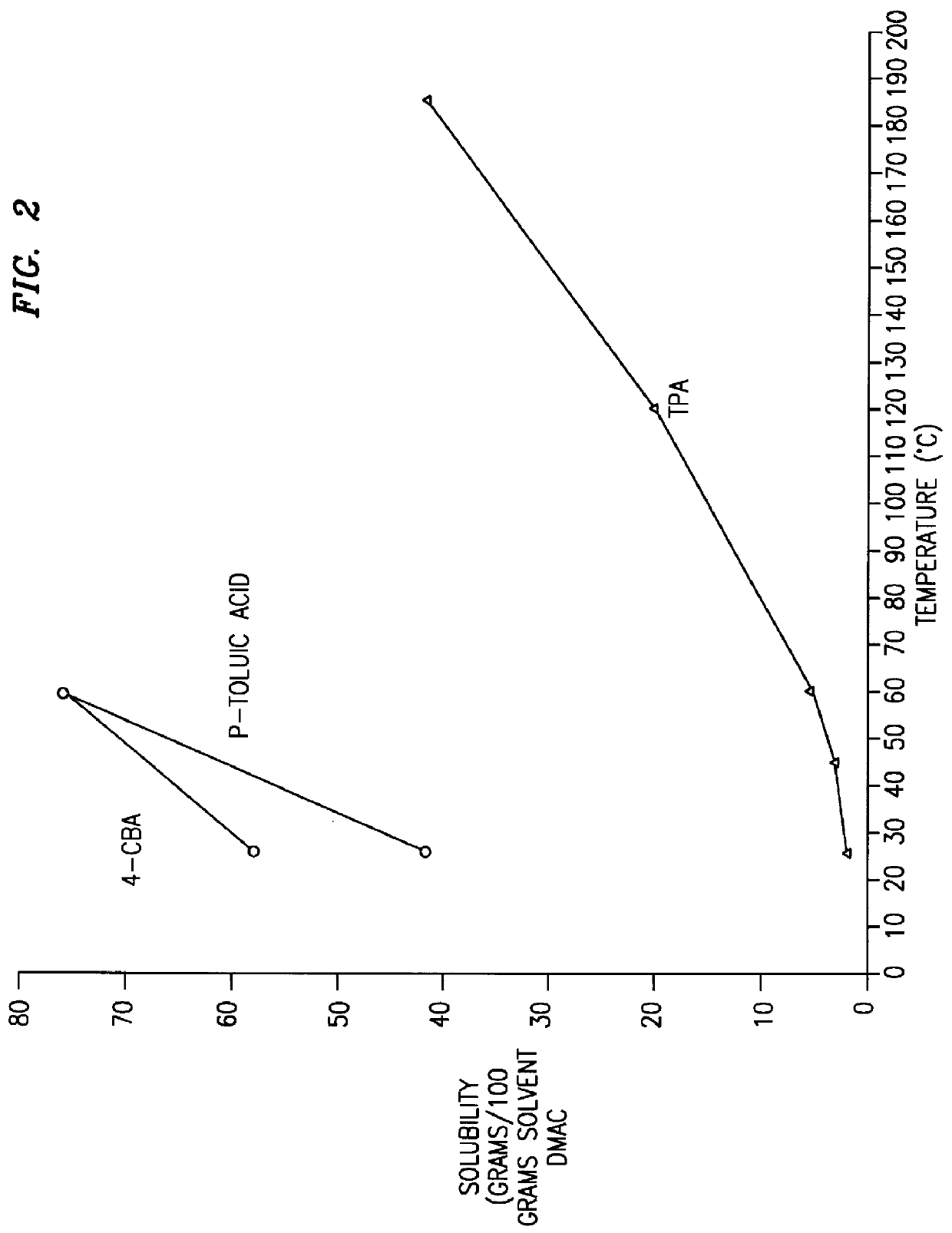
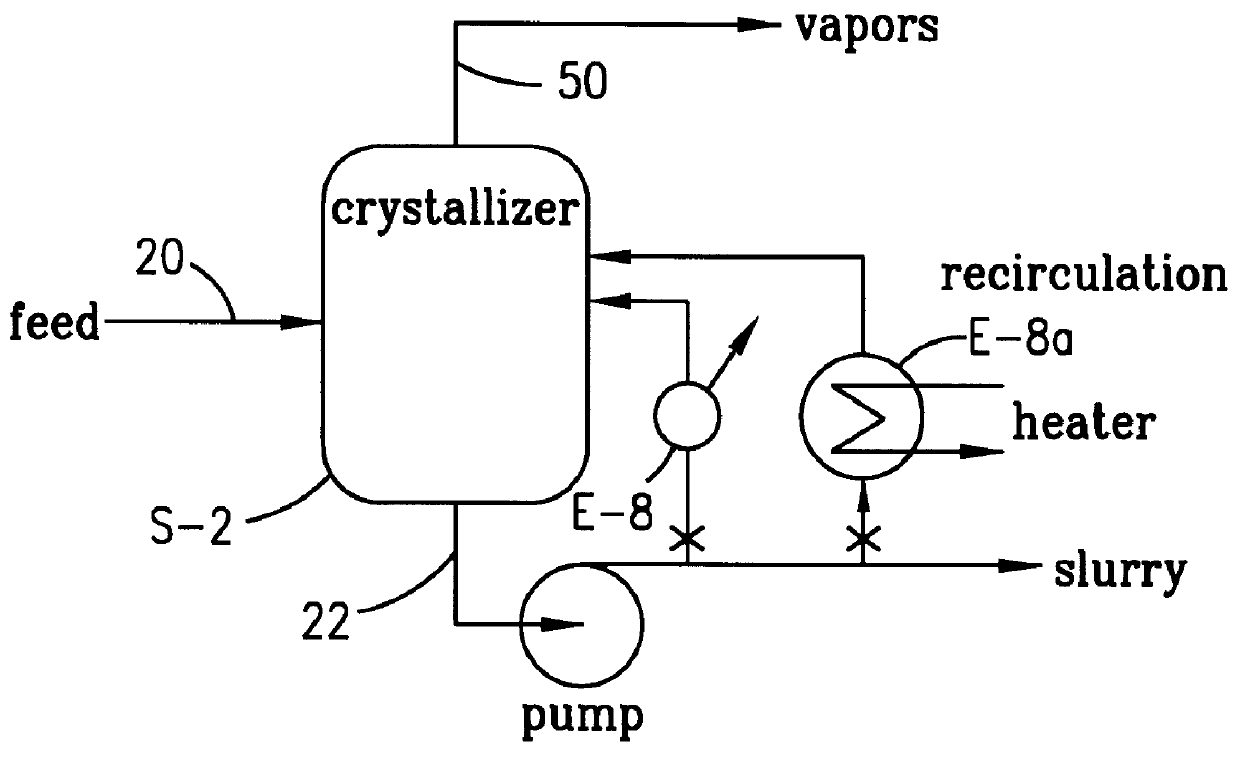
Method and apparatus for preparing purified terephthalic acid
InactiveUS6013835AOrganic compound preparationCarboxylic preparation by oxidationPolyester resinSide reaction
A method and apparatus for purifying crude terephthalic acid from a liquid dispersion thereof also containing impurities selected from unreacted starting materials, solvents, products of side reactions and / or other undesired materials is provided. The method comprises the steps of filtering the dispersion to form a crude terephthalic acid filter cake, dissolving the filter cake in a selective crystallization solvent at an elevated temperature to form a solution, crystallizing purified terephthalic acid from the solution in the crystallization solvent by reducing the pressure and temperature of the solution, and separating the crystallized purified terephthalic acid from the solution. According to the invention, the selective crystallization solvent is non-aqueous, non-corrosive and essentially non-reactive with terephthalic acid. Preferably, the selective crystallization solvent is N-methyl pyrrolidone. The method and apparatus produces purified terephthalic acid having a purity desired for use in forming polyester resin and other products at an economically attractive rate and at operating conditions of reduced severity which require a lower capital investment and simplified processing.
Owner:GTC TECHNOLOGY INC
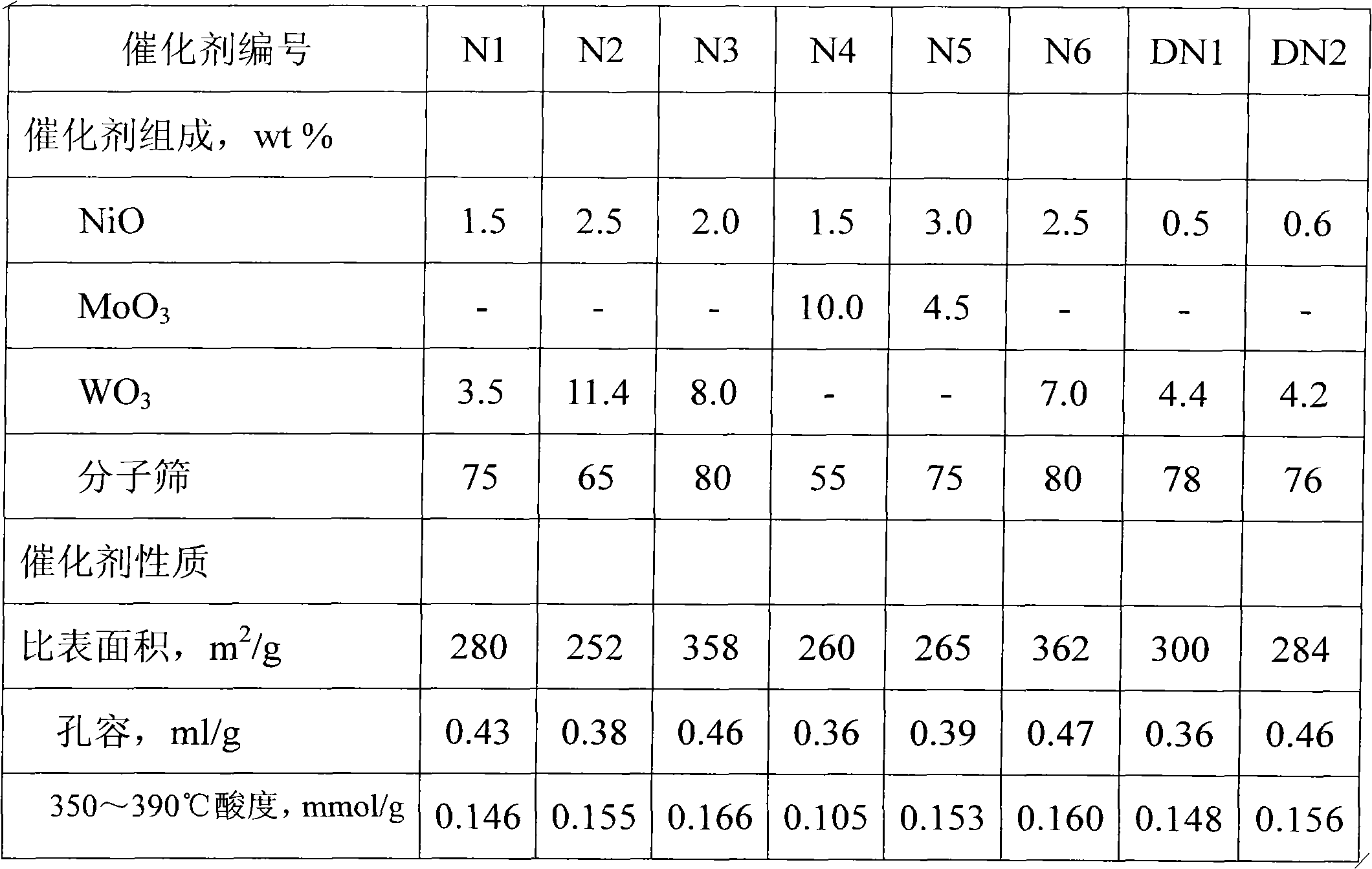
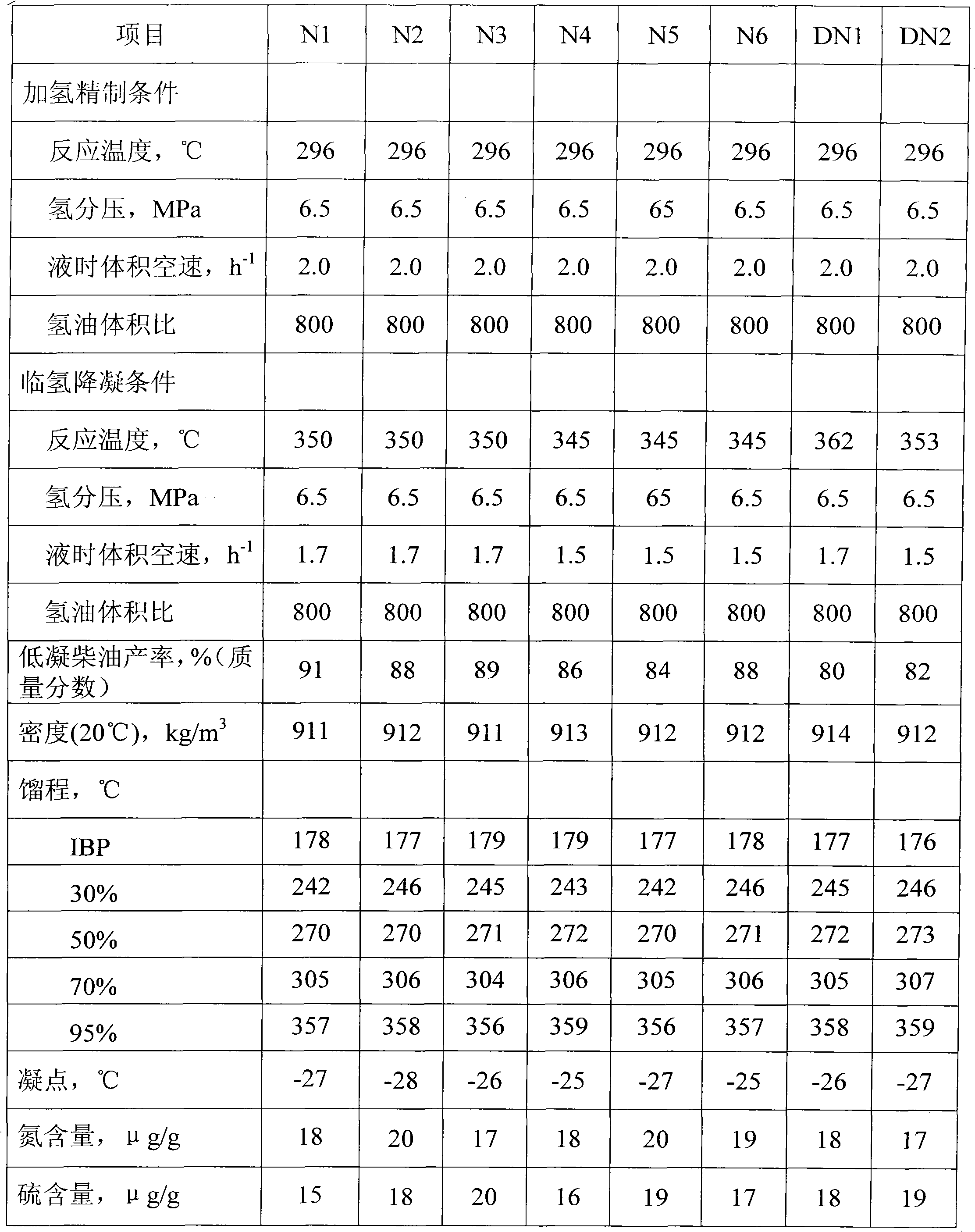
Diesel oil hydrogenation pour point depressing method
ActiveCN102051232ASuitable pore structureAppropriate pore inner surface propertiesCatalyst carriersMolecular sieve catalystsMolecular sieveWax
The invention discloses a diesel oil hydrogenation pour point depressing method. A series flow of hydrofining and hydrogenation pour point depressing is adopted in the method, wherein a hydrogenation pour point depressing catalyst is modified by metal auxiliary agent and silicon, effectively regulates the acidity and orifice shape of the inner surface and the outer surface of a shape-selective cracking molecular sieve, is favorable for dispersing wax molecules in a diesel oil raw material into pore canals of the molecular sieve to perform shape-selective cracking reaction, and has high catalytic activity and selectivity of a target product; and because the acidity of the outer surface of the molecular sieve is weak, side reactions such as secondary cracking and the like are reduced, the pour point depressing effect of the diesel oil is good, and the fraction yield of the diesel oil is high.
