Patents
Literature
10443results about "Hydrogen" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Apparatus, system, and method for generating a gas from solid reactant pouches
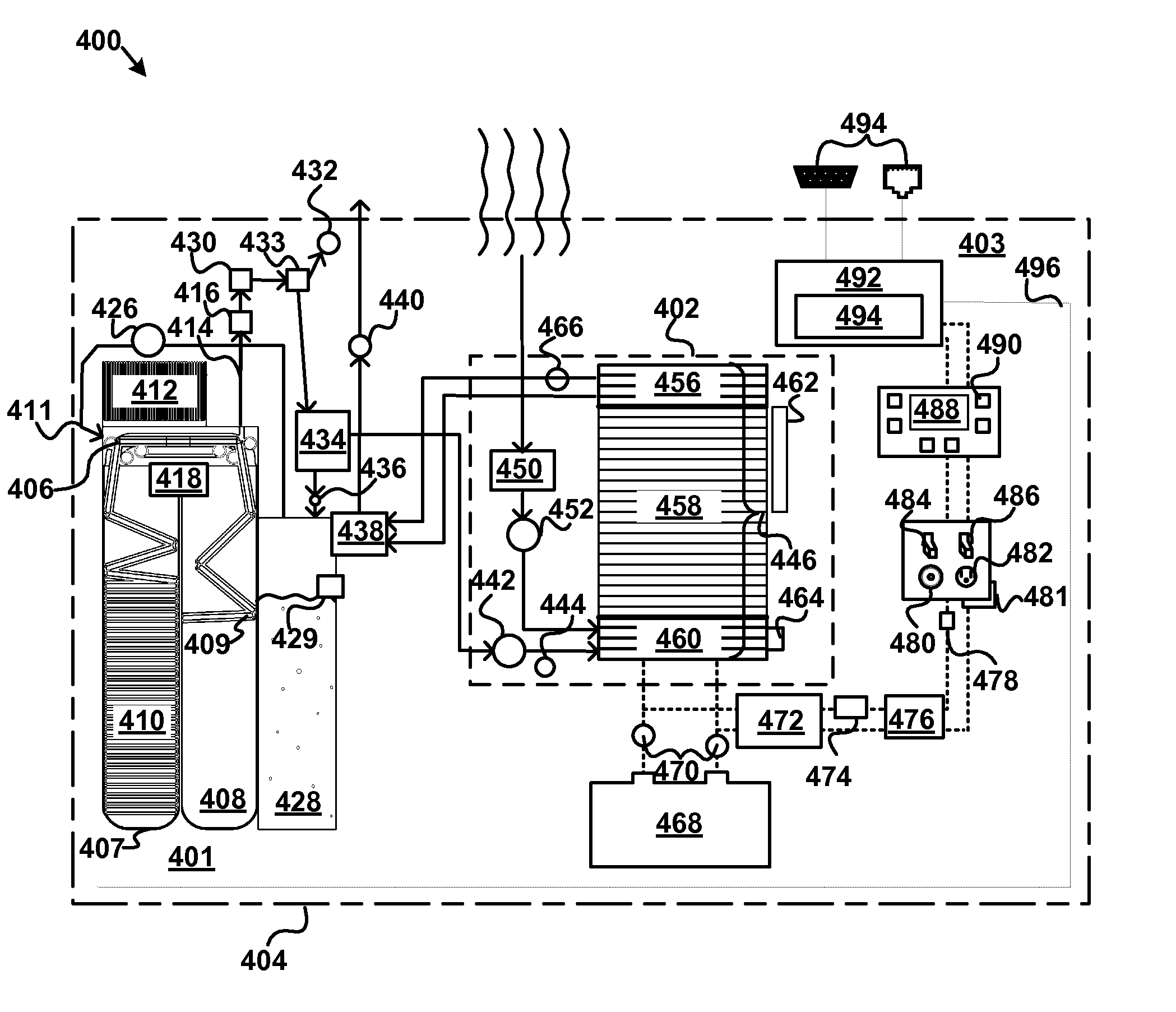
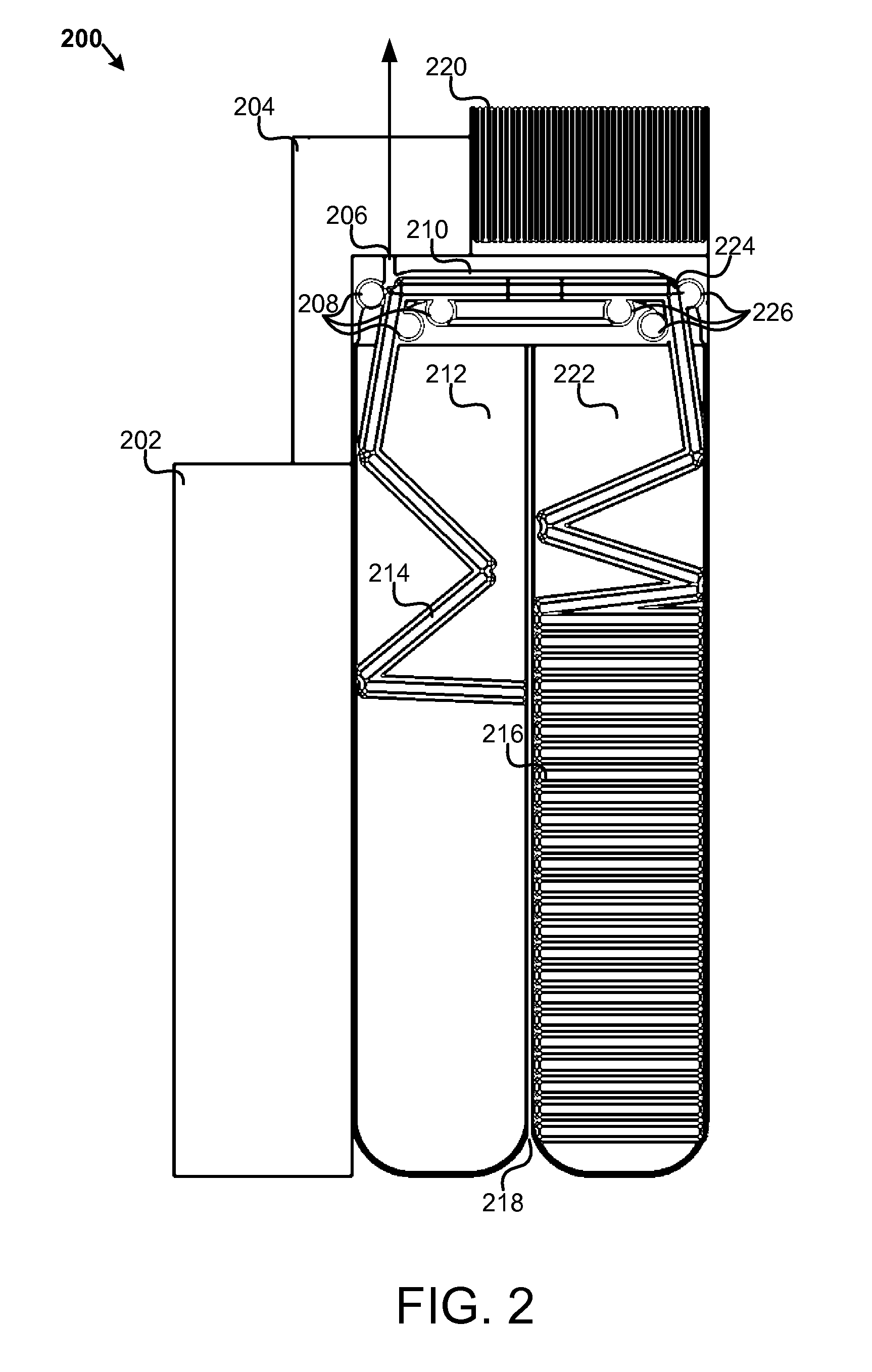
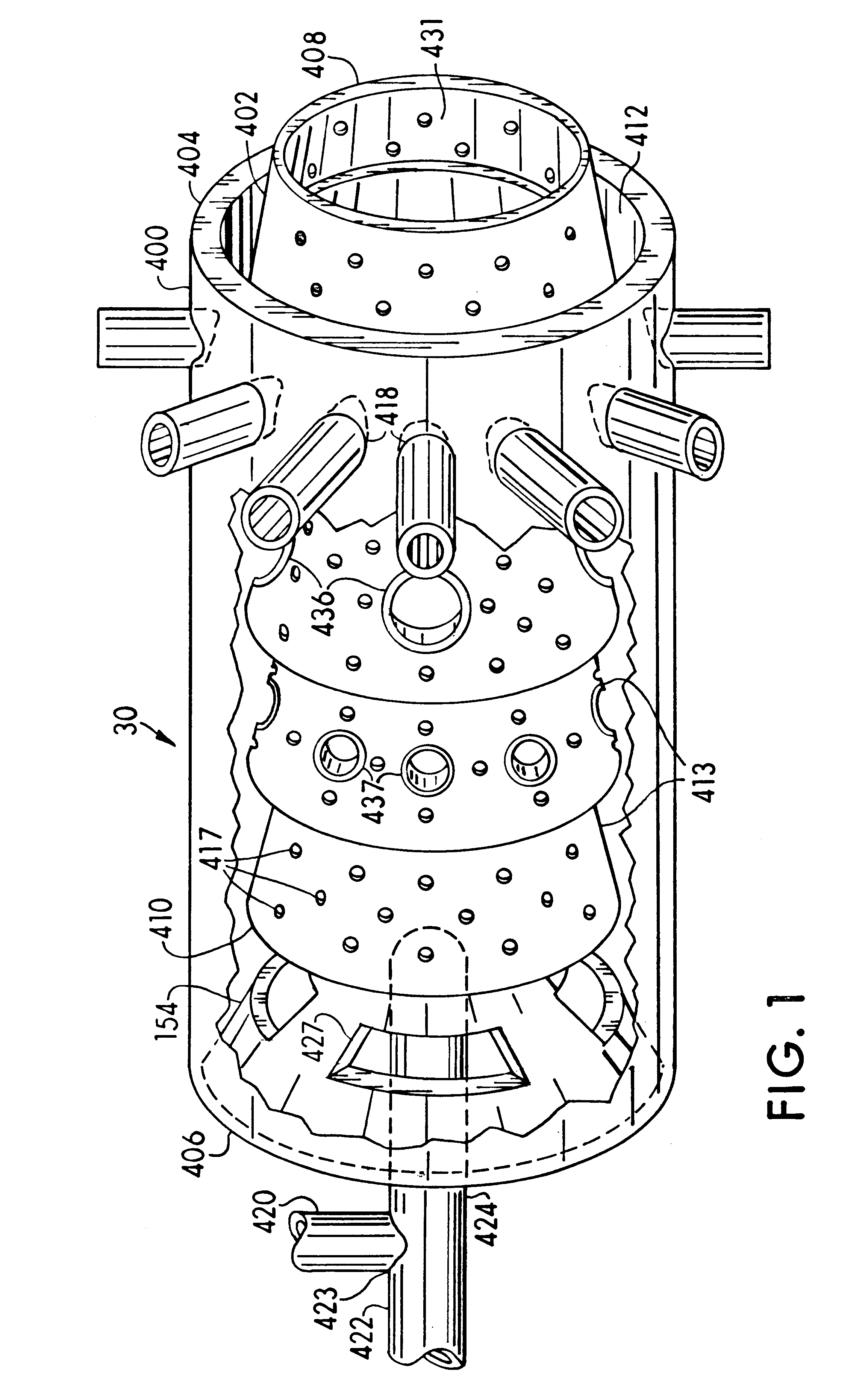
An apparatus, system, and method are disclosed for generating a gas. One or more liquid permeable pouches each define a cavity that contains a solid anhydrous reactant, such as a chemical hydride. A reaction chamber made of a heat, chemical and / or pressure resistant material receives the one or more pouches from a pouch feeder that transfers the one or more pouches into the reaction chamber successively at a feed rate. One or more liquid sources inject a liquid reactant into the reaction chamber so that the liquid reactant contacts a portion of the one or more pouches. The one or more liquid sources inject the liquid reactant at an injection rate that corresponds to the feed rate. A gas outlet releases a gas, such as hydrogen, oxygen, ammonia, borazine, nitrogen, or a hydrocarbon, that is produced by a reaction between the solid reactant and the liquid reactant.