Owner:CHINA PETROLEUM & CHEM CORP +1
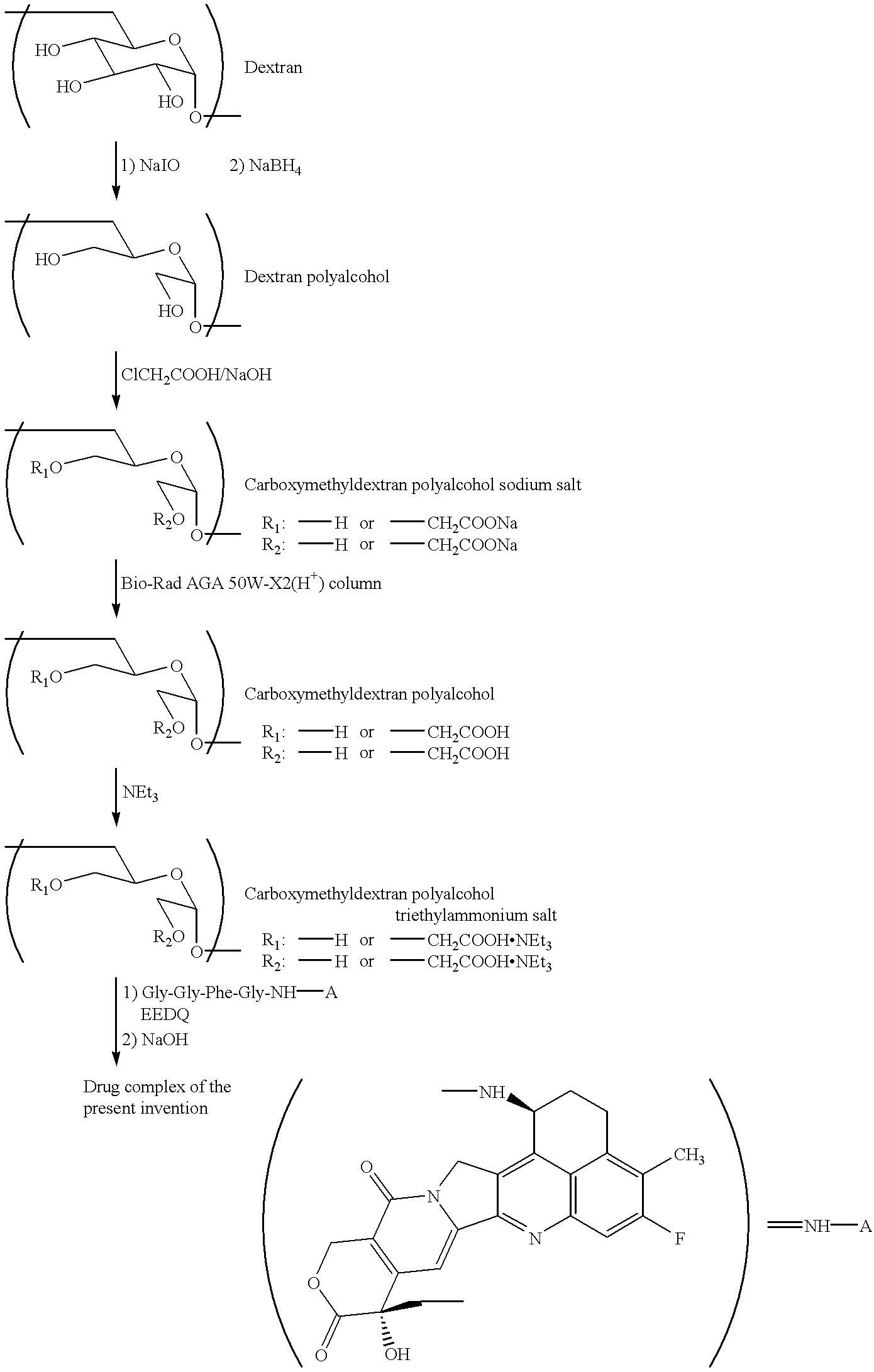
Process for producing drug complexes
InactiveUS6291671B1Efficient productionProduce significantEsterified saccharide compoundsSugar derivativesDrug compoundSide reaction
A method for preparing a drug complex in which a polysaccharide derivative having carboxyl groups and a residue of a drug compound are bound to each other by means of a spacer comprising an amino acid or a spacer comprising peptide-bonded 2 to 8 amino acids, or a drug complex in which a polysaccharide derivative having carboxyl groups and a residue of a drug compound are bound to each other without the spacer, characterized in that an organic amine salt of the polysaccharide derivative having carboxyl groups is reacted with the drug compound or the spacer bound to the drug compound in a non-aqueous system. The reaction between the polysaccharide derivative having carboxyl groups and the drug compound bound with the spacer or the like can be carried out in high yields, and when a drug compound having a lactone ring is subjected to the reaction, side reactions can be reduced.
Owner:DAIICHI PHARMA CO LTD
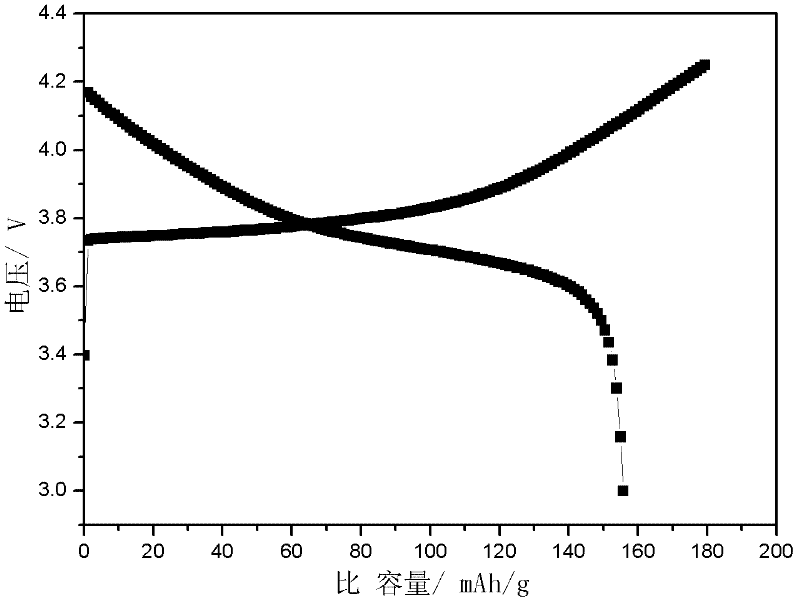
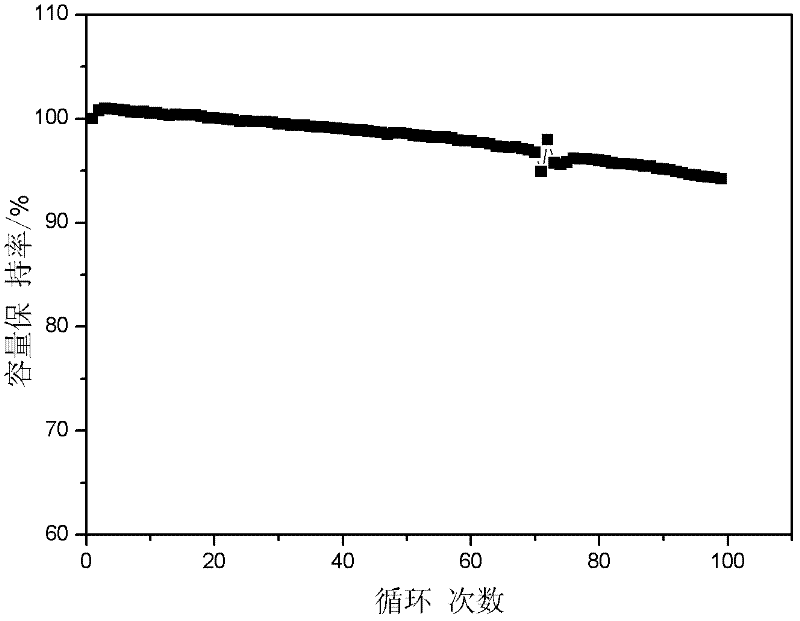
Preparation method of lithium ion battery ternary cathode material
The invention discloses a preparation method of a lithium ion battery ternary cathode material. According to the preparation method, full grinding is performed through a colloid mill to improve the reactivity of a precursor and a lithium salt and the uniformity of a mixed material; and a carbon chain organic additive is added in the process of grinding to improve the viscosity of a sizing material, inhibit raw material segregation in the process of drying, quickly balance the temperature of each part of a system in the process of high-temperature reaction and inhibit agglomeration among particles, so that a uniform-appearance and regular mono-crystal ternary cathode material is obtained. Meanwhile, the ternary cathode material is doped and subjected to surface coating modification, so that the structure of the material is stabilized, the side reaction between the material and electrolyte is inhibited, and the high-temperature cycle performance of the material is improved.
Owner:PULEAD TAIAN TECH IND
Positive electrode active material and its manufacturing method, positive electrode for lithium secondary cell using same, and lithium secondary cell
InactiveUS20060177739A1Deteriorated in battery performanceGood charge and discharge cycle performanceActive material electrodesNon-aqueous electrolyte accumulator electrodesLithiumEngineering
A positive active material is provided which can inhibit side reactions between the positive electrode and an electrolyte even at a high potential and which, when applied to a battery, can improve charge / discharge cycle performance without impairing battery performances even in storage in a charged state. Also provided are: a process for producing the active material; a positive electrode for lithium secondary batteries which employs the active material; and a lithium secondary battery which has improved charge / discharge cycle performance while retaining intact battery performances even after storage in a charged state and which can exhibit excellent charge / discharge cycle performance even when used at a high upper-limit voltage. The positive active material comprises: base particles able to dope and release lithium ions; and an element in Group 3 of the periodic table present on at least part of that part of the base particles which is able to come into contact with an electrolyte. It is produced by, e.g., a process which comprises: producing base particles containing lithium and able to dope and release lithium ions; and then imparting an element in Group 3 of the periodic table to the base particles so that the element can be present on at least part of that part of the base particles which is able to come into contact with an electrolyte.
Owner:GS YUASA INT LTD
Normal-temperature synthesis method for polycarboxylic acid water-reducing agent
The invention discloses a normal-temperature synthesis method for a polycarboxylic acid water-reducing agent, and belongs to the field of cement concrete water-reducing agents. The water-reducing agent is prepared by the following steps of: copolymerizing polyoxyethylene ether monomer or polyoxyethylene ester monomer a containing unsaturated double bonds, unsaturated monocarboxylic acid and derivative monomer thereof b, unsaturated dicarboxylic acid c and unsaturated sulfonic acid or salt monomer thereof d in aqueous solution under the action of an oxidation reduction initiator, and finally neutralizing the solution by using alkali solution to obtain the water-reducing agent. The reaction can be performed at room temperature by adopting an oxidation reduction initiating system, and the appropriate reaction temperature is between 5 and 30 DEG C; and the synthesis process does not need heating, so energy is saved, and high-temperature side reaction is effectively controlled. The prepared polycarboxylic acid high-performance water-reducing agent has the characteristics of high water-reducing rate and good collapse protecting performance. The water-reducing agent has wide application range, and the method has low production process requirement and is suitable for industrialized large-scale production.
Owner:BEIJING UNIV OF TECH
Method for directly converting synthetic gas into low-carbon olefins
ActiveCN102234212AExcellent thermal effectStable temperatureHydrocarbon from carbon oxidesBulk chemical productionRetention timeSlurry
The invention discloses a method for preparing low-carbon olefins from synthetic gas, and belongs to the field of olefins. In order to solve the problems that due to a backmixing phenomenon, the retention time is inconsistent, reaction products are widely distributed, and side reactions of olefin secondary reaction and water gas reaction and the like are intensified so as to influence the economy of the process in the traditional process for preparing low-carbon olefins from synthetic gas, a rotating packed bed reactor is used as a reactor, a solid Fischer-Tropsch synthesis catalyst is used or a catalyst is prepared into slurry, and the synthetic gas is converted into the low-carbon olefins. By the method, the selectivity of the low-carbon olefins can be greatly improved, and reaction heat can be quickly removed to stabilize the temperature of a reaction bed.
Owner:CHINA PETROLEUM & CHEM CORP +1
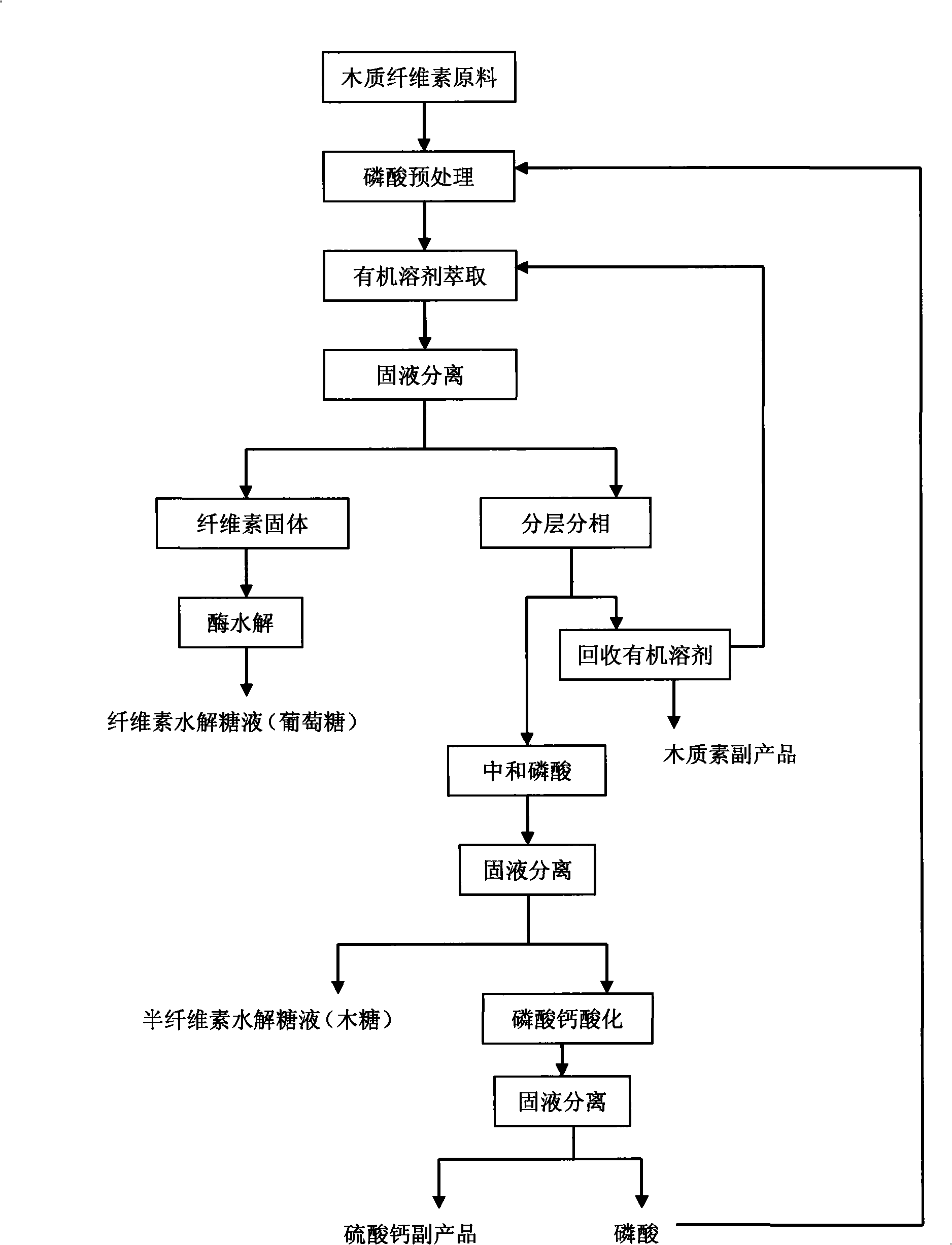
Process for preparing multicomponent liquid glucose and lignose while effectively hydrolyzing lignocellulosic biomass
InactiveCN101285106ASimple recycling processNot volatileSulfate preparationLignin derivativesLiquid glucoseFiltration
The invention discloses a method for efficiently hydrolyzing lignocellulosic biomass and synchronously preparing multi-component liquid glucose and lignin. The method comprises the steps that: the lignocellulosic biomass through physical crushing is added with phosphoric acid to perform acid hydrolysis; then organic solvent is added to extract the lignin, the layering and the phase separation are performed, then the lignin is extracted out while the organic solvent is reclaimed under the condition of pressure reduction and distillation; phosphoric acid can be reclaimed through steps such as neutralization, filtration, acidification and so on, hemicellulose hydrolyzed liquid glucose is obtained at the same time; and the remained cellulose undergoes the zymohydrolysis to prepare cellulosic hydrolyzed liquid glucose. The method can separate lignin, hemicellulose and cellulose, remarkably decreases the degree of crystallinity of the cellulose hydrolyzed by phosphoric acid, and remarkably improves the zymohydrolysis efficiency; and the prepared hydrolyzed liquid glucose does not contain fermentation inhibitors. The method has mild treatment conditions, simple process and less side reactions; the phosphoric acid and the organic solvent can both be reclaimed and circularly used; and the method is environment-friendly, and has broad social and economic benefits.
Owner:NANJING UNIV OF TECH
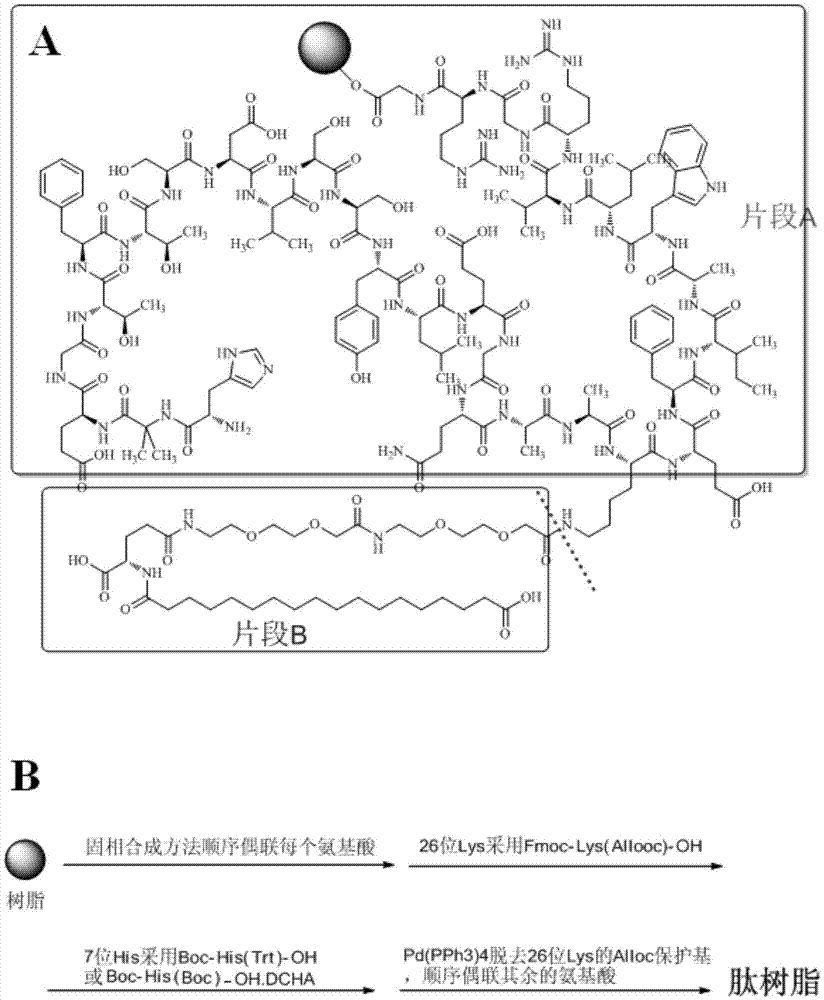
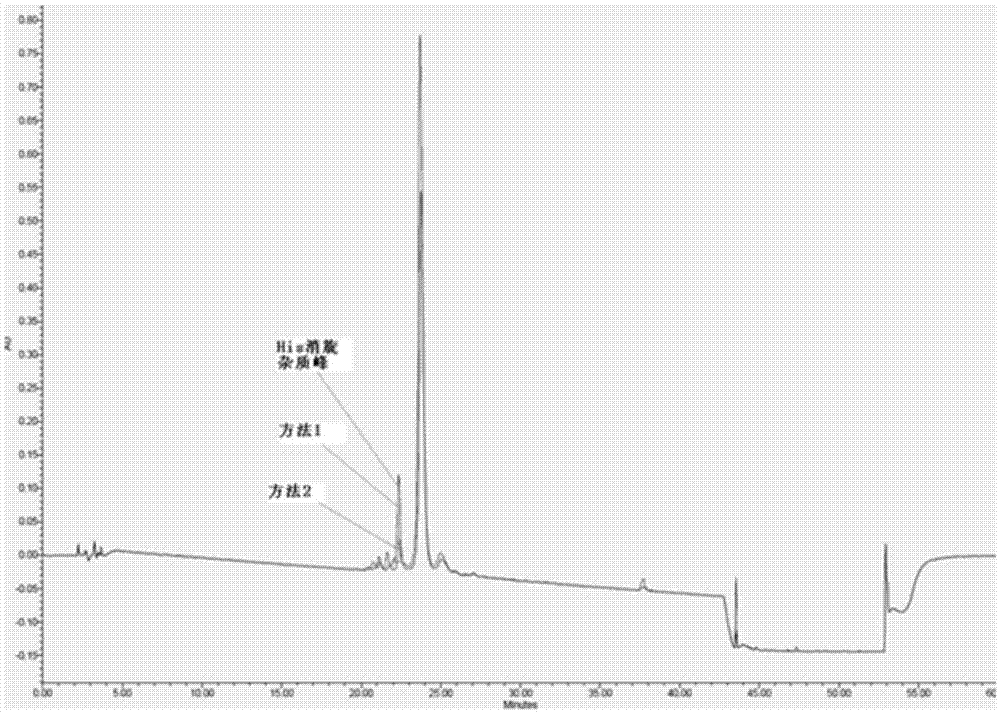
Method for preparing semaglutide
InactiveCN106928343AEasy to operateNo side effectsPeptide-nucleic acidsPeptide preparation methodsSide reactionSemaglutide
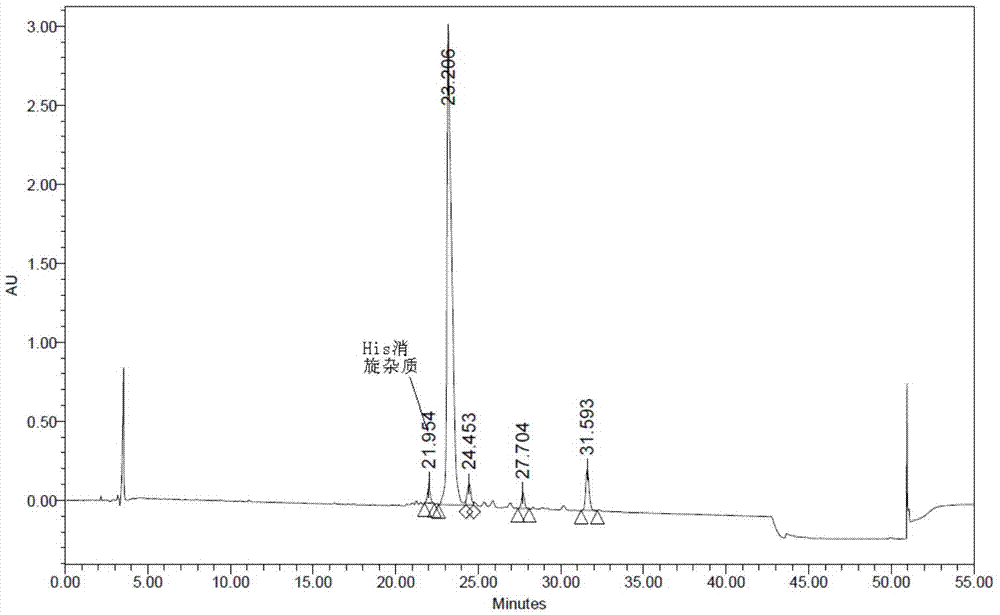
The invention relates to the field of polypeptides, in particular to a method for preparing semaglutide. The method has the advantages that Fmoc-Lys (Alloc)-OH protection amino acid is used as a raw material, de-protection is carried out by the aid of selected Pd (PPh3) 4, accordingly, operation procedures are simple, only 1-2 times of simple elimination reaction operation are required, each elimination reaction operation is carried out for 10-30 min, side reaction is prevented, the operation procedures are safe, and enlarged production can be facilitated; Boc-His (Boc)-OH. DCHA and Boc-His (Trt)-OH are used as raw materials in the procedures, and accordingly His racemization risks can be reduced to the greatest extent; special fragments are coupled, and accordingly the synthesis efficiency can be improved.
Owner:HYBIO PHARMA
Electrolyte of lithium-ion battery and lithium-ion battery containing electrolyte
ActiveCN106505249AExcellent high temperature storageExcellent high temperature cycle performanceSecondary cells servicing/maintenanceOrganic electrolytesHigh temperature storageSide reaction
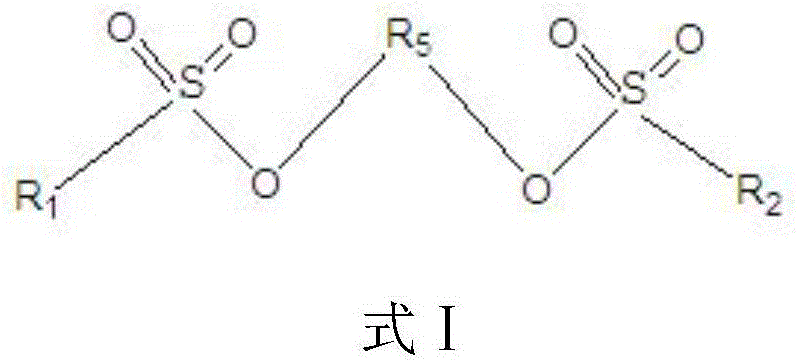
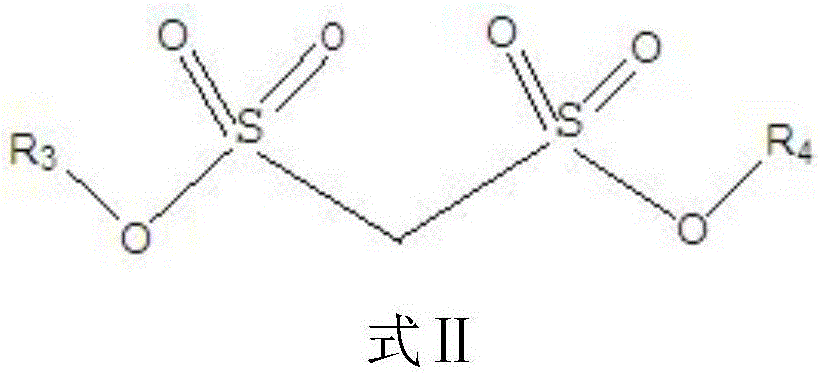
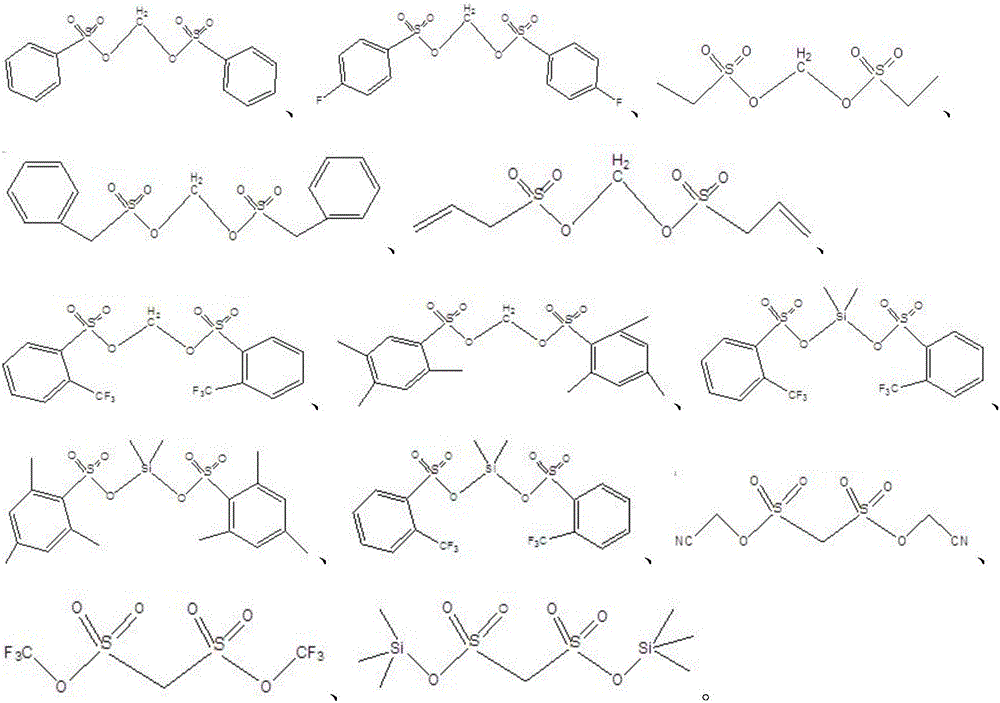
The invention relates to the technical field of lithium-ion batteries, in particular to an electrolyte of the lithium-ion battery and the lithium-ion battery containing the electrolyte. The electrolyte comprises a lithium salt, a non-aqueous organic solvent and an additive, wherein the additive comprises a film-forming additive A and a stabilizing additive B; the stabilizing additive B is a chainlike disulfonic acid ester compound as shown in a formula I and / or a formula II; and the film-forming additive A forms an SEI film on a negative electrode surface of the battery and the side reaction of a negative electrode interface and the electrolyte is reduced, so that the film-forming additive A is an necessary precondition that the battery has relatively good cycle performance. The stabilizing additive B can form a CEI film on a positive electrode surface, the activity of a positive electrode interface and the electrolyte is inhibited, direct contact oxidation of a positive electrode and the electrolyte is reduced, meanwhile, the structure stability of a positive electrode material is improved and the structure is not easy to collapse and crush due to generation of the stress, so that the electrolyte has excellent high-temperature storage performance and high-temperature cycle performance through combination of the additive A and the additive B.
Owner:DONGGUAN SHANSHAN BATTERY MATERIALS
Reagents for oligonucleotide cleavage and deprotection
InactiveUS6664388B2Convenient and ready-to-use formatContaminate the synthesized oligonucleotides can be minimizedSugar derivativesRecombinant DNA-technologyChemical synthesisAryl
The present invention provides a process for the removal of protecting groups, i.e. deprotection, from chemically synthesized oligonucleotides. In one embodiment, the invention provides reagents suitable for use in such a process, and kits incorporating such reagents in a convenient, ready-to-use format. By use of the process and reagents of the invention, side-reactions leading to certain impurities that contaminate the synthesized oligonucleotides can be minimized.Methods and reagents are provided for deprotection of an oligonucleotide by reacting a protected oligonucleotide with a deprotection reagent wherein the deprotection reagent comprises an active methylene compound and an amine reagent. The active methylene compound has the structure:where substituent EWG is an electron-withdrawing group and R is hydrogen, C1-C12 alkyl, C6-C20 aryl, heterocycle or an electron-withdrawing group.
Owner:APPL BIOSYSTEMS INC
Low metal content catalyst compositions and processes for making and using same
ActiveUS7125821B2Reduce metal contentReduce the amount requiredMolecular sieve catalystsMolecular sieve catalystMolecular sieveOxygenate
The invention provides low metal content molecular sieve catalyst compositions, processes for making such catalysts, and processes for using such catalysts in the conversion of an oxygenate into one or more light olefins. Preferably, the catalyst composition comprises a matrix material having a low metal content. By utilizing matrix materials having low metal contents, the amount of metal-catalyzed side reaction byproducts formed in a reaction system, particularly in an oxygenate-to-olefin reaction system, can be advantageously reduced.
Owner:EXXONMOBIL CHEM PAT INC
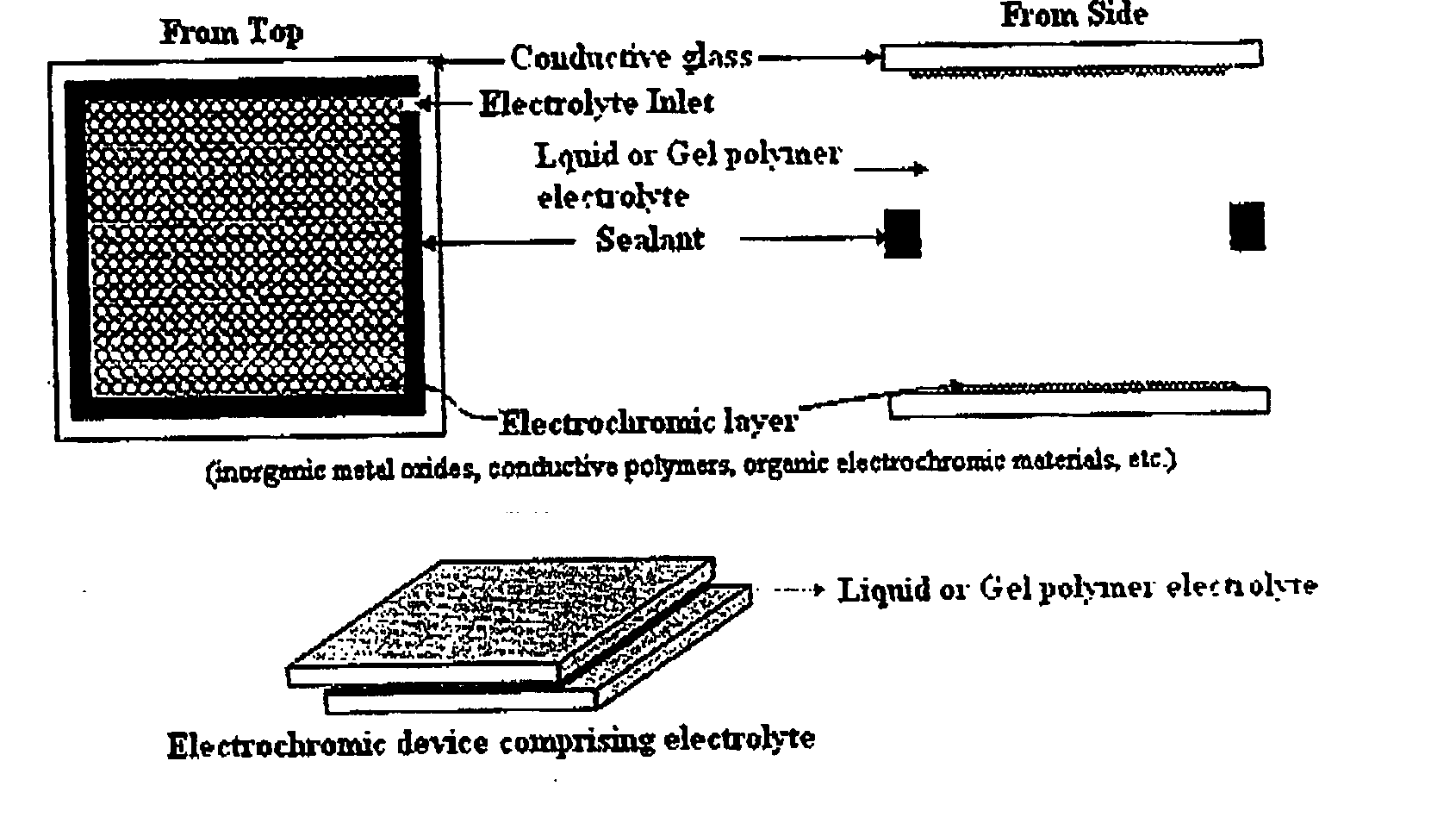
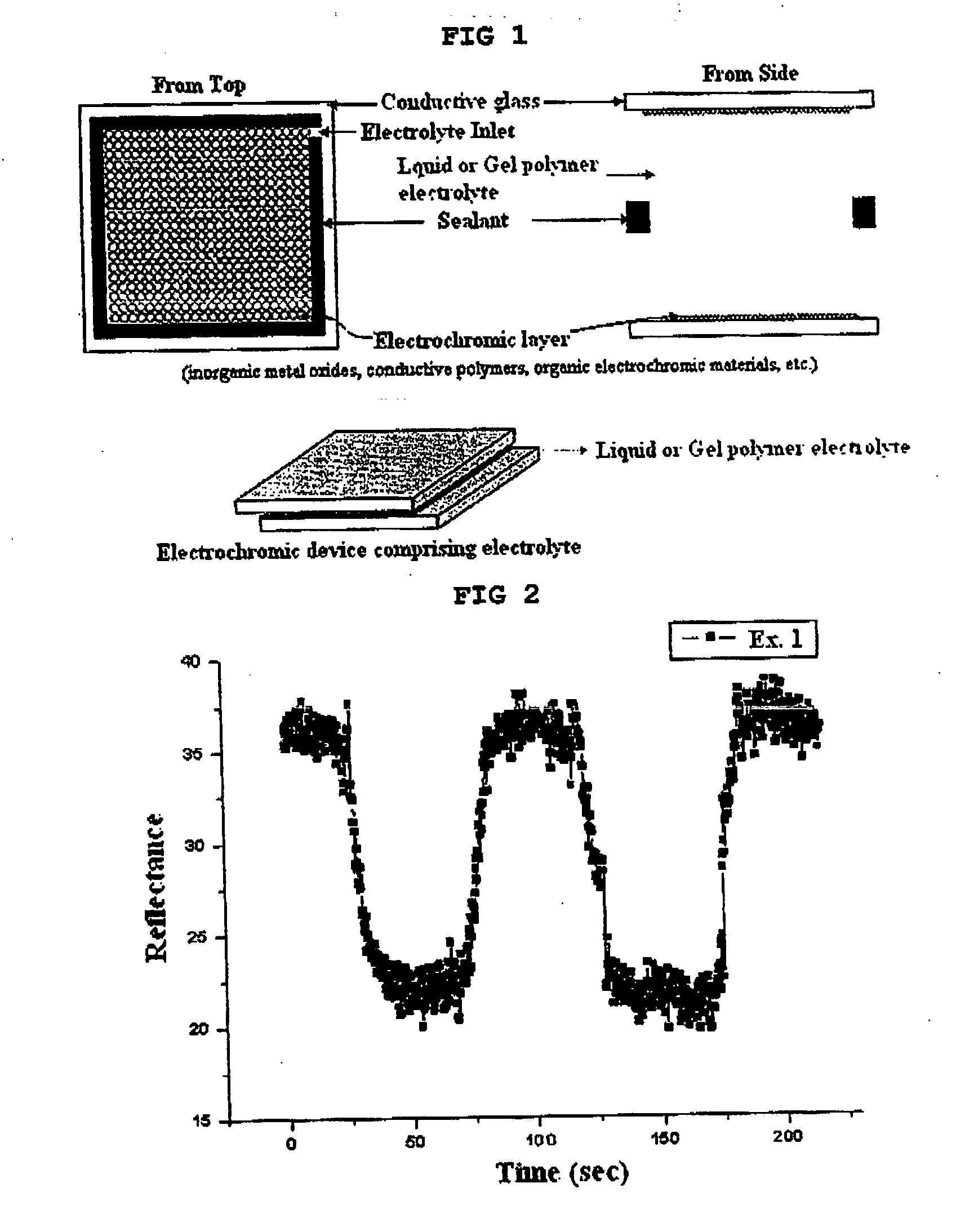
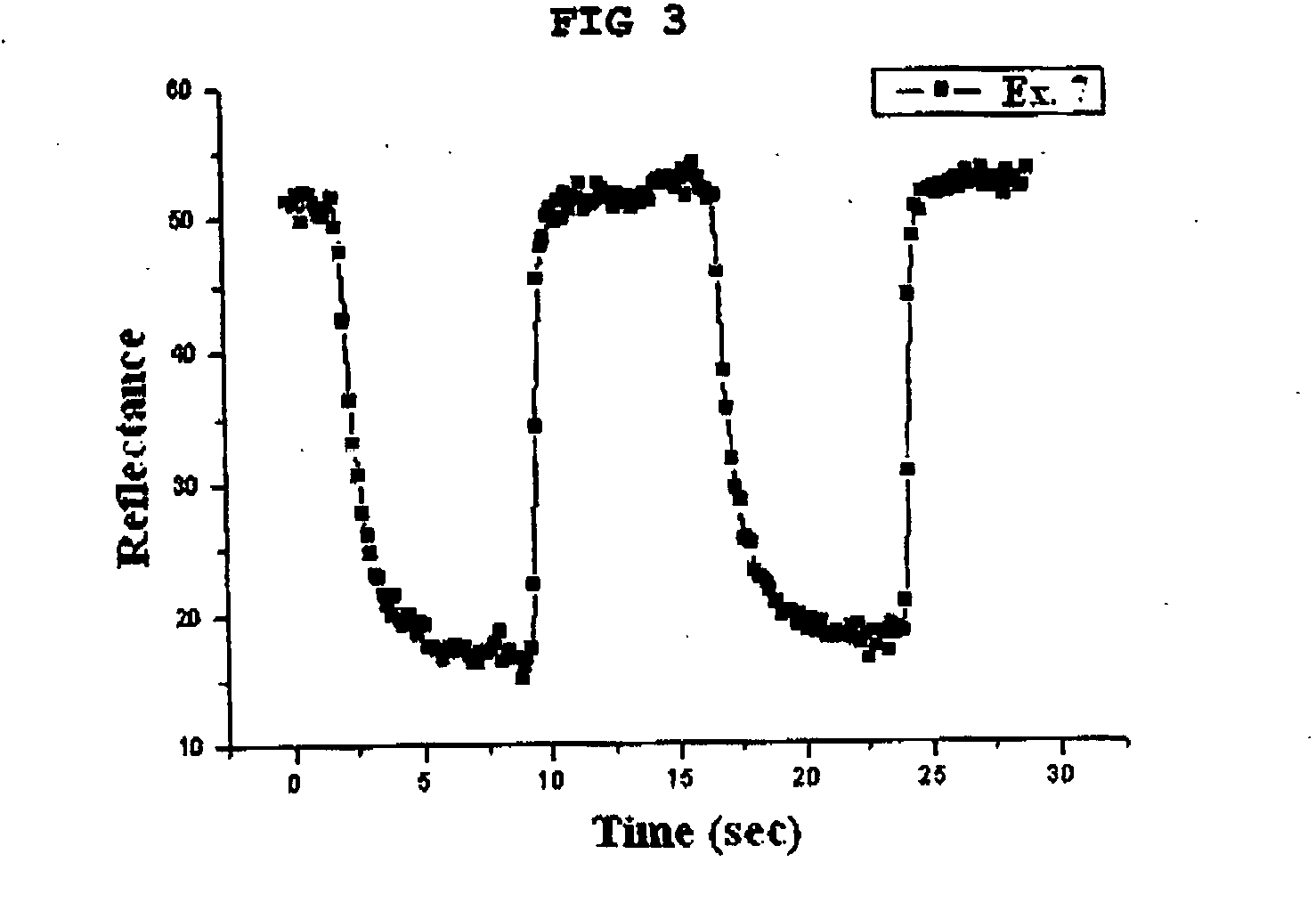
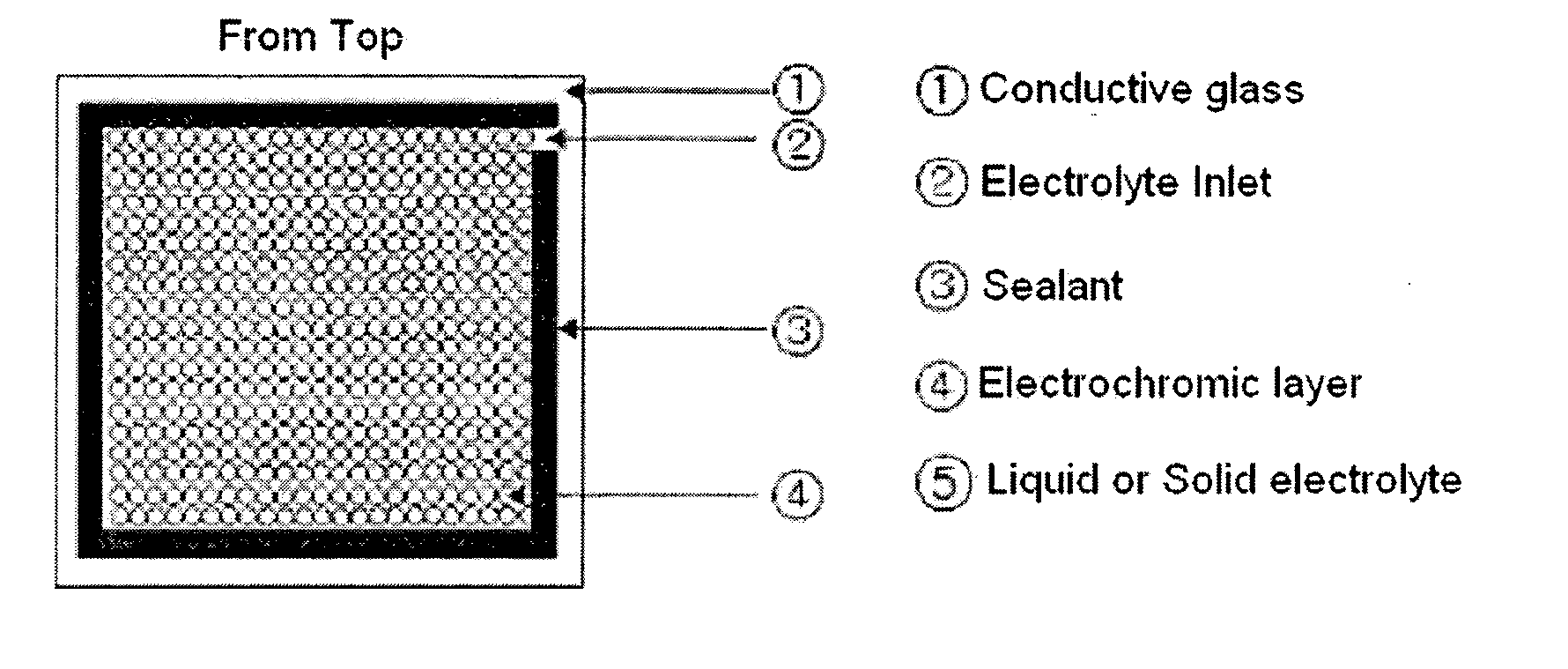
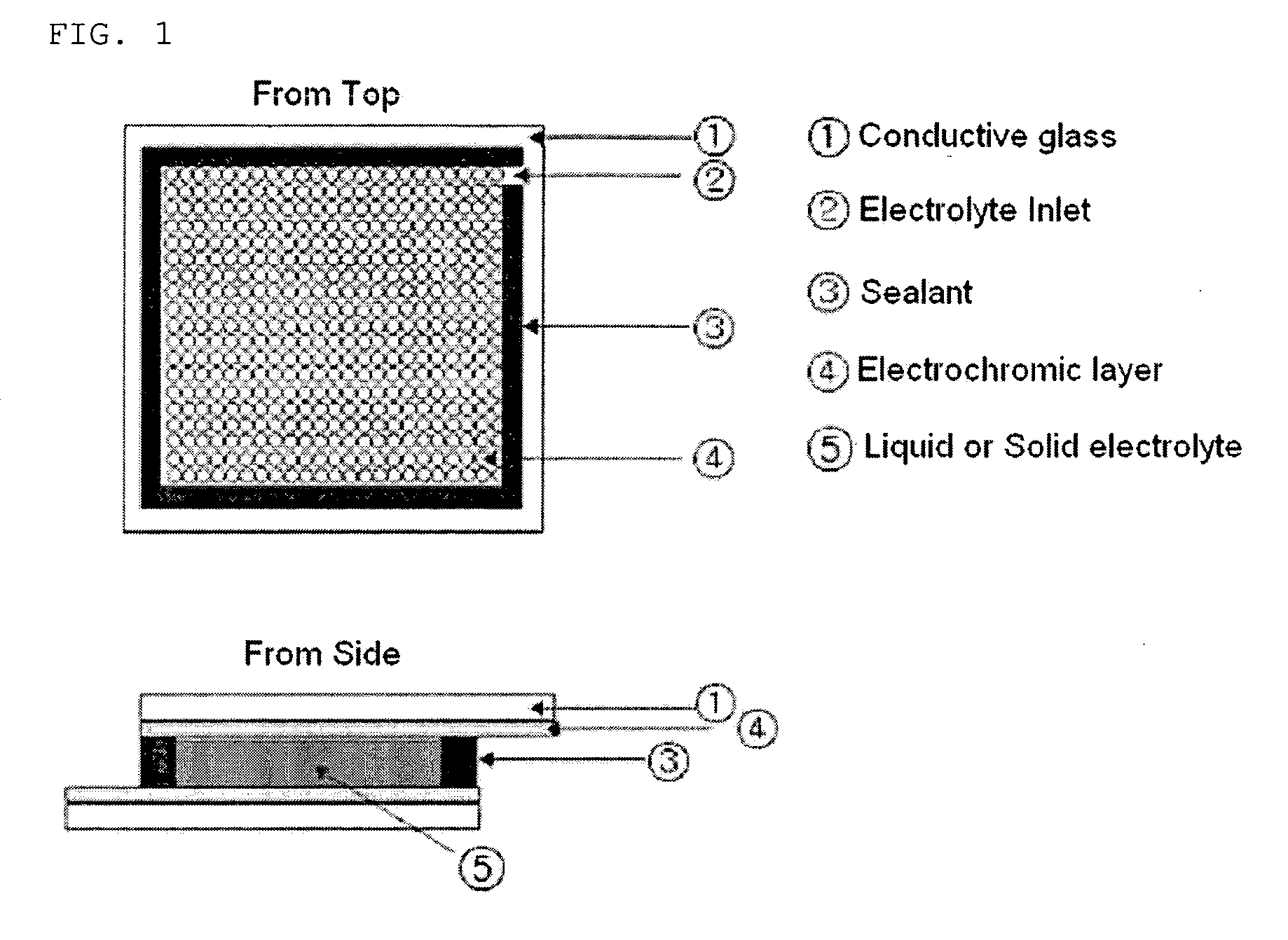
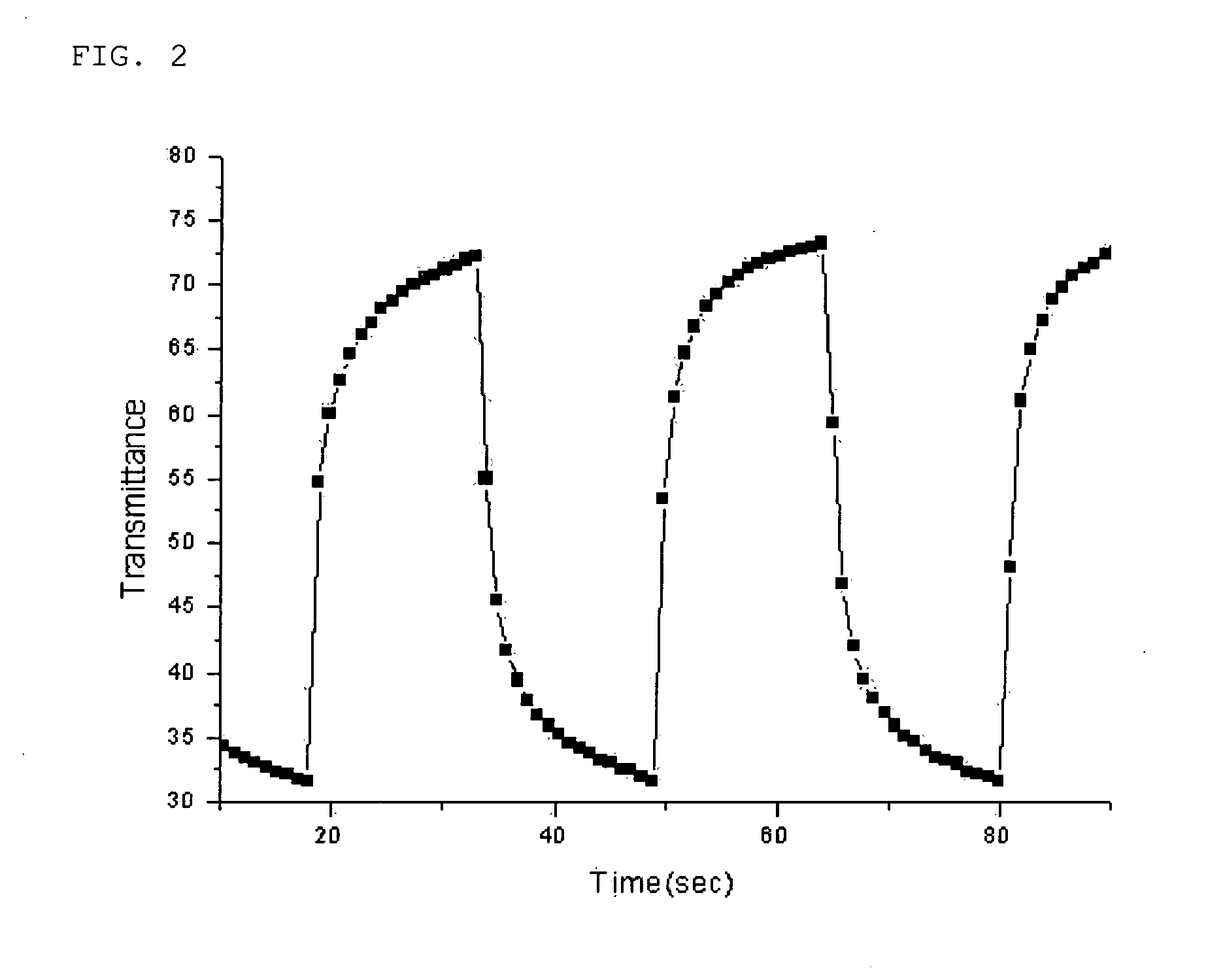
Gel polymer electrolyte containing ionic liquid and electrochromic device using the same
ActiveUS7411716B2Solve the real problemIncrease chanceLapping machinesTenebresent compositionsStructural deformationPolymer electrolytes
Owner:LG ENERGY SOLUTION LTD
Anode coated with lithium fluoride compounds, method for preparing the same, and lithium secondary battery having the same
ActiveUS20110177388A1Prevent degradationElectrochemical processing of electrodesActive material electrodesBattery chargePhysical chemistry
An anode for a lithium ion secondary battery includes an anode, and a LiF-based coating layer formed with LiF-based particles on a surface of the anode. The LiF-based coating layer has a thickness of 0.05 to 1 μm. The anode allows the LiF-based coating layer created by side reaction of LiPF6 during a battery charging / discharging process to be relatively uniformly formed on the anode surface, thereby elongating the life cycle of a lithium ion secondary battery.
Owner:LG ENERGY SOLUTION LTD
Modified composite material containing silicon-base material, preparation method thereof, and application thereof in lithium ion battery
PendingCN109301184AImprove performanceEasy to crushMaterial nanotechnologyCell electrodesStructural stabilitySide reaction
The invention discloses a modified composite material containing silicon-base material, a preparation method thereof, and an application thereof in a lithium ion battery. The composite material includes a silicon-base core and a modifying layer covering same. The modifying layer includes a polymer covering film and a nano-conductive material embedded therein. The modified composite material not only can isolate an electrolyte to inhibit side reactions of the electrolyte on the surface of the material, thus improving overall performance of the battery, but also can improve electric conductivityand improve structural stability due to the unique structure by embedding the nano-conductive material in the polymer. Tension is absorbed by means of deformation, so that problems of breaking and pulverization and stripping of the electrode during processing and use of the silicon-base negative material are solved. The problems of pulverization and stripping of the electrode due to large expansion during charge / discharge processes of the silicon-base material are solved, thus stabilizing the structure of the electrode and improving cycle performance of the cell.
Owner:JIANGSU ZENIO NEW ENERGY BATTERY TECH CO LTD
Manufacturing method of copolyester for low acetaldehyde content of PET bottles
InactiveUS6489434B2Reduce productionLow in acetaldehydeLayered productsBottlesPolyethylene terephthalate glycolBottle
The present invention provides a manufacturing method of copolyester for low acetaldehyde content of PET bottles. The polyethylene terephthalate (PET) polymer is added with an appropriate modifier in order to decrease the production of acetaldehyde caused by pyrolysis side reaction during the blow molding process of PET bottles. The modifier comprises stabilizer and primary antioxidant, wherein the stabilizer is an inorganic phosphorous compound with an addition quantity of 0.003~0.5 weight % based on the weight of the total copolyester copolymer and the primary antioxidant is a hindered phenolic antioxidant containing Ca+2 with an addition quantity of 0.005~5.0 weight % based on the weight of the total copolyester copolymer. The present invention owns an improving effect of decreasing the production of side product-acetaldehyde at least 30% than those without the addition of said modifier.
Owner:NANYA PLASTICS CORP
Porous polypropylene particle suspension solid phase graft-modification method
The invention relates to a method for modifying grafting for porous polypropylene particle floating solid phase. The invention modifies floating solid phase grafting for polypropylene particle absorbed with monomer and initiator in salt solution, to increase grafting depth, reduce degradation and side reaction, and improve mechanics property for polypropylene. The modified product can also be used as compatible agent for PP and other polymer to increase material compatibility and then improve mechanics property.
Owner:BEIJING UNIV OF CHEM TECH
Gel polymer electrolyte containing ionic liquid and electrochromic device using the same
ActiveUS20050231785A1Good effectLow viscosityLapping machinesTenebresent compositionsStructural deformationElectrolytic agent
Disclosed is an electrochromic device comprising: (a) a first electrode; (b) a second electrode; (c) an electrochromic material; and (d) a gel polymer electrolyte containing an ionic liquid. A method for manufacturing the same is also disclosed. The electrochromic device uses a gel polymer electrolyte comprising an ionic liquid. Therefore, there is no problem related with electrolyte leakage. Additionally, it is possible to manufacture electrochromic devices by using plastic materials, because the ionic liquid gel polymer electrolyte permits structural deformation with ease. Further, because the electrochromic device uses an ionic liquid, it is possible to minimize side reactions between constitutional elements of an electrochromic device and electrolyte.
Owner:LG ENERGY SOLUTION LTD
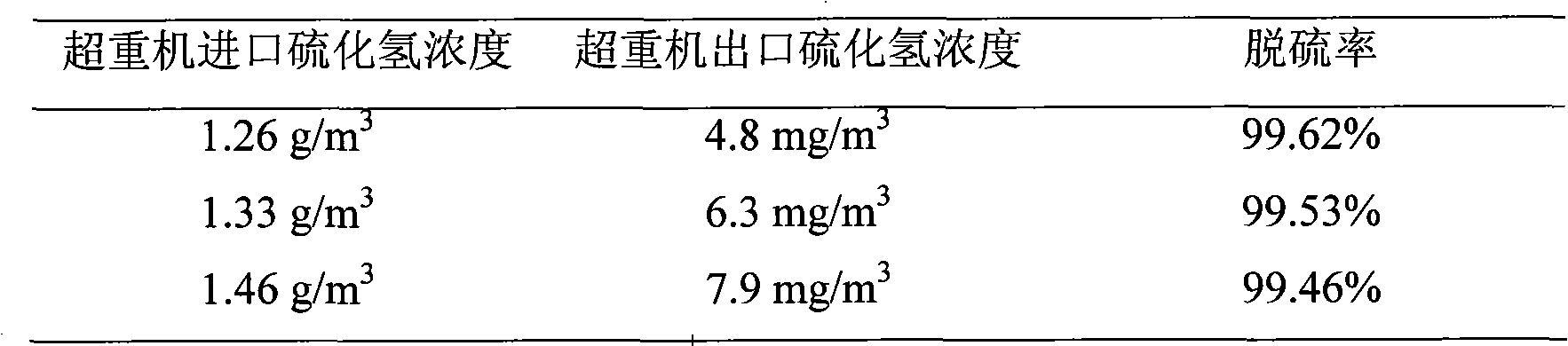
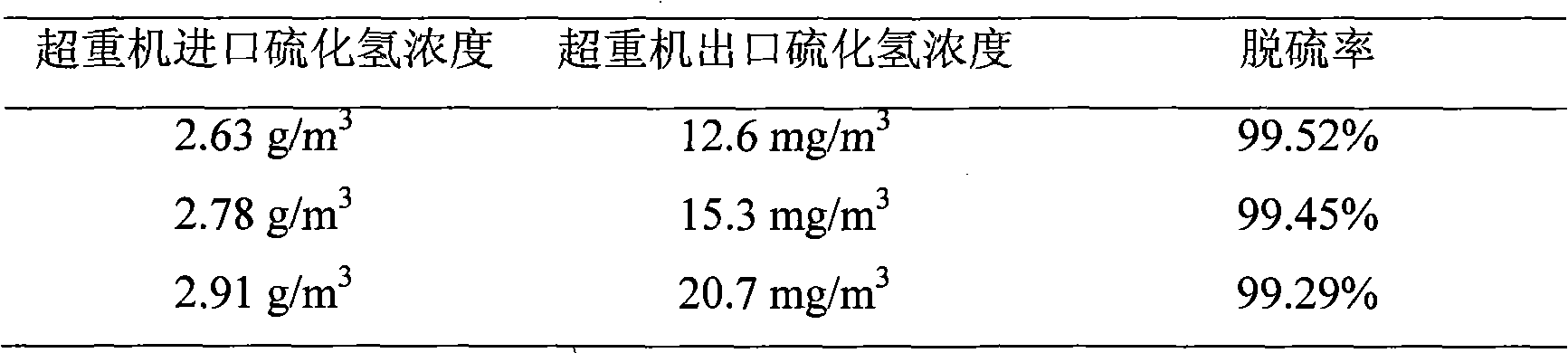
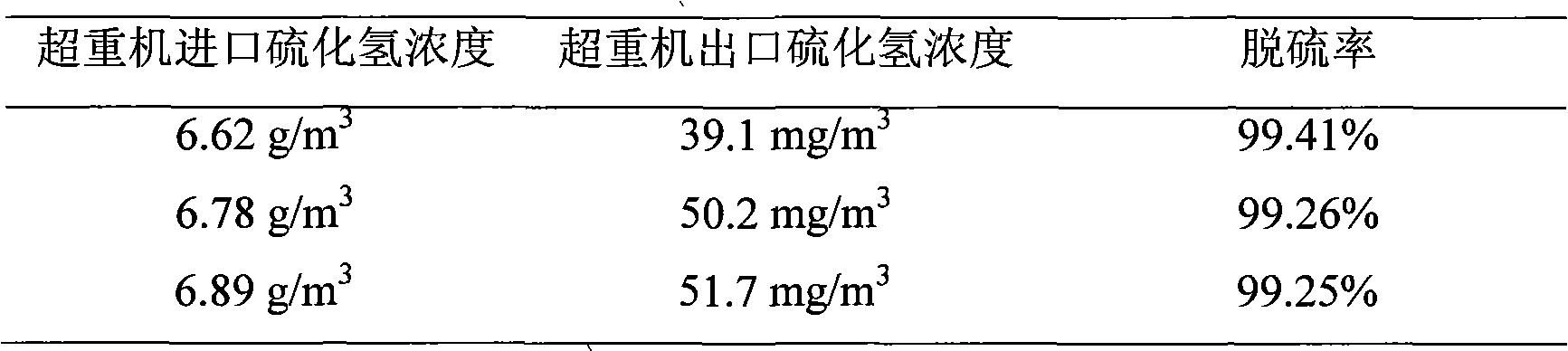
Iron complex desulfurizer suitable for super-gravity desulfurization
ActiveCN101874968AReduce corrosionReduce corrosion rateDispersed particle separationSulfur granulesIron salts
The invention belongs to the technical field of gas purification, and in particular relates to an iron complex desulfurizer suitable for super-gravity desulfurization. The active ingredients of the iron complex desulfurizer are the following components: a soluble iron salt, an iron salt complexing agent, hydrosulfide absorbants comprising alkaline matters and alcamines, and additives comprising astabilizing agent, a synergist, a sulfur granule settling agent and a corrosion inhibitor. The iron complex desulfurizer has the advantages of large accumulative saturation sulfur capacity (reaching 0.60g / L), high absorption speed, high efficiency, stable solution performance without degradation, easy recovery of sulfur, less side reactions, low running cost and the like; and the accumulative saturation sulfur capacity and the absorption speed are obviously improved, so the iron complex desulfurizer is suitable for the characteristics of small liquid circulating volume and short retention time of the super-gravity desulfurization, fully takes the obvious advantages of the super-gravity technology in the wet desulphurization, and is a high-efficiency, economic, practical and environment-friendly iron complex desulfurizer.
Owner:ZHONGBEI UNIV
Manufacturing method of copolyester for low acetaldehyde content of PET bottles
The present invention provides a manufacturing method of copolyester for low acetaldehyde content of PET bottles. The polyethylene terephthalate (PET) polymer is added with an appropriate modifier in order to decrease the production of acetaldehyde caused by pyrolysis side reaction during the blow molding process of PET bottles. The modifier comprises stabilizer and primary antioxidant, wherein the stabilizer is an inorganic phosphorous compound with an addition quantity of 0.003~0.5 weight % based on the weight of the total copolyester copolymer and the primary antioxidant is a hindered phenolic antioxidant containing Ca+2 with an addition quantity of 0.005~5.0 weight % based on the weight of the total copolyester copolymer. The present invention owns an improving effect of decreasing the production of side product-acetaldehyde at least 30% than those without the addition of said modifier.
Owner:NANYA PLASTICS CORP
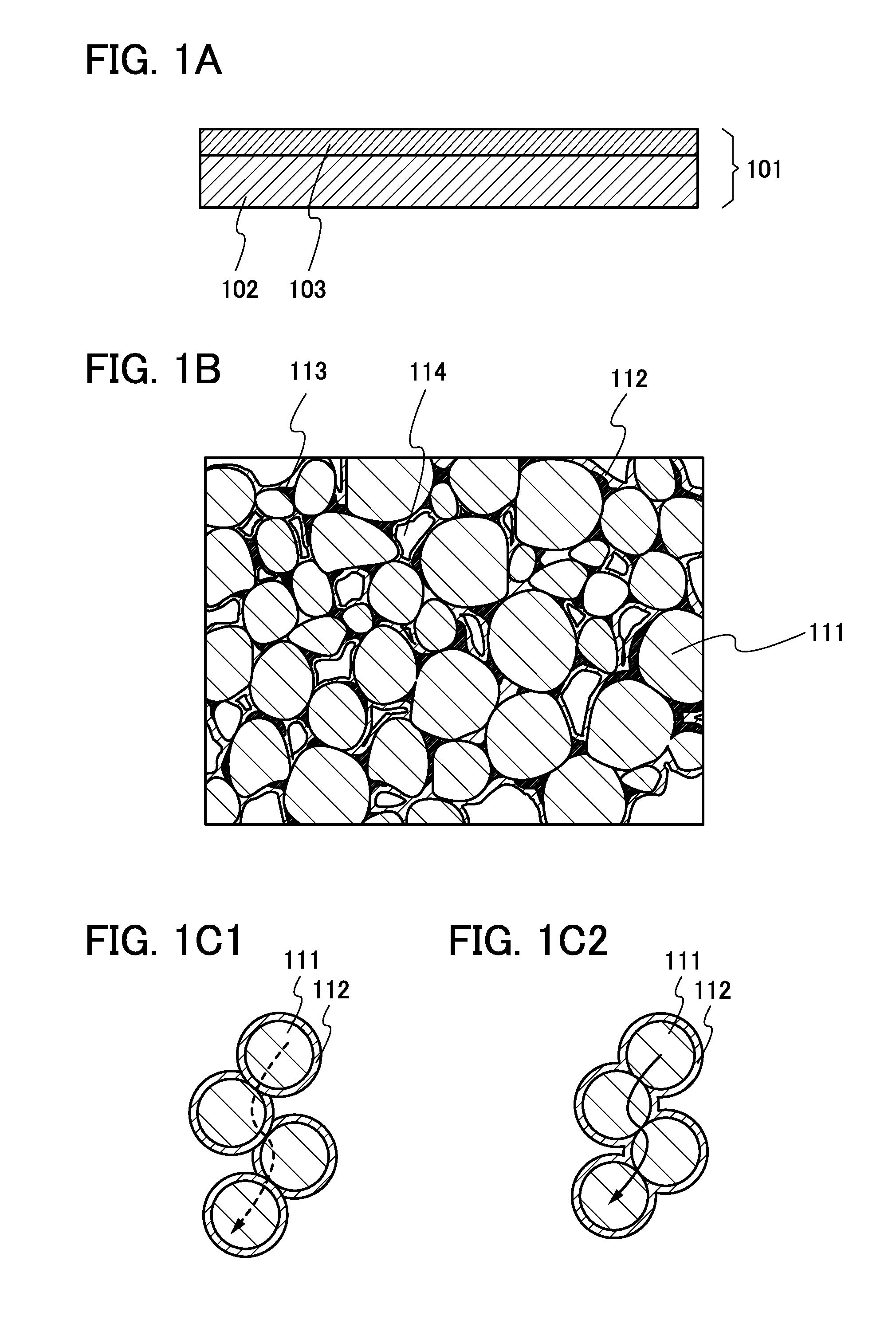
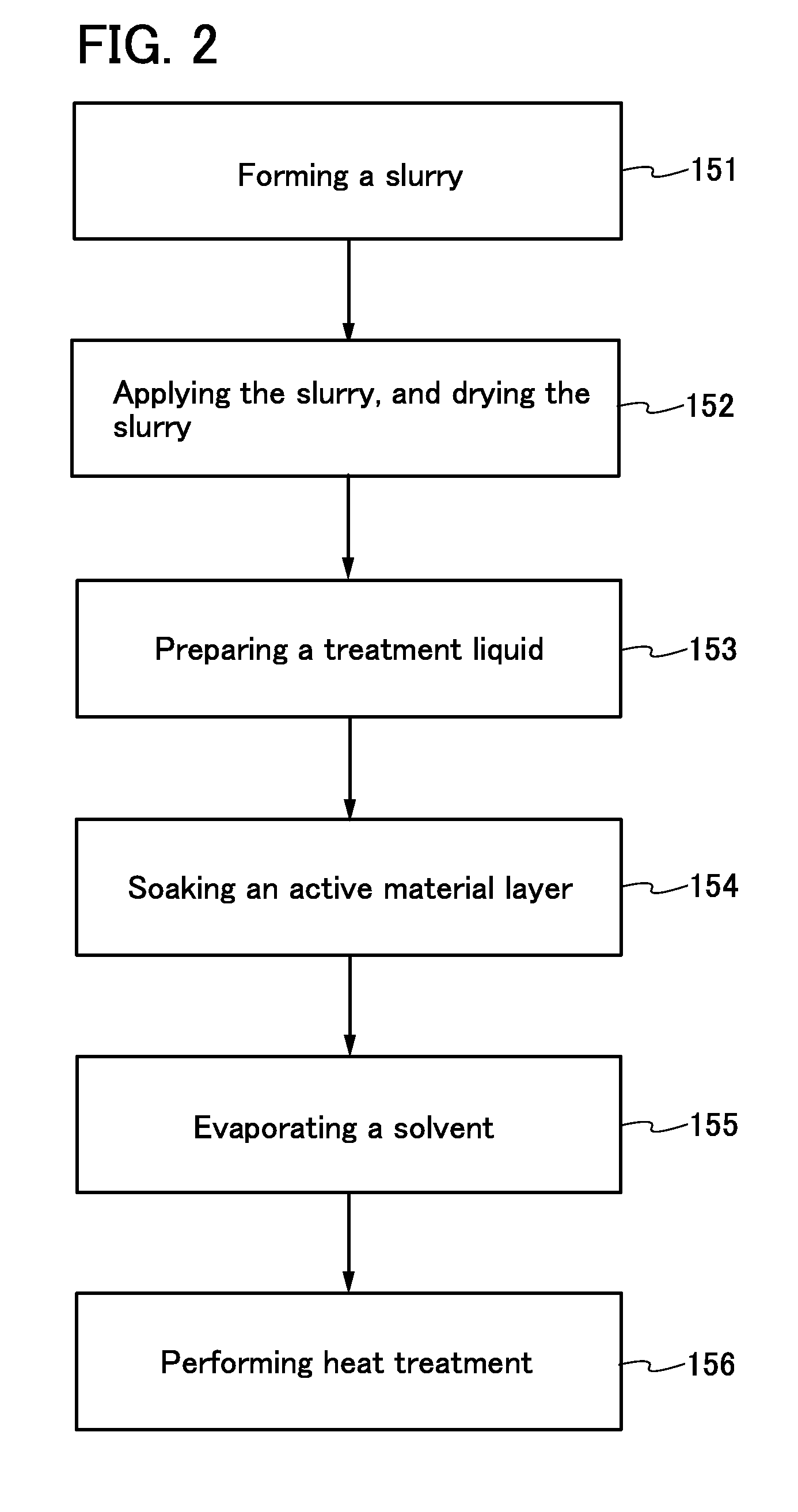
Electrode for power storage device, power storage device, and manufacturing method of electrode for power storage device
ActiveUS20140127567A1Minimize electrochemical decompositionMinimizing reactionHybrid capacitor electrodesActive material electrodesDecompositionSilicon oxide
To improve the long-term cycle performance of a lithium-ion battery or a lithium-ion capacitor by minimizing the decomposition reaction of an electrolytic solution and the like as a side reaction of charge and discharge in the repeated charge and discharge cycles of the lithium-ion battery or the lithium-ion capacitor. A current collector and an active material layer over the current collector are included in an electrode for a power storage device. The active material layer includes a plurality of active material particles and silicon oxide. The surface of one of the active material particles has a region that is in contact with one of the other active material particles. The surface of the active material particle except the region is partly or entirely covered with the silicon oxide.
Owner:SEMICON ENERGY LAB CO LTD
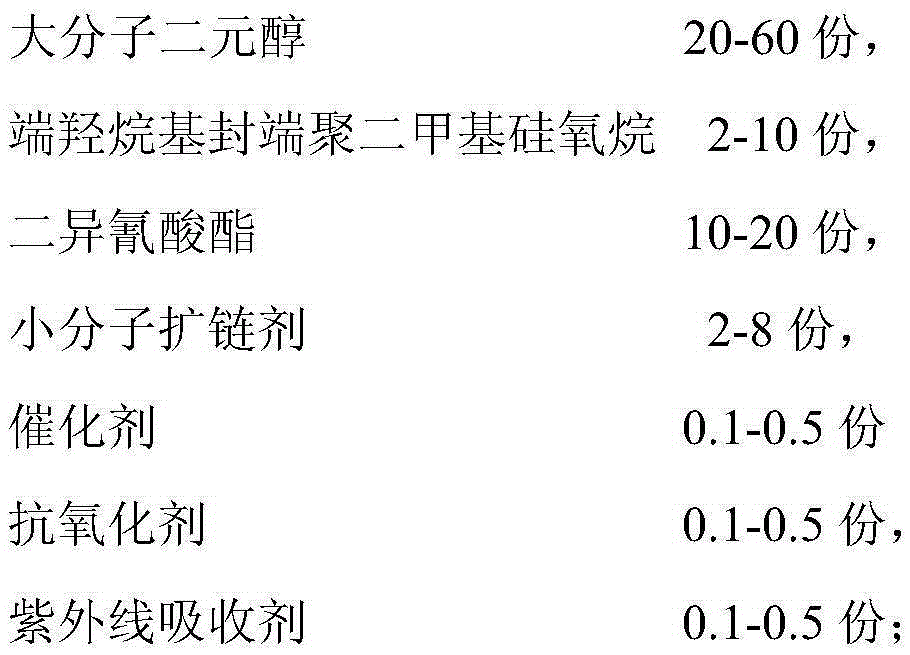
Organic silicon modified thermoplastic polyurethane elastomer (TPU) and preparation method thereof
ActiveCN105399917AImprove hydrophobicityImprove thermal stabilityChemical structureStructural regularity
The invention belongs to the field of high polymer material research, and provides an organic silicon modified thermoplastic polyurethane elastomer (TPU) in order to overcome the defects of poor temperature resistance and hydrophobic property of the existing TPU. The elastomer consists of the following components in parts by weight: 20-60 parts of macromolecular dihydric alcohol, 2-10 parts of terminal hydroxyalkyl end-blocked polydimethylsiloxane, 10-20 parts of diisocyanate, 2-8 parts of a micromolecular chain extender, 0.1-0.5 part of a catalyst, 0.1-0.5 part of an antioxidant and 0.1-0.5 part of an ultraviolet light absorber. Due to the unique chemical structure and composition, organic silicon has a series of excellent properties, such as high temperature and low temperature resistance and hydrophobic property, which just can make up the defects of the TPU. The invention also provides a preparation method of the elastomer, the preparation method adopts a prepolymer two-step process and is less in side reaction, and the prepared TPU is good in structural regularity.
Owner:HANGZHOU JIHUA POLYMER MATERIAL CO LTD
Electrolyte comprising eutectic mixture and electrochemical device using the same
ActiveUS20070042266A1Hybrid capacitor electrolytesElectrolytic capacitorsEvaporationElectrochemical window
Disclosed is an electrolyte comprising a eutectic mixture formed of: (a) an amide group-containing compound; and (b) a lithum-free ionizable salt. An electrochemical device comprising the electrolyte is also disclosed. The electrolyte improves the quality of and electrochemical device due to the excellent conductivity of the metal cation contained in the eutectic mixture, a broad electrochemical window and low viscosity. Additionally, since the eutectic mixture has excellent thermal and chemical stability, it is possible to solve the problems of evaporation, exhaustion and ignition of electrolytes, to minimize side reactions between constitutional elements of the device and the electrolyte, and to improve the safety of the electrochemical device.
Owner:LG ENERGY SOLUTION LTD
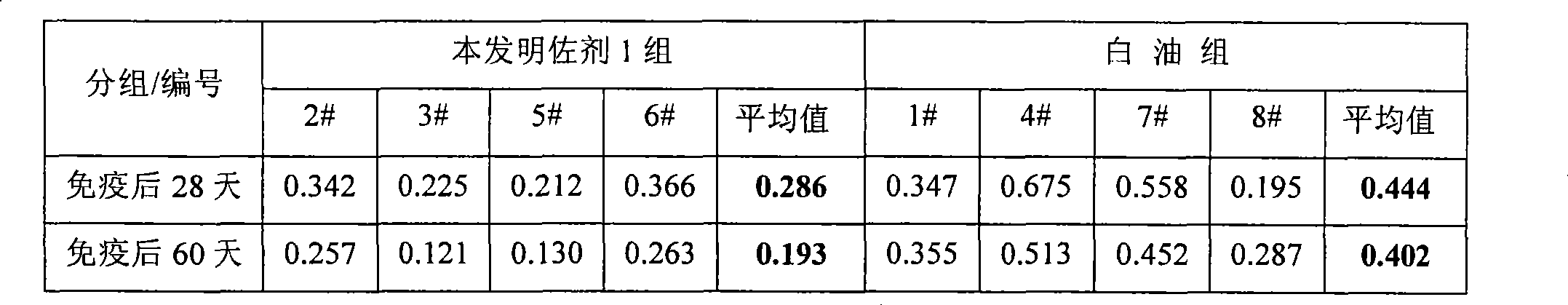
Novel vaccine adjuvant
ActiveCN101428145AGood immune boosterWide range of usePowder deliveryViral antigen ingredientsEmulsionBody fluid
The invention discloses a nanometer particle adjuvant, a preparation method thereof, and the application thereof in the preparation of inactivated vaccines. According to the invention, a novel nanometer particle adjuvant is obtained through preparation by the emulsion preparation and the nanometer treatment following the dissolving of different immunopotentiator and surfactant in water and oil respectively. The adjuvant provided by the invention not only can remarkably improve the humoral immune response and the cellular immune response of various bacterial and viral inactivated vaccines, butalso has unique advantages in the preparation process, the stability, the immunity duration and the side reaction of vaccines.
Owner:BEIJING CENT BIOLOGY
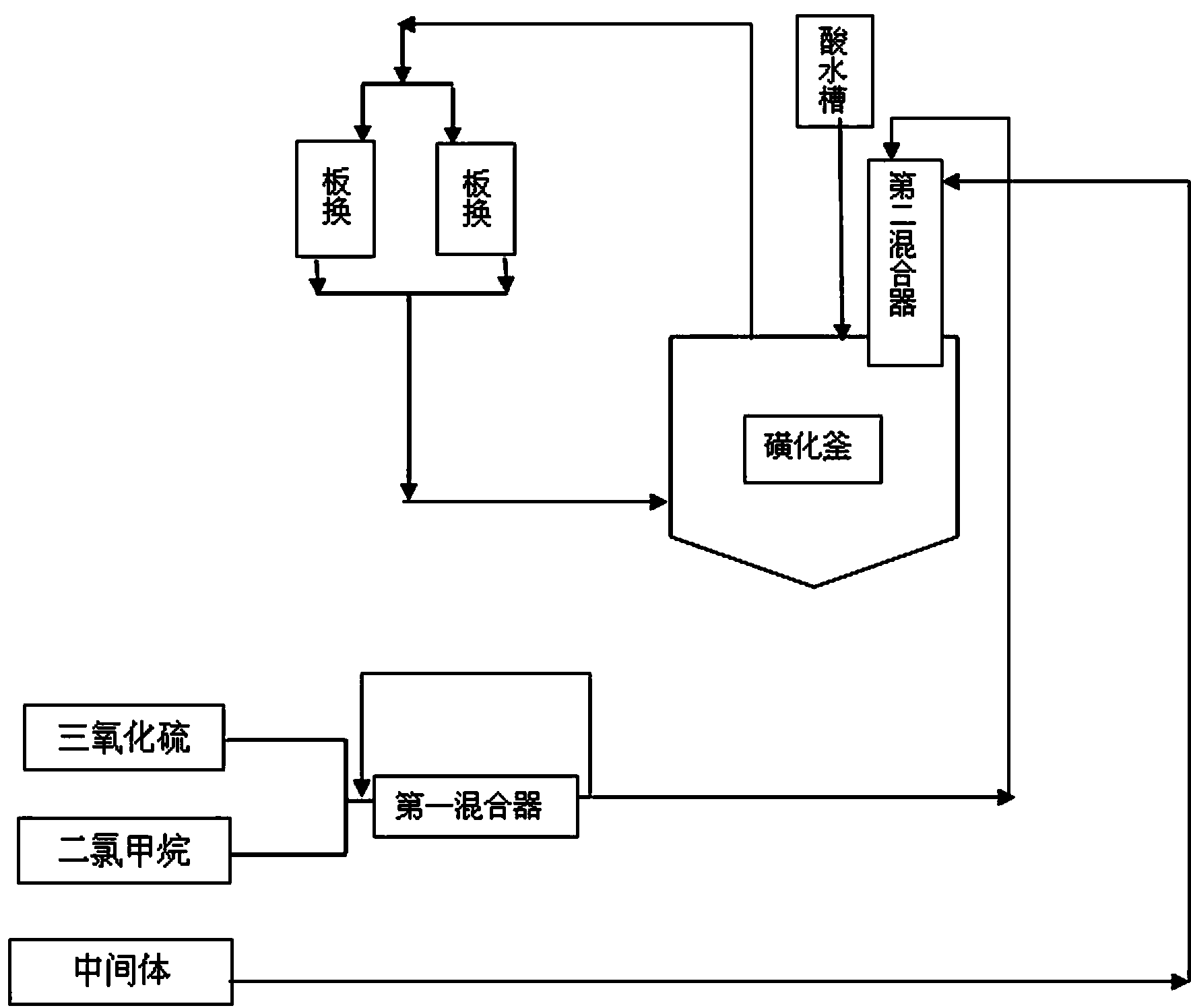
Acesulfame potassium cyclization continuous production method
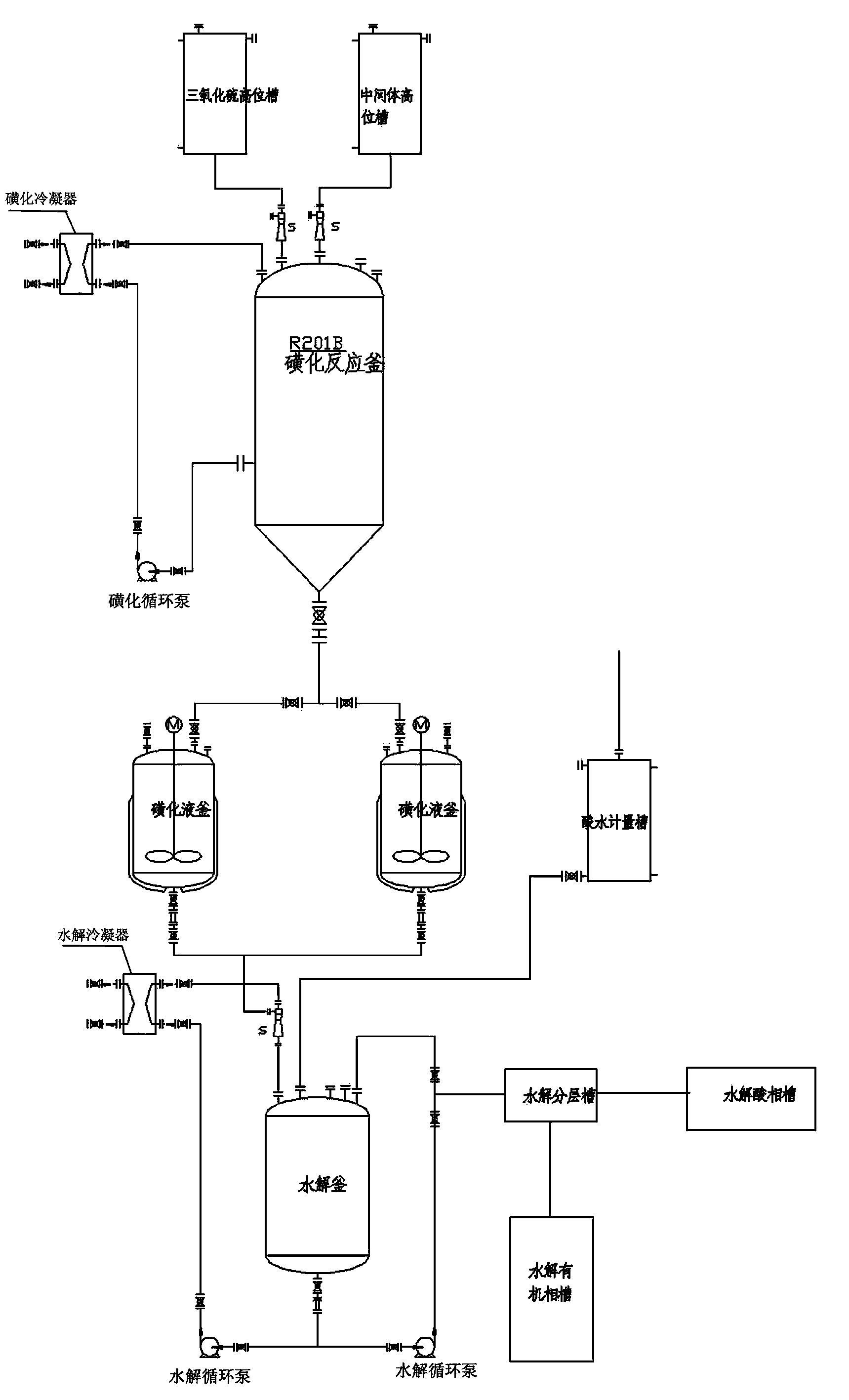
ActiveCN103613566AImprove continuityReduce labor intensityOrganic chemistryReaction temperatureSulfur trioxide
The invention relates to an acesulfame potassium cyclization continuous production method. The method is characterized in that the sulfonation reaction and hydrolysis reaction comprise the following steps: step a, separately pumping an intermediate generated during a synthesis reaction and sulfur trioxide into a sulfonation reactor in a certain speed, carrying out sulfonation reactions in the sulfonation reactor; step b, gasifying dichloromethane when the concentration of the reactants in the sulfonation reactor reach a certain level, spraying the sulfonation liquid into a hydrolysis reactor; step c, dropwise adding acidic water into the hydrolysis reactor to carry out hydrolysis reactions. The method has the advantages that an cyclization one-step reaction technology is adopted, thus the continuity of production operation is realized, and the work strength of workers is reduced; compared to the conventional intermittent production technology, the one-step reaction method has a higher stability, improves the service life of the reactor, shortens the reaction time, and reduces the side reactions. Furthermore, in the method, the sulfonation reaction temperature is raised, then dichloromethane gasification is utilized to reduce the reaction temperature, so that low temperature production is avoided, and a deep cooling ice machine is stopped, so the production efficient is greatly improved, and the energy consumption is largely reduced.
Owner:ANHUI JINGHE IND
Method for preparing low-melting-point polyester composite fiber by adopting continuous polymerization and melt direct-spinning
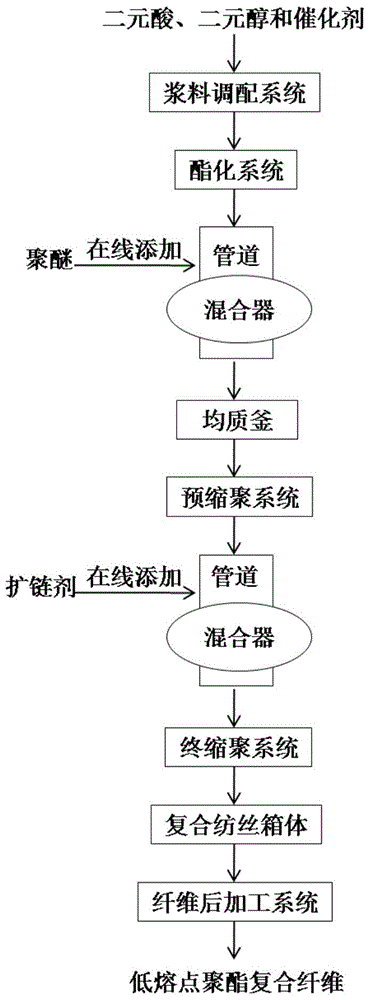
ActiveCN105063797AImprove thermal stabilitySuppress thermal degradation side reactionsArtificial filament heat treatmentConjugated synthetic polymer artificial filamentsOligomerTransesterification
The invention relates to a method for preparing a modified copolyester composite fiber, in particular to a method for preparing a low-melting-point polyester composite fiber by adopting continuous polymerization and melt direct-spinning. The method comprises the following steps: slurry is prepared from raw materials in proportion and added continuously into an esterification system for an esterification reaction, an obtained oligomer is uniformly mixed with polyether, the mixture enters a homogenizing kettle for transesterification and pre-polycondensation, a product is uniformly mixed with a chain extender for final polycondensation, and an obtained low-melting-point polyester melt and a common polyester melt enter a composite spinning assembly to prepare the low-melting-point polyester composite fiber which comprises a common polyester core layer and a low-melting-point polyester cortex layer. The method has the advantages as follows: a side reaction of thermal degradation of polyether components is inhibited effectively, and the color and spinnability of the low-melting-point polyester melt are improved; the problem of poor performance of composite spinning of the low-melting-point polyester on the cortex layer and the common polyester on the core layer due to large dynamic viscosity difference of the melt is solved, and the composite fiber is well formed; the production efficiency of the low-melting-point polyester composite fiber is improved.