Owner:TRULITE INC
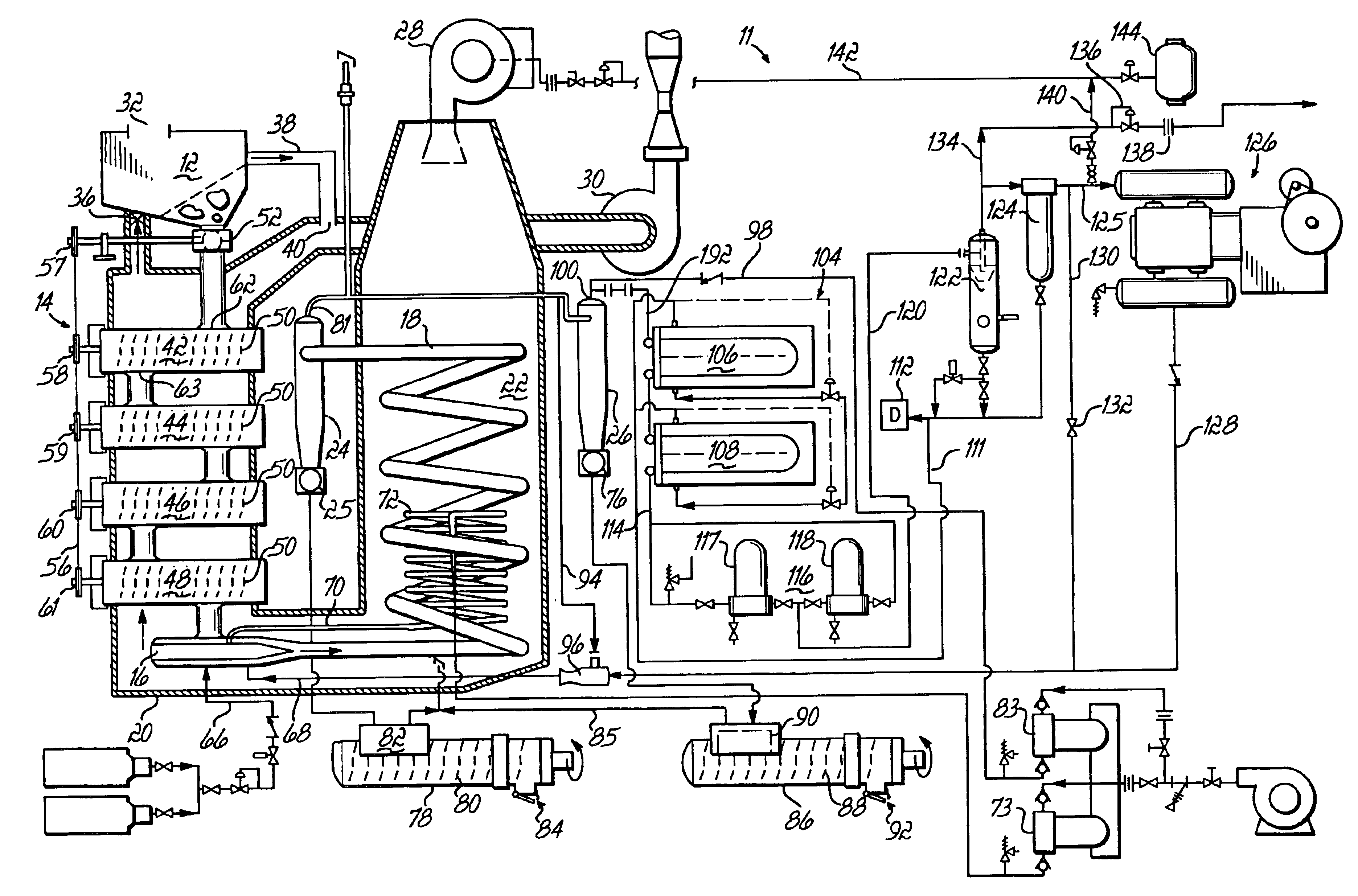
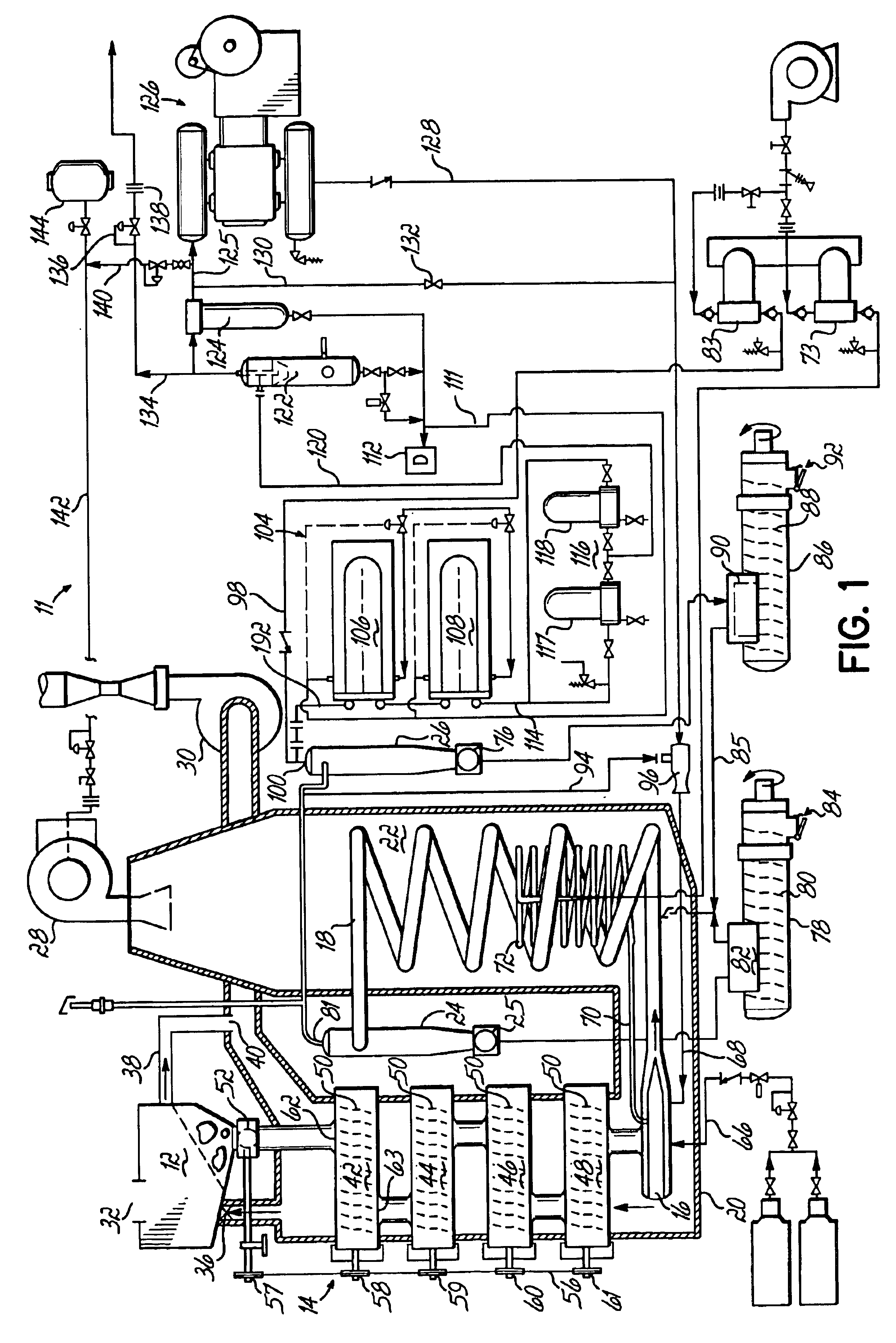
Staged combustion of a low heating value fuel gas for driving a gas turbine
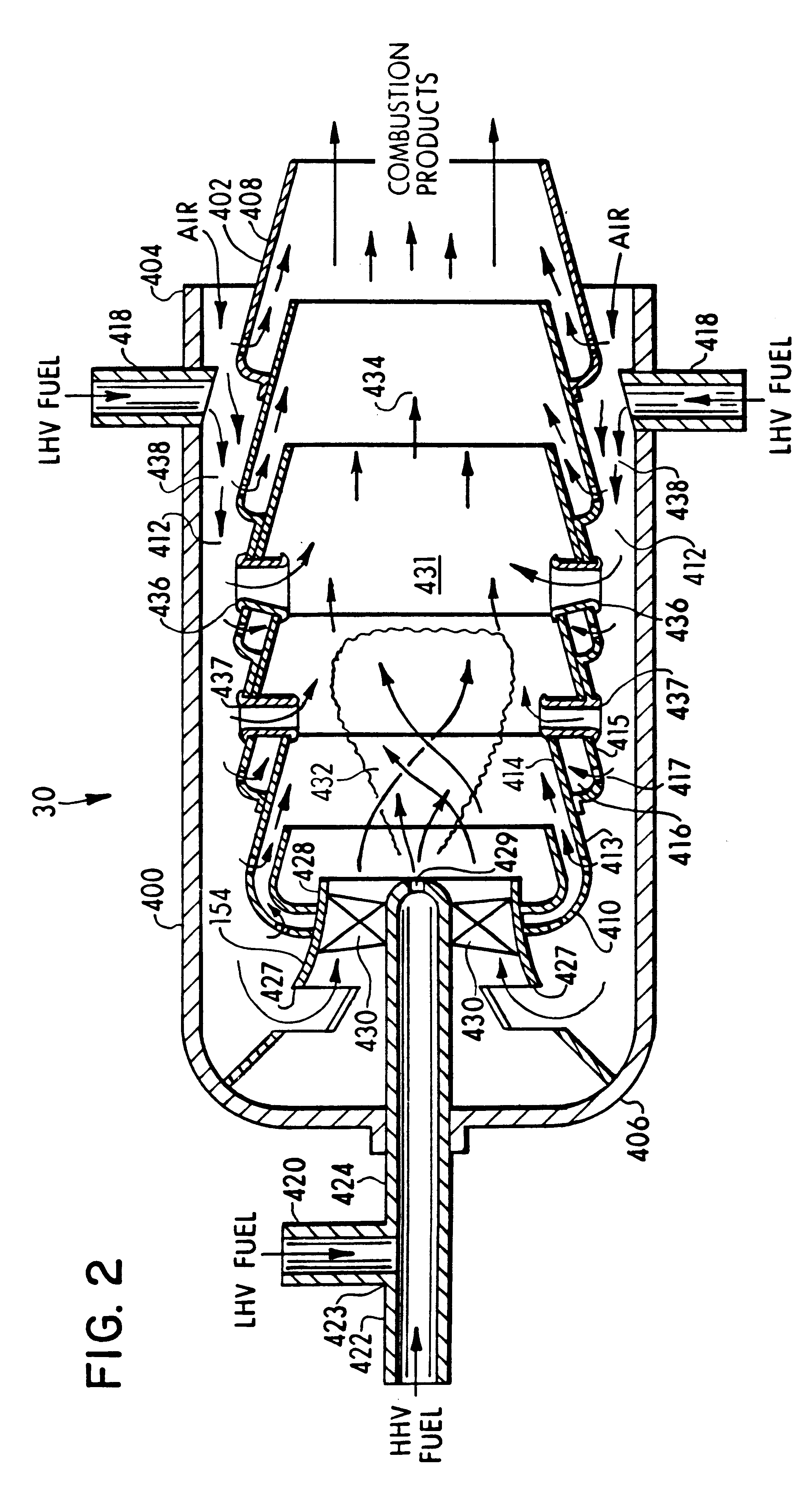
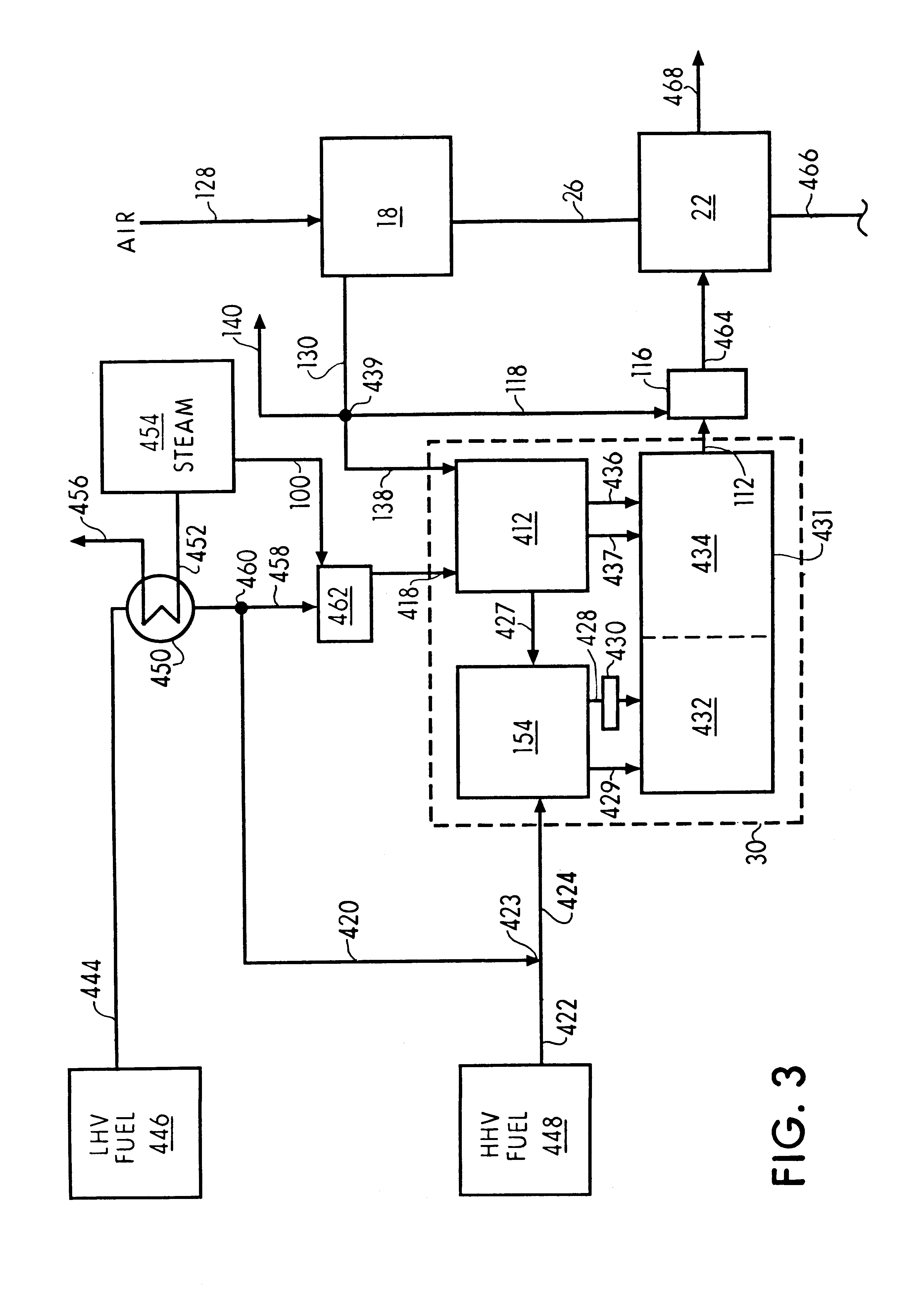
A process is provided for combusting a low heating value fuel gas in a combustor to drive an associated gas turbine. A low heating value fuel gas feed is divided into a burner portion and a combustion chamber portion. The combustion chamber portion and a combustion air are conveyed into a mixing zone of the combustor to form an air / fuel mixture. The burner portion is conveyed into a flame zone of the combustor through a burner nozzle while a first portion of the air / fuel mixture is conveyed into the flame zone through a burner port adjacent to the burner nozzle. The burner portion and first portion of the air / fuel mixture are contacted in the flame zone to combust the portions and produce flame zone products. The flame zone products are conveyed into an oxidation zone of the combustor downstream of the flame zone while a second portion of the air / fuel mixture is also conveyed into the oxidation zone. The second portion is combusted in the oxidation zone in the presence of the flame zone products to produce combustion products. The combustion products are conveyed into the associated gas turbine and drive the gas turbine.
Owner:MARATHON OIL CO +1
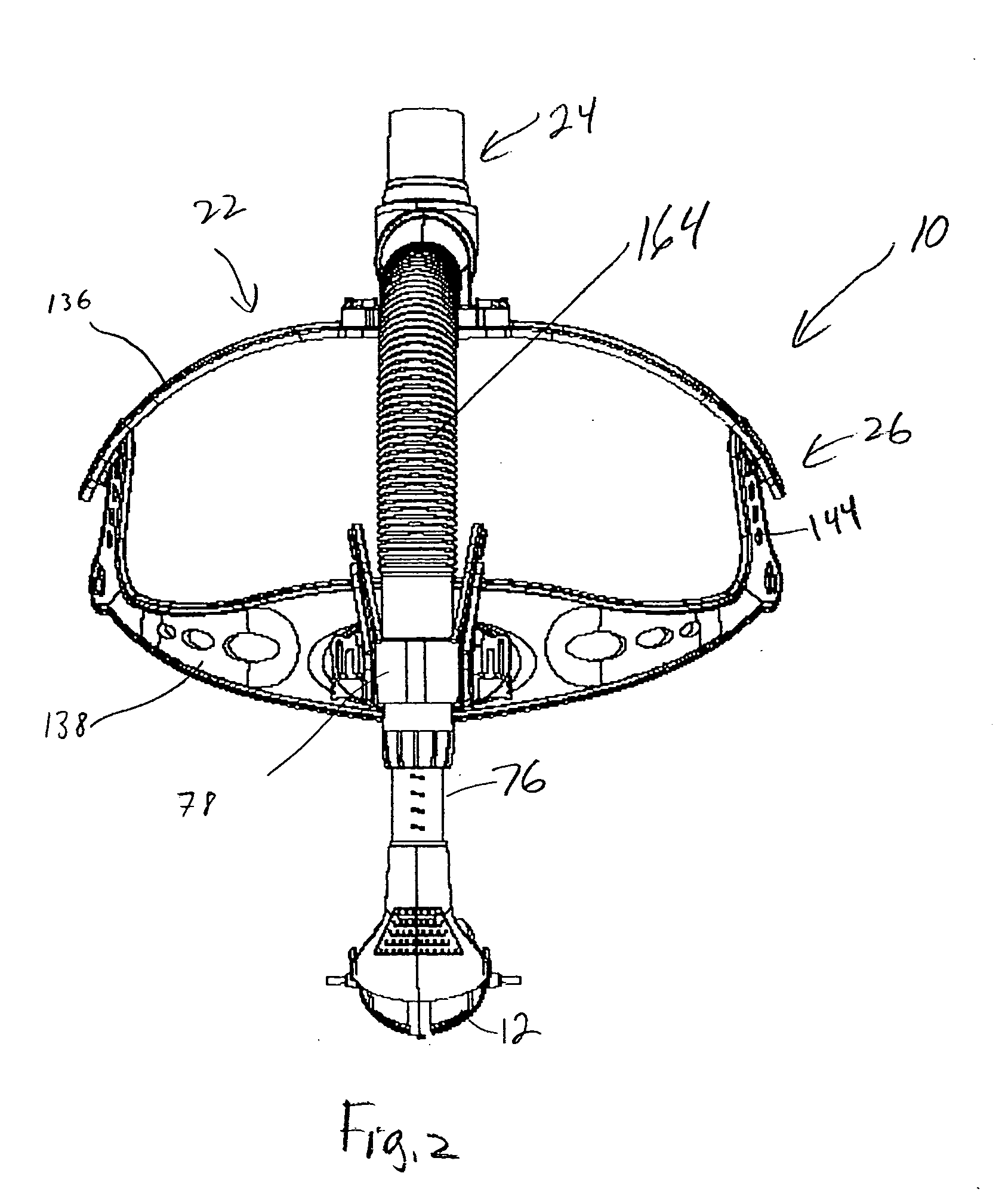
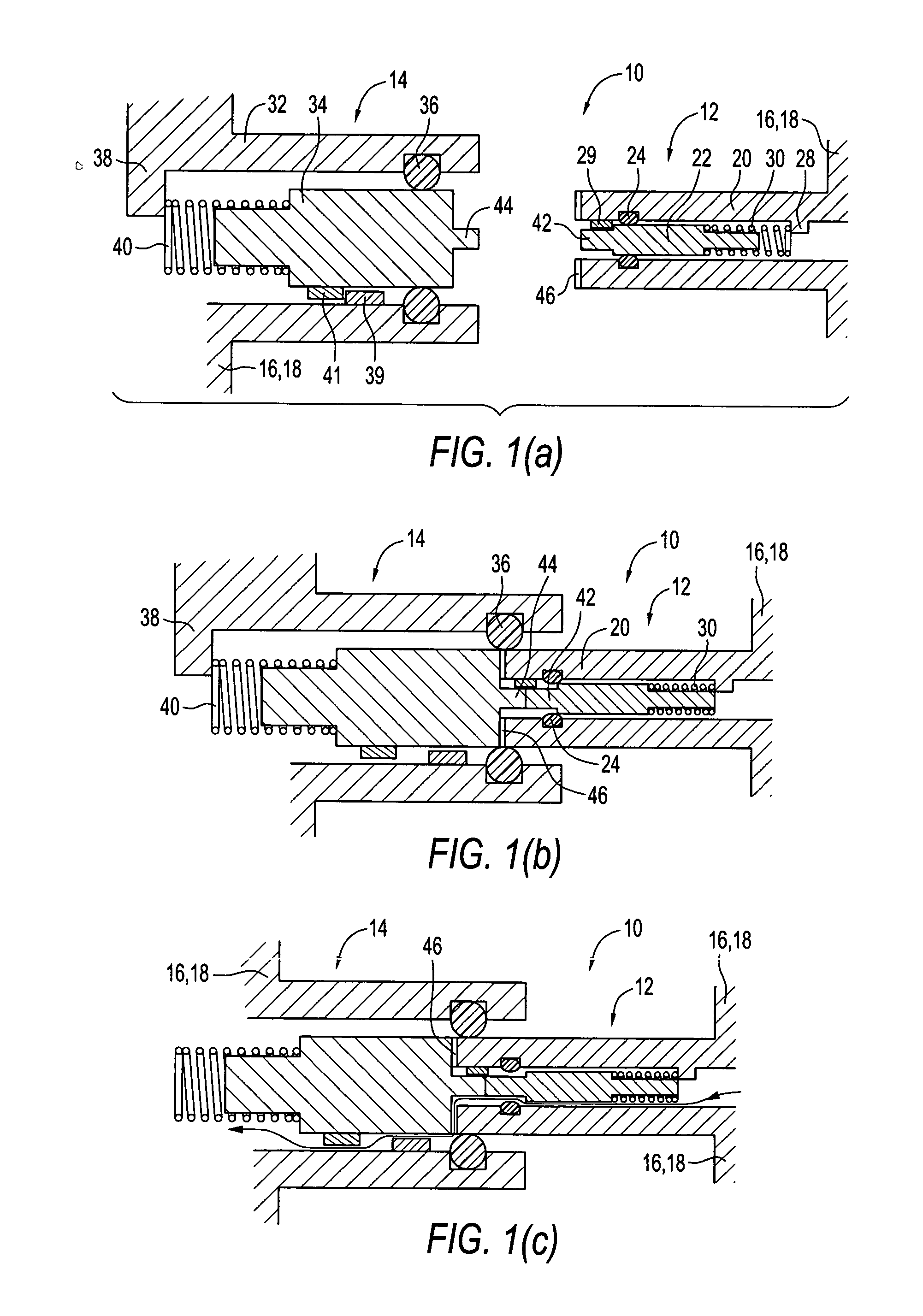
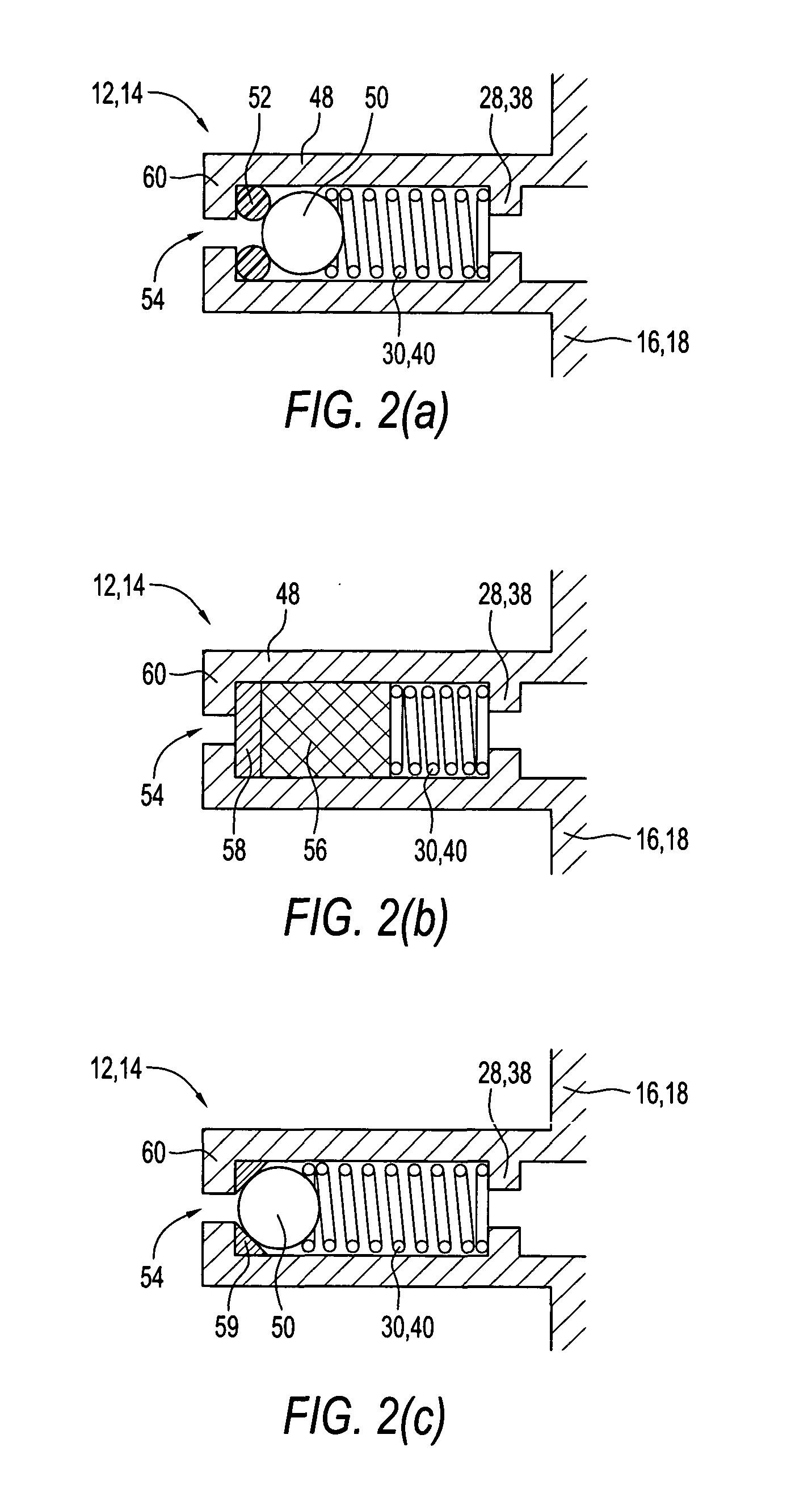
Patient inteface assembly and system using same
ActiveUS20050076913A1Improved patient interface stability and overall comfortOvercomes shortcomingPhysical therapyBreathing filtersNasal cavityMinimal contact
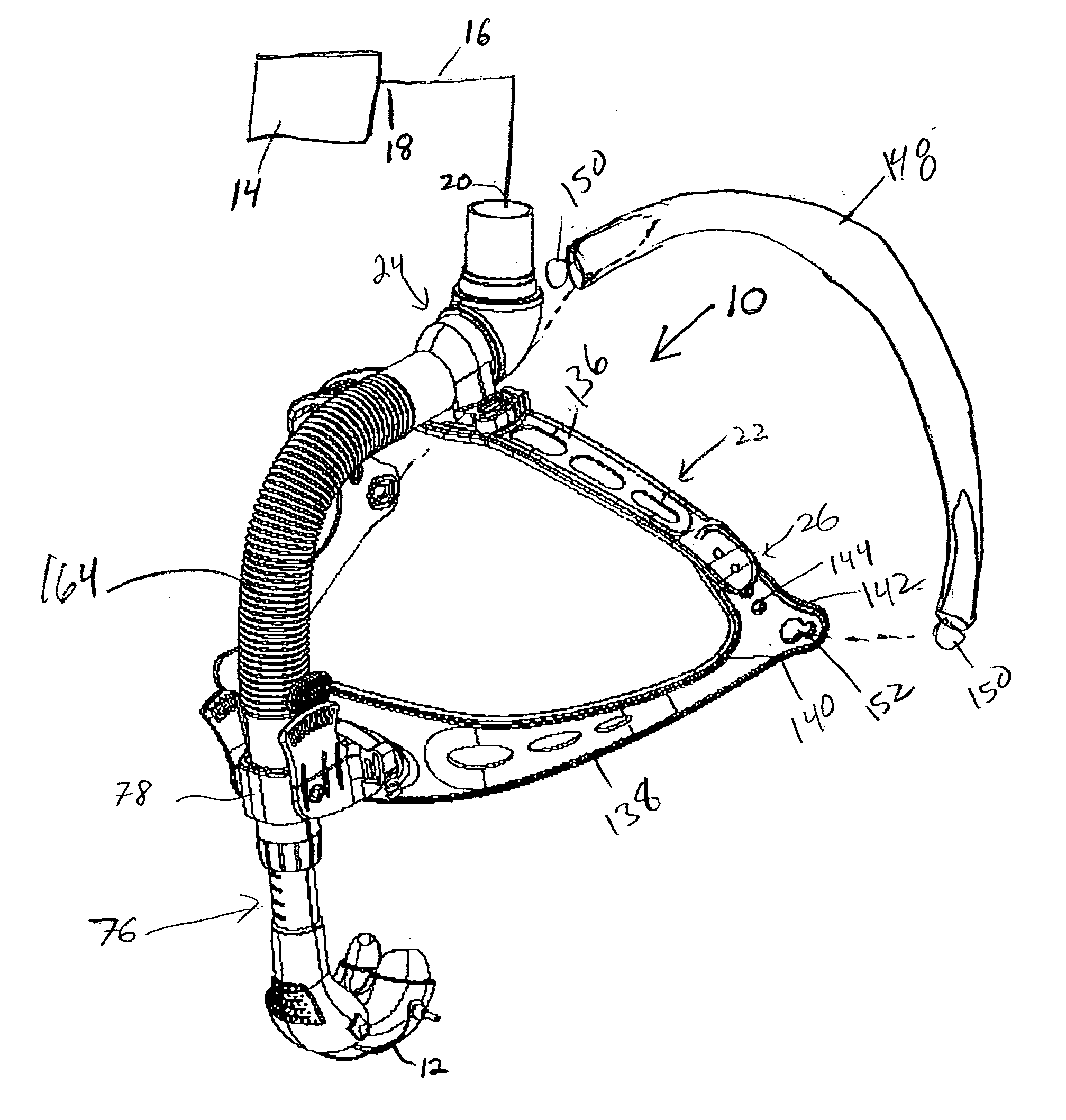
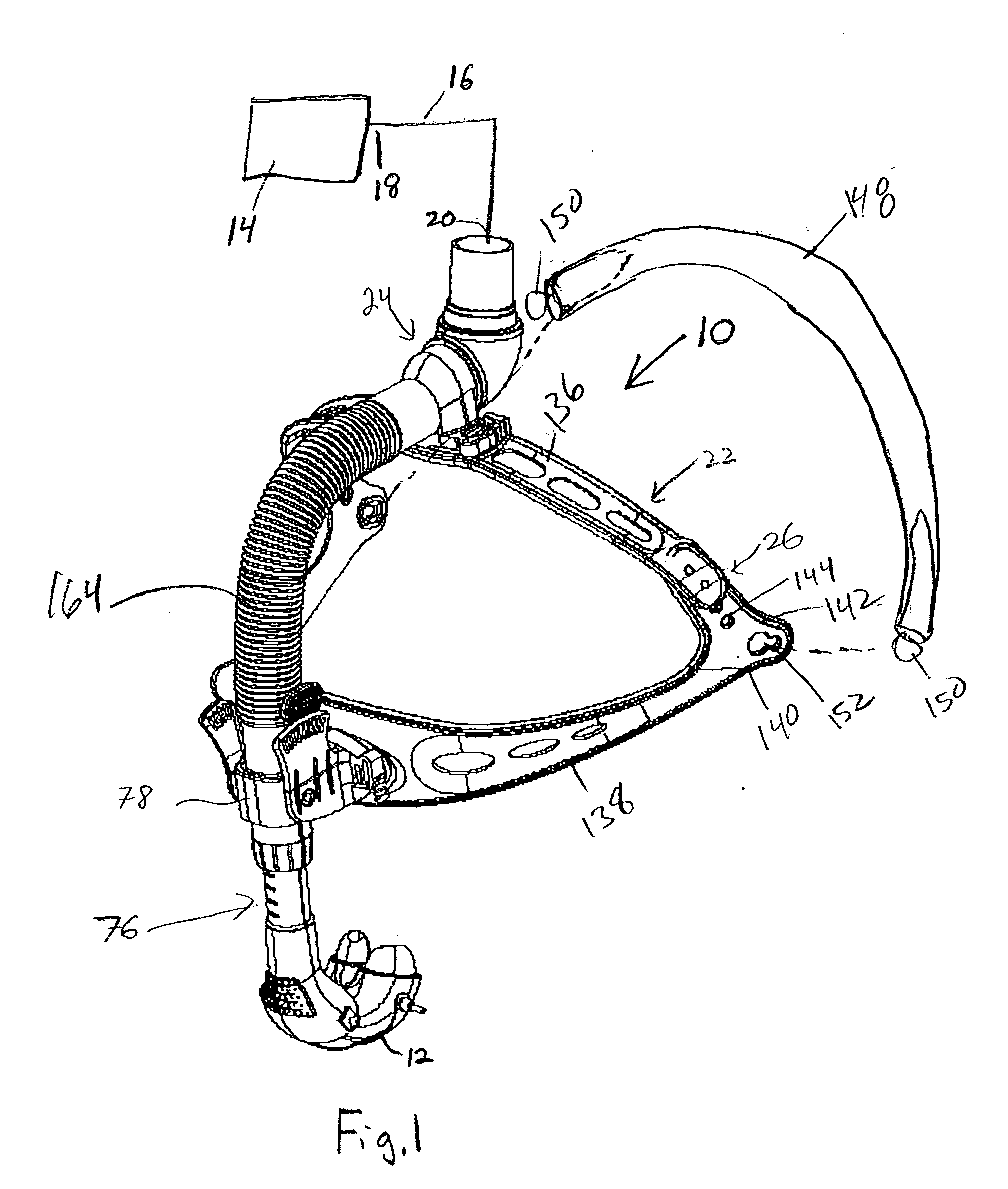
A patient interface assembly that includes a patient interface device, a headgear, and coupling member joining the interface device to a headgear assembly. In one embodiment, a spring biases the patient interface device against the patient's face when in use. In a further embodiment, the patient interface device is a nasal cushion that includes a formable support mounted to the nasal cushion for providing support and adjustment of the nasal cushion to improve fit and comfort. The headgear assembly in one embodiment is a semi-rigid, minimal contact harness assembly and includes an adjustment assembly that allows for a simultaneous adjustment of multiple straps. An adjustment assembly on the headgear provide a gross adjustment of the position of the interface relative to the patient and a biasing force to urge the patient interface device against the patient's face. In a further embodiment, a pair of rigid connecting members coupling the patient interface device to the headgear.
Owner:RIC INVESTMENTS LLC
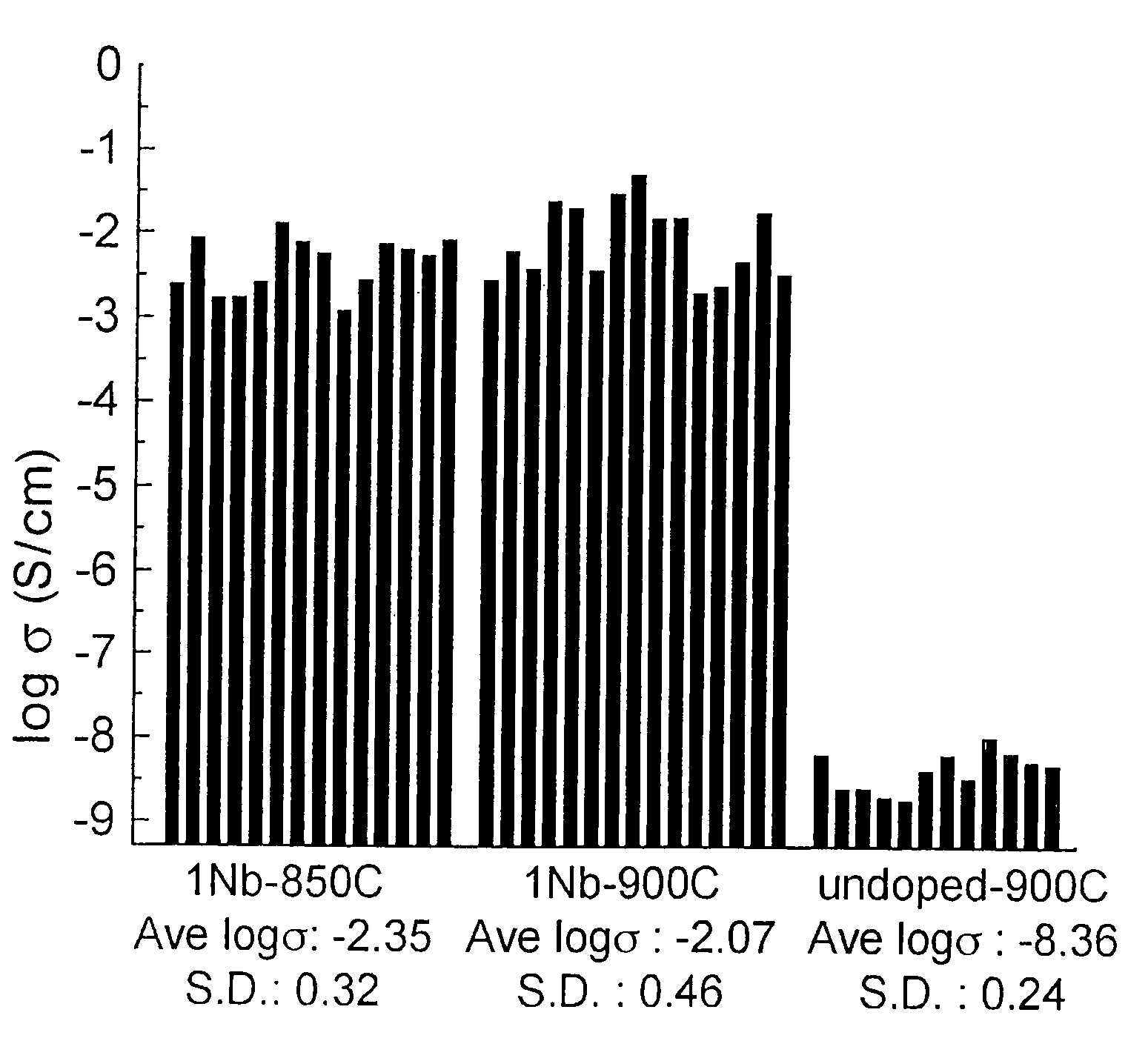
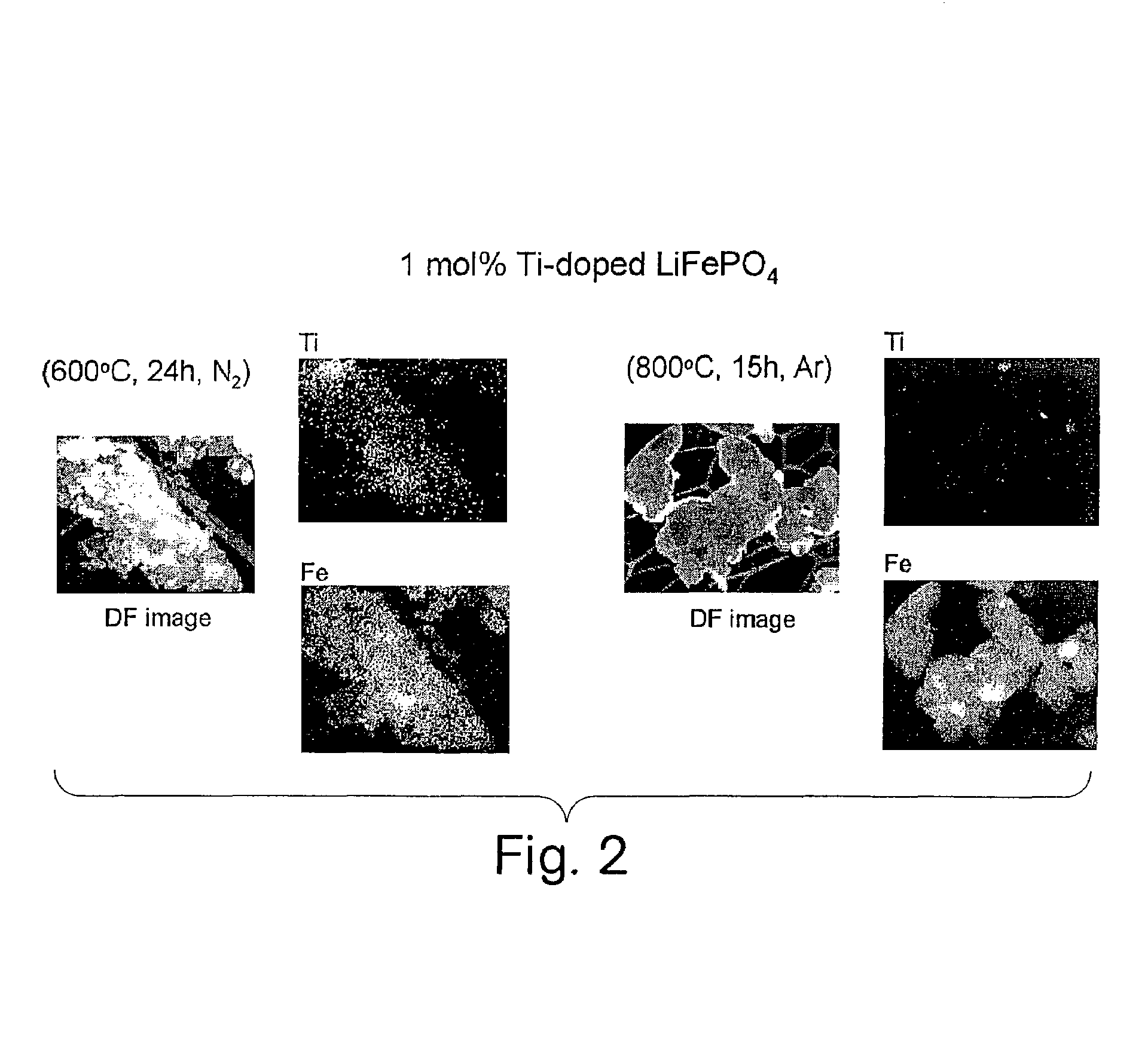
Conductive lithium storage electrode
A compound comprising a composition Ax(M′1-aM″a)y(XD4)z, Ax(M′1-aM″a)y(DXD4)z, or Ax(M′1-aM″a)y(X2D7)z, and have values such that x, plus y(1-a) times a formal valence or valences of M′, plus ya times a formal valence or valence of M″, is equal to z times a formal valence of the XD4, X2D7, or DXD4 group; or a compound comprising a composition (A1-aM″a)xM′y(XD4)z, (A1-aM″a)xM′y(DXD4)z(A1-aM″a)xM′y(X2D7)z and have values such that (1-a)x plus the quantity ax times the formal valence or valences of M″ plus y times the formal valence or valences of M′ is equal to z times the formal valence of the XD4, X2D7 or DXD4 group. In the compound, A is at least one of an alkali metal and hydrogen, M′ is a first-row transition metal, X is at least one of phosphorus, sulfur, arsenic, molybdenum, and tungsten, M″ any of a Group IIA, IIIA, IVA, VA, VIA, VIIA, VIIIA, IB, IIB, IIIB, IVB, VB, and VIB metal, D is at least one of oxygen, nitrogen, carbon, or a halogen, 0.0001<a≦0.1, and x, y, and z are greater than zero. The compound can have a conductivity at 27° C. of at least about 10−8 S / cm. The compound can be a doped lithium phosphate that can intercalate lithium or hydrogen. The compound can be used in an electrochemical device including electrodes and storage batteries and can have a gravimetric capacity of at least about 80 mAh / g while being charged / discharged at greater than about C rate of the compound.
Owner:MASSACHUSETTS INST OF TECH
Fuel cartridge with connecting valve
A shut-off valve or connecting valve capable of connecting a fuel supply to a fuel cell is disclosed. The valve comprises a first valve component and a second valve component. Each valve component has an outer housing and a biased slidable member disposed inside the housing forming an internal seal. During the connection process, the two valve components establish an inter-component seal. Afterward, in one suitable embodiment the slidable member moves inward and opens the internal seal in the valve component to establish a flow path. In another embodiment, the slidable member moves inward and exposes a first filler and the first filler abuts a second filler in the other valve component to establish a flow path. In other embodiments, at least one valve component is sized and dimensioned to limit access to the internal seal.
Owner:INTELLIGENT ENERGY LTD
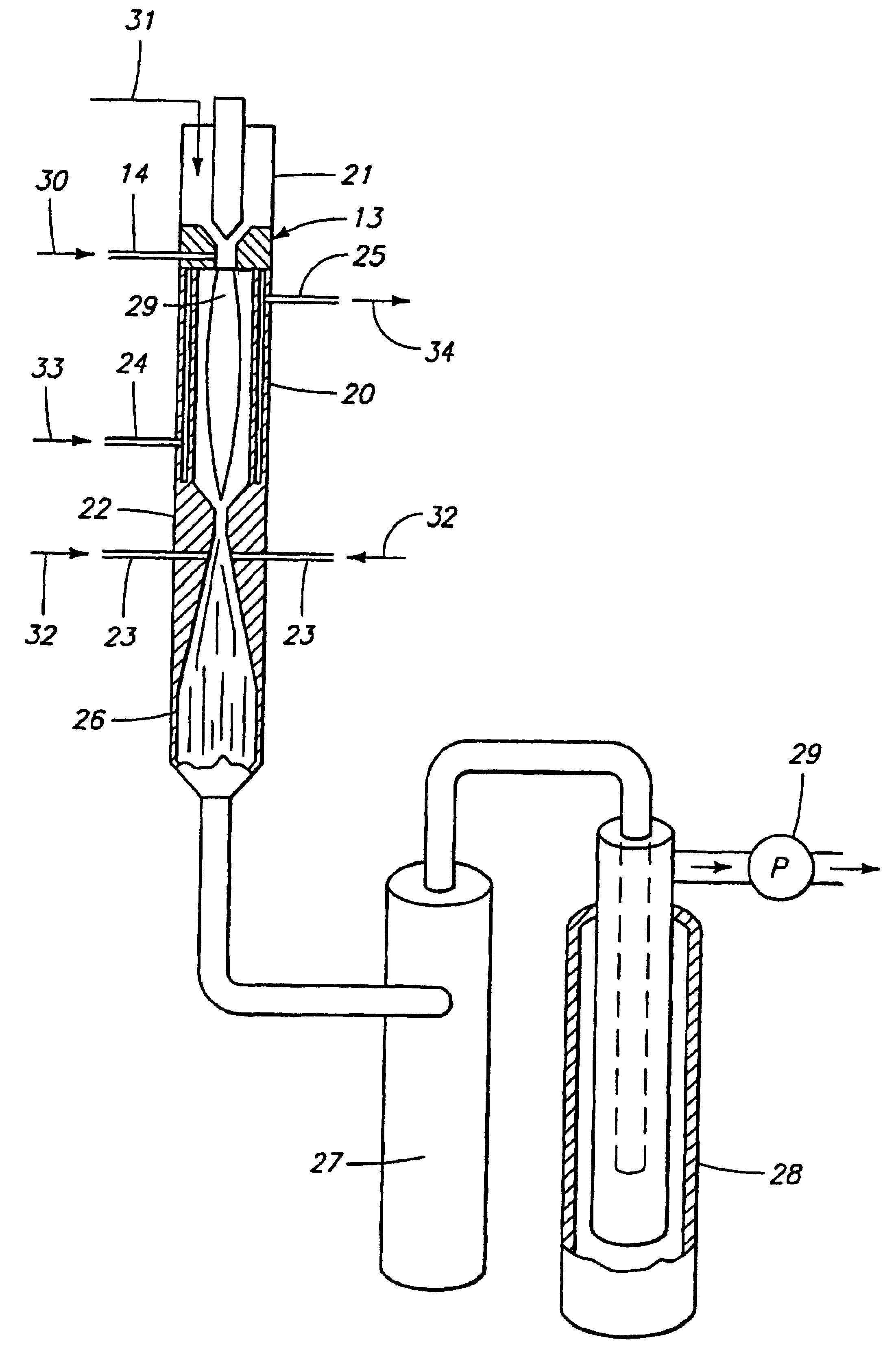
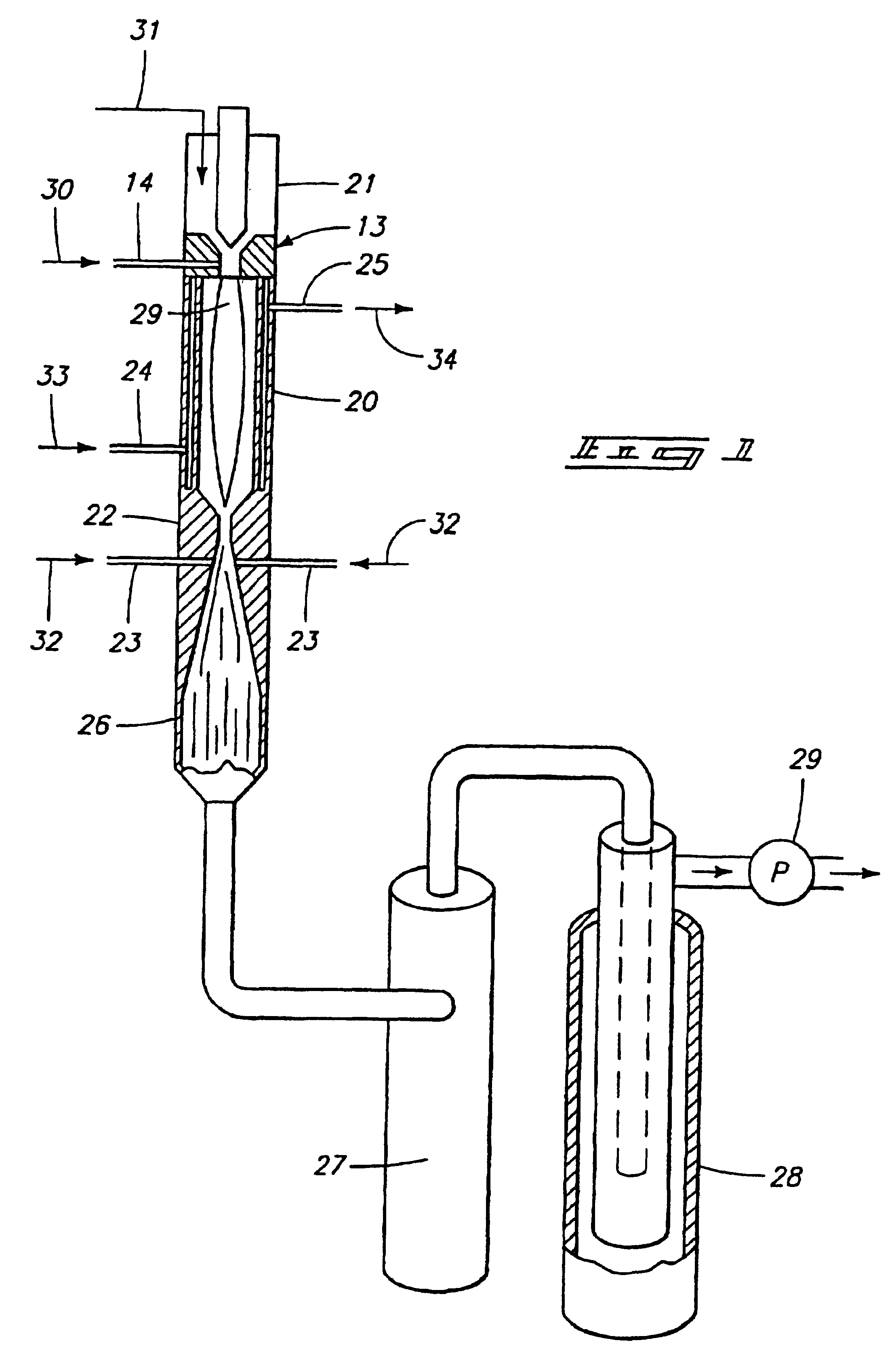
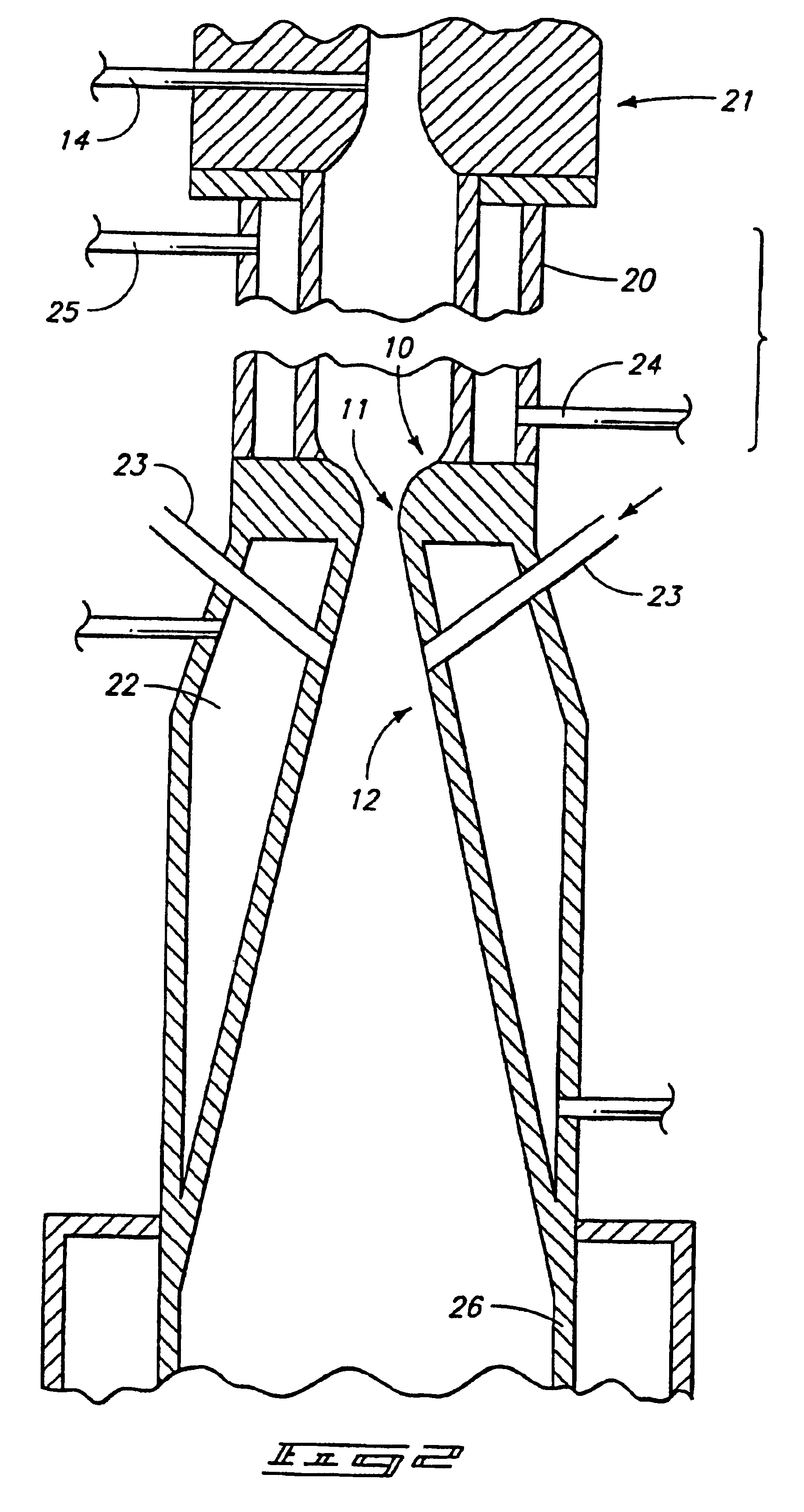
Fast quench reactor and method
InactiveUSRE37853E1Minimizing back reactionRapid coolingMaterial nanotechnologyIndirect heat exchangersThermodynamicsProcess engineering
A fast quench reaction includes a reactor chamber having a high temperature heating means such as a plasma torch at its inlet and a restrictive convergent-divergent nozzle at its outlet end. Reactants are injected into the reactor chamber. The resulting heated gaseous stream is then rapidly cooled by passage through the nozzle. This "freezes" the desired end product(s) in the heated equilibrium reaction stage.
Owner:BATTELLE ENERGY ALLIANCE LLC
Electro-mechanical interfaces to mount robotic surgical arms
In one embodiment of the invention, a method of mounting a surgical robotic arm to a set-up arm of a robotic surgical system is provided that includes sliding a pair of guide slots of the surgical robotic arm over a pair of guide tabs in the set-up arm; aligning electrical connectors in the set-up arm to electrical connectors of the surgical robotic arm; and coincidentally mating male electrical connectors to female electrical connectors while finally mating the guide tabs in the set-up arm to flanges of a housing of the surgical robotic arm.
Owner:INTUITIVE SURGICAL OPERATIONS INC
Compositions for Catalytic Gasification of a Petroleum Coke
InactiveUS20090090056A1Cost-effective and high-yielding productionPromote gasificationHydrogenGaseous fuelsParticle compositionMethane gas
The present invention relates to particulate compositions of a lower ash type petroleum coke containing at least two preselected components (alkali metal and calcium) that exhibit an efficient, enhanced-yielding gasification to value added gaseous products, particularly when used in a steady-state integrated gasification process. The compositions of the present invention are particularly useful for catalytic gasification of petroleum coke at moderate temperatures ranging from about 450° C. to about 900° C. Advantageously, the compositions can be readily incorporated into fluidized bed gasification units, and can result in a cost-effective, high-yielding production of methane gas from petroleum coke.
Owner:SURE CHAMPION INVESTMENT LTD
Method and apparatus for producing synthesis gas from carbonaceous materials
A method of producing syn gas from biomass or other carbonaceous material utilizes a controlled devolatilization reaction in which the temperature of the feed material is maintained at less than 450° F. until most available oxygen is consumed. This minimizes pyrolysis of the feed material. The method and apparatus utilizes the formed synthesis gas to provide the energy for the necessary gasification. This provides for a high purity syn gas and avoids production of slag.
Owner:JBK EXTRACTIONS
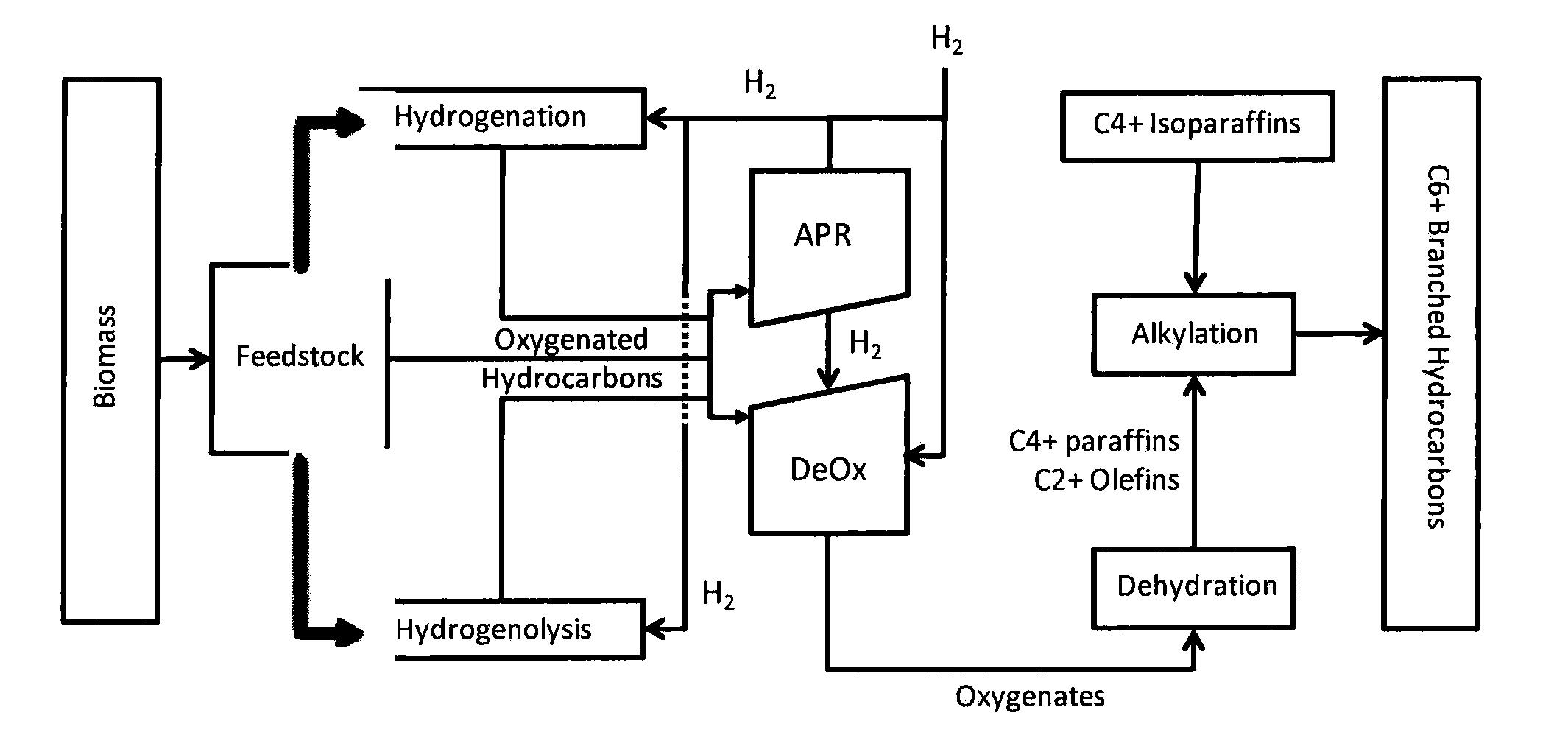
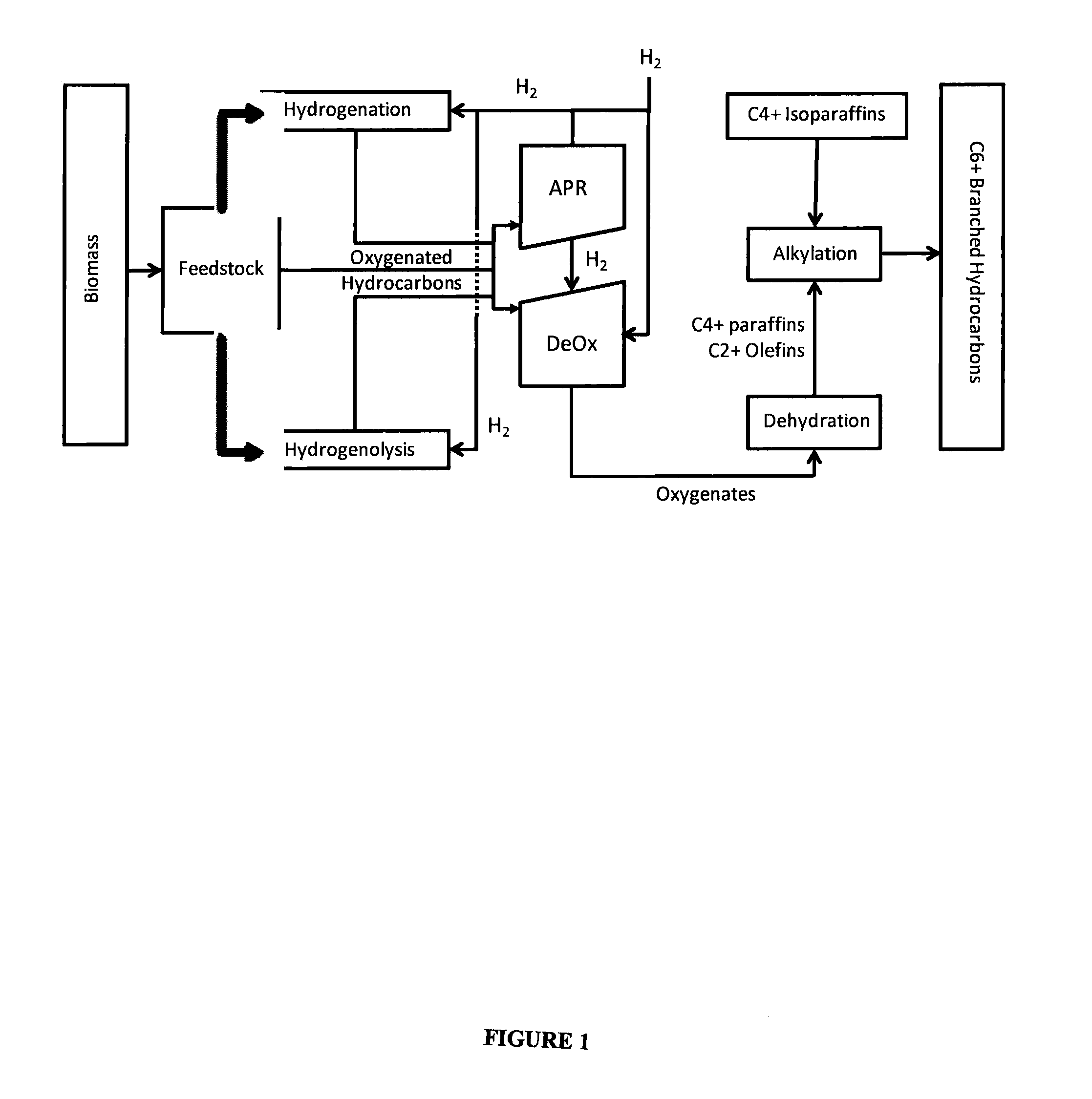
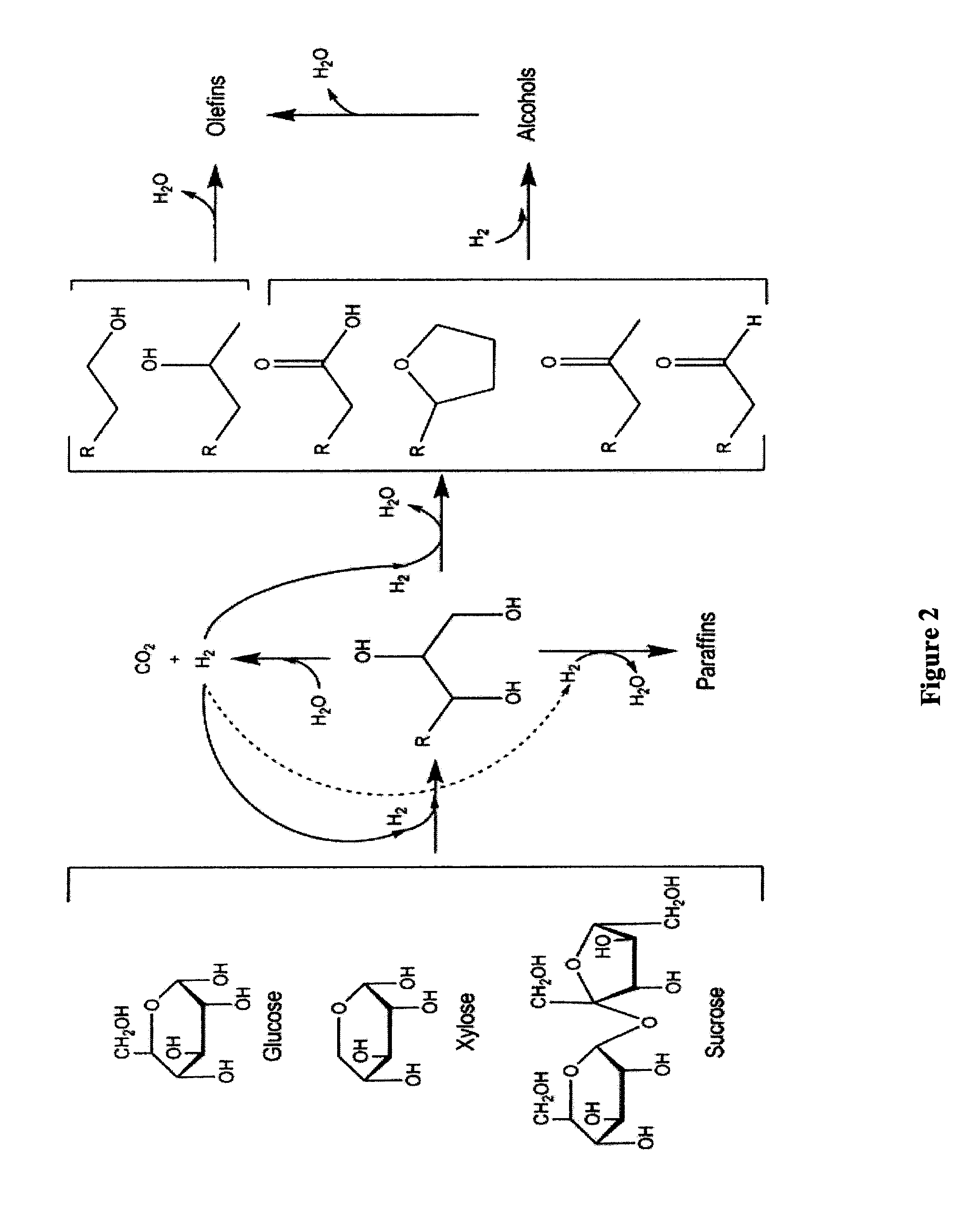
Synthesis of liquid fuels from biomass
ActiveUS20100076233A1Hydrocarbon by metathesis reactionLiquid hydrocarbon mixture productionFuranAlkane
Processes and reactor systems are provided for the conversion of oxygenated hydrocarbons to paraffins useful as liquid fuels. The process involves the conversion of water soluble oxygenated hydrocarbons to oxygenates, such as alcohols, furans, ketones, aldehydes, carboxylic acids, diols, triols, and / or other polyols, followed by the subsequent conversion of the oxygenates to paraffins by dehydration and alkylation. The oxygenated hydrocarbons may originate from any source, but are preferably derived from biomass.
Owner:VIRENT
Manifold designs, and flow control in multichannel microchannel devices
InactiveUS20050087767A1Equally distributedLow costChemical/physical/physico-chemical microreactorsSemiconductor/solid-state device manufacturingEngineering
Owner:VELOCYS CORPORATION
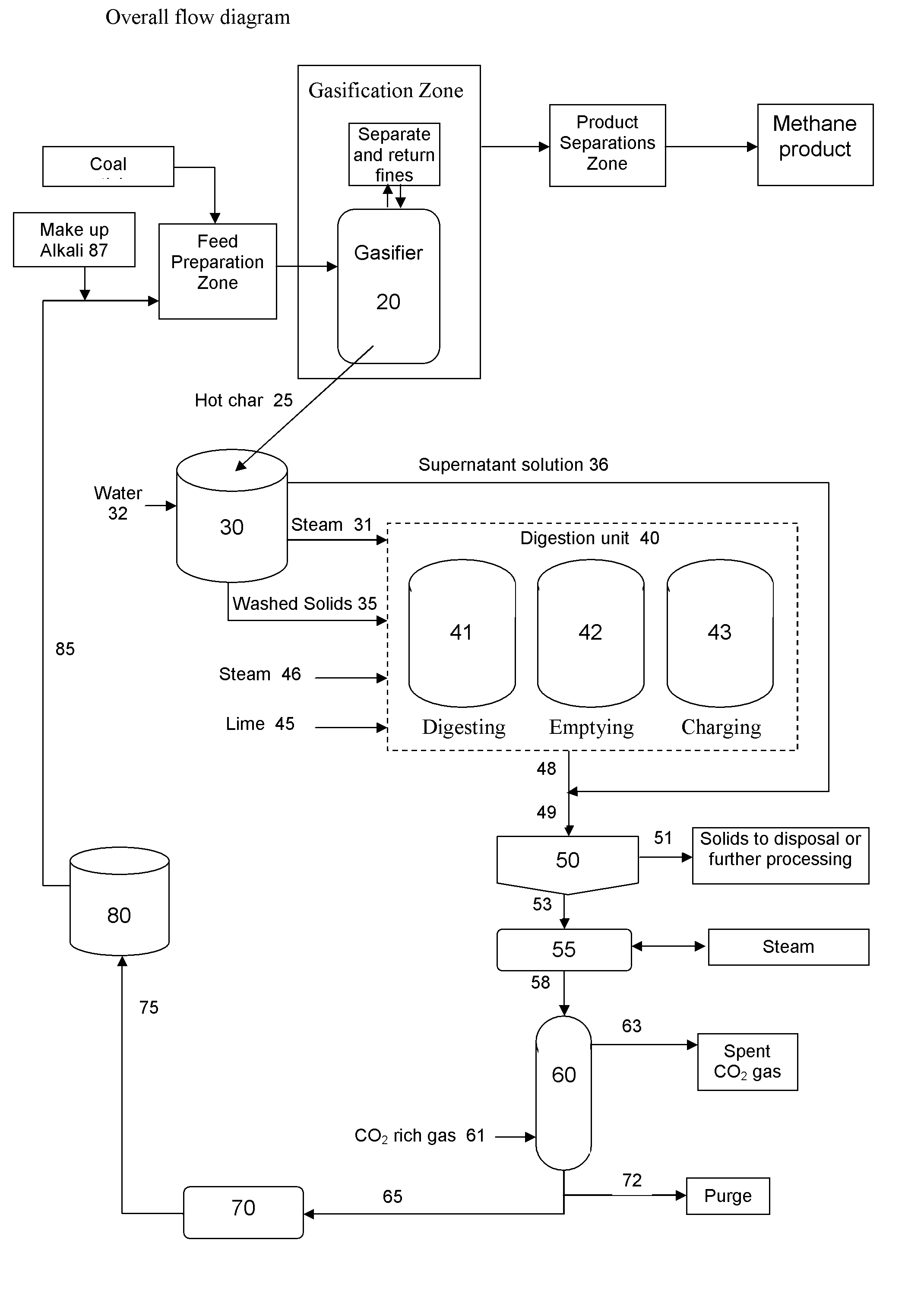