Owner:CHINESE TEXTILE ACAD
Method used for preparing methyl acetate
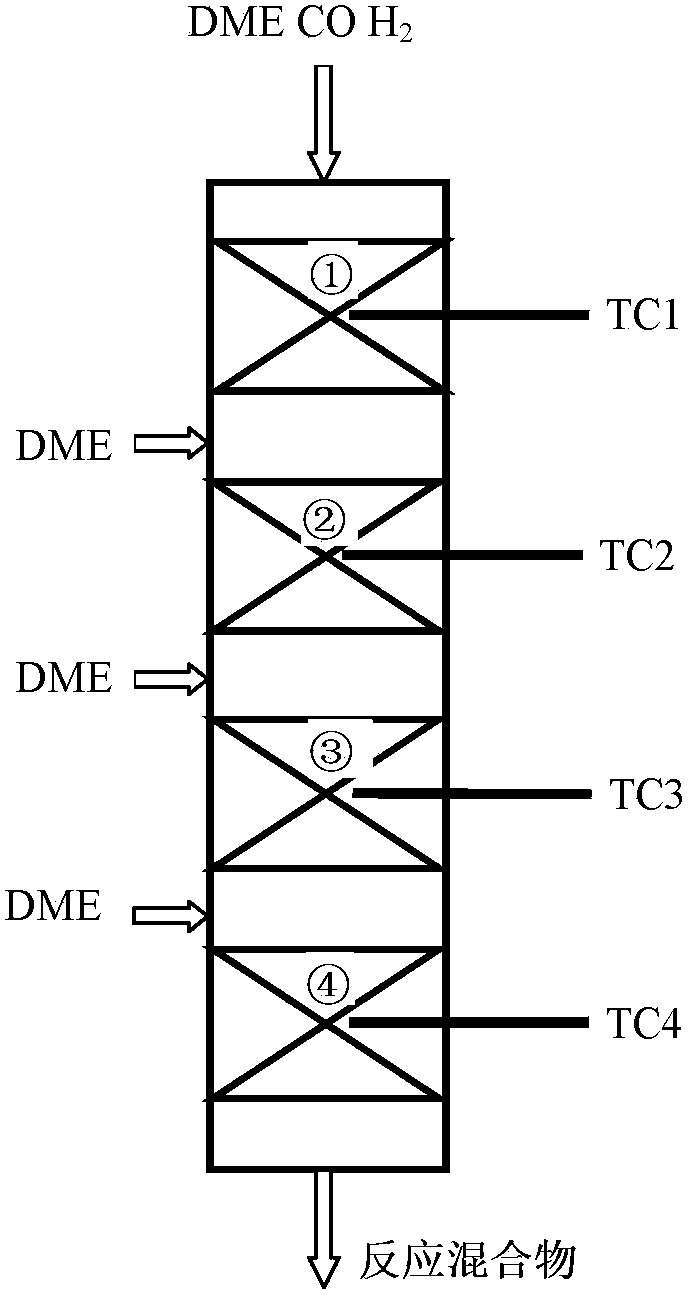
ActiveCN103896768AExtend your lifeControl temperature distributionMolecular sieve catalystsCatalyst activation/preparationMolecular sieveHydrogen
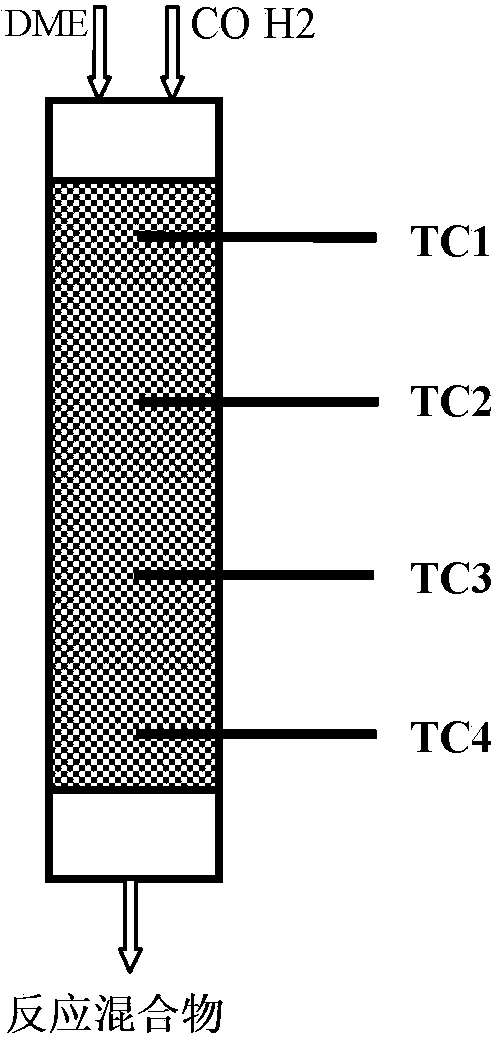
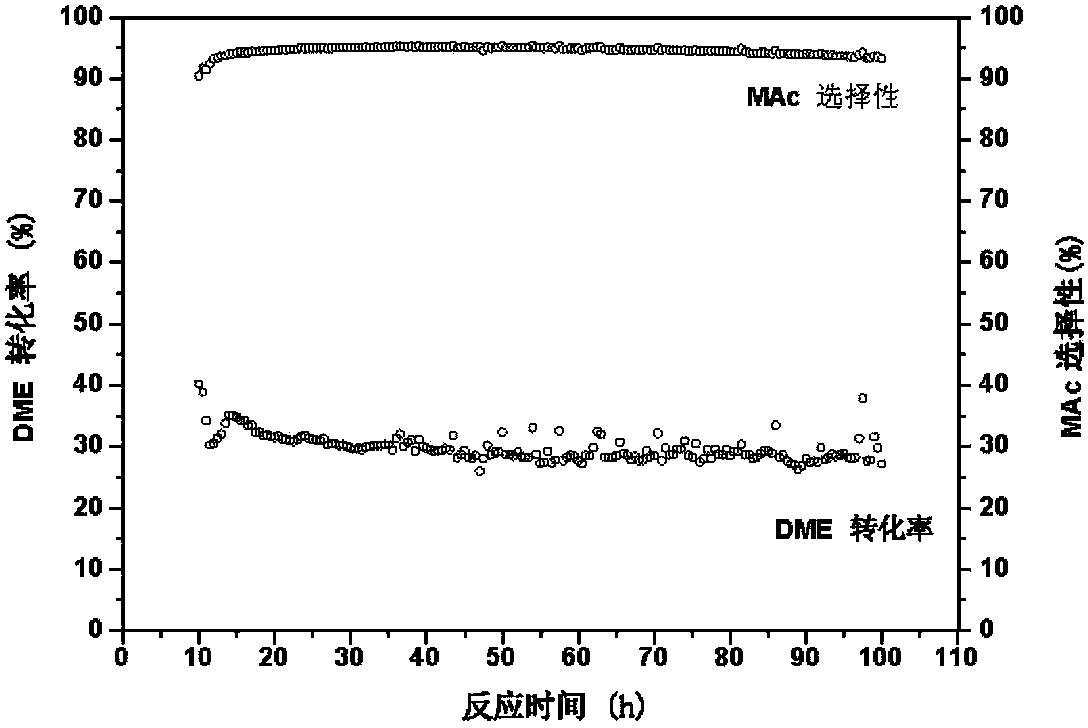
Provided in the present invention is a process for preparing methyl acetate through carbonylation of dimethyl ether, comprising: passing a feed gas containing dimethyl ether, carbon monoxide, and optional hydrogen gas through a reactor loaded with a mordenite molecular sieve catalyst, and preparing methyl acetate by reacting same at a reaction temperature of 19 to 320°C, a reaction pressure of 0.5 to 20.0 Mpa, and a gas volume space velocity of 500 to 5000 h-1; wherein the mordenite molecular sieve catalyst is a hydrogen-type mordenite molecular sieve catalyst with an adsorbed organic amine, the molar ratio of the dimethyl ether to carbon monoxide in the feed gas is DME / CO = 1 / 1 to 1 / 15, the molar ratio of the hydrogen gas to carbon monoxide is H2 / CO = 0 to 10 / 1, and the dimethyl ether in the feed gas is charged into the reactor by means of feeding in stages. The present invention is characterized in that: through treatment by organic amine adsorption, the temperature distribution of catalyst bed can be effectively controlled so as to avoid the occurrence of hot points, and thereby side reactions are reduced, the selectivity of target product is improved, and the lifetime of the catalyst is greatly extended.
Owner:YANCHANG ZHONGKE (DALIAN) ENERGY TECH CO LTD
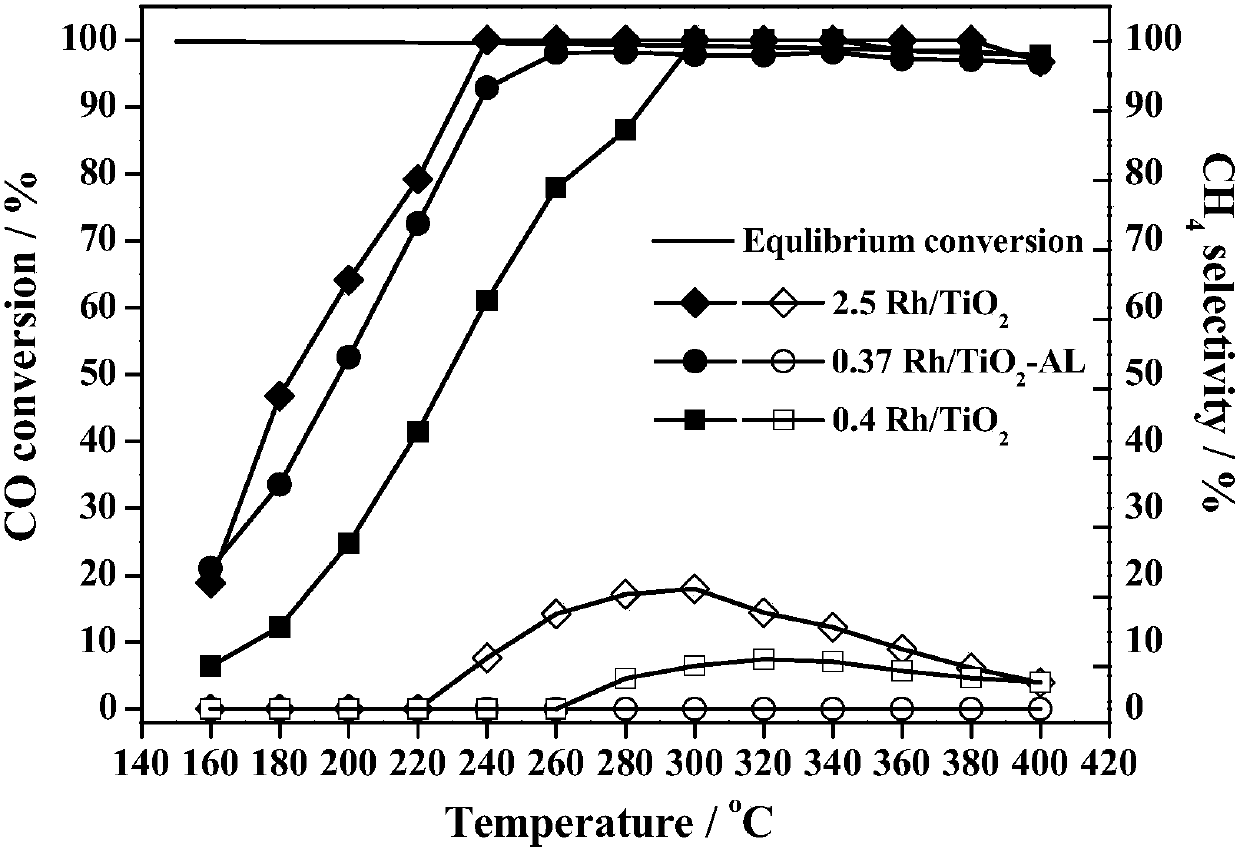
Monoatomic dispersed noble metal catalyst and application thereof
ActiveCN107649124AHave substantive characteristicsEnhance intrinsic reactivityHydrogenCatalyst activation/preparationIridiumMethanation
The invention relates to a monoatomic dispersed platinum group metal based catalyst with a higher loading capacity and an application of the catalyst. Specifically, the invention relates to a bicomponent catalyst of titanium oxide and one selected from metals rhodium (Rh), ruthenium (Ru), platinum (Pt), iridium (Ir) and palladium (Pd) as well as preparation and an application of the catalyst. Themetal is highly dispersed in the form of a single atom on the carrier titanium oxide, and the metal content is up to 0.4-2%. The catalyst provided by the invention is suitable for the CO water gas shift reaction, and can improve the CO conversion rate and inhibit a methanation side reaction; and meanwhile, the catalyst also has very good low-temperature activity and use stability on catalytic decomposition of the liquid single-component propellant level anhydrous hydrazine used in a satellite attitude control engine in the aerospace industry.
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
Metal-organic frame material based composite battery diaphragm and preparation method and application thereof
InactiveCN108807798AAvoid direct contactAvoid short circuitFuel and secondary cellsNon-aqueous electrolyte accumulatorsPorosityIon transport number
The invention discloses a metal-organic frame material based composite battery diaphragm and a preparation method and application thereof. The preparation method comprises the following steps: (1), synthesizing a metal-organic frame material precursor; (2), compounding the metal-organic frame material precursor and a two-dimensional material or a polymer material to obtain the metal-organic framematerial based composite battery diaphragm. The diaphragm is high in porosity and large in specific surface area, the electrolyte wettability of the diaphragm can be improved, and the ion transport number of the diaphragm is greatly increased; the diaphragm has the advantage of an adjustable pore size, and through a suitable pore size, shuttling of electrolyte ions can be effectively controlled, occurrence of adverse side reactions can be inhibited, the battery capacity can be increased and the cycle life can be prolonged; through a uniform pore structure, the passing ions can be uniformly dispersed on the surface of an electrode, so that growth of dendrites are fundamentally inhibited, the cycle life of a battery is effectively prolonged, and the safety performance of the battery is improved; the metal-organic frame material based composite battery diaphragm has good flexibility and mechanical properties and can be applied to assembly of a practical soft pack battery.
Owner:NANJING UNIV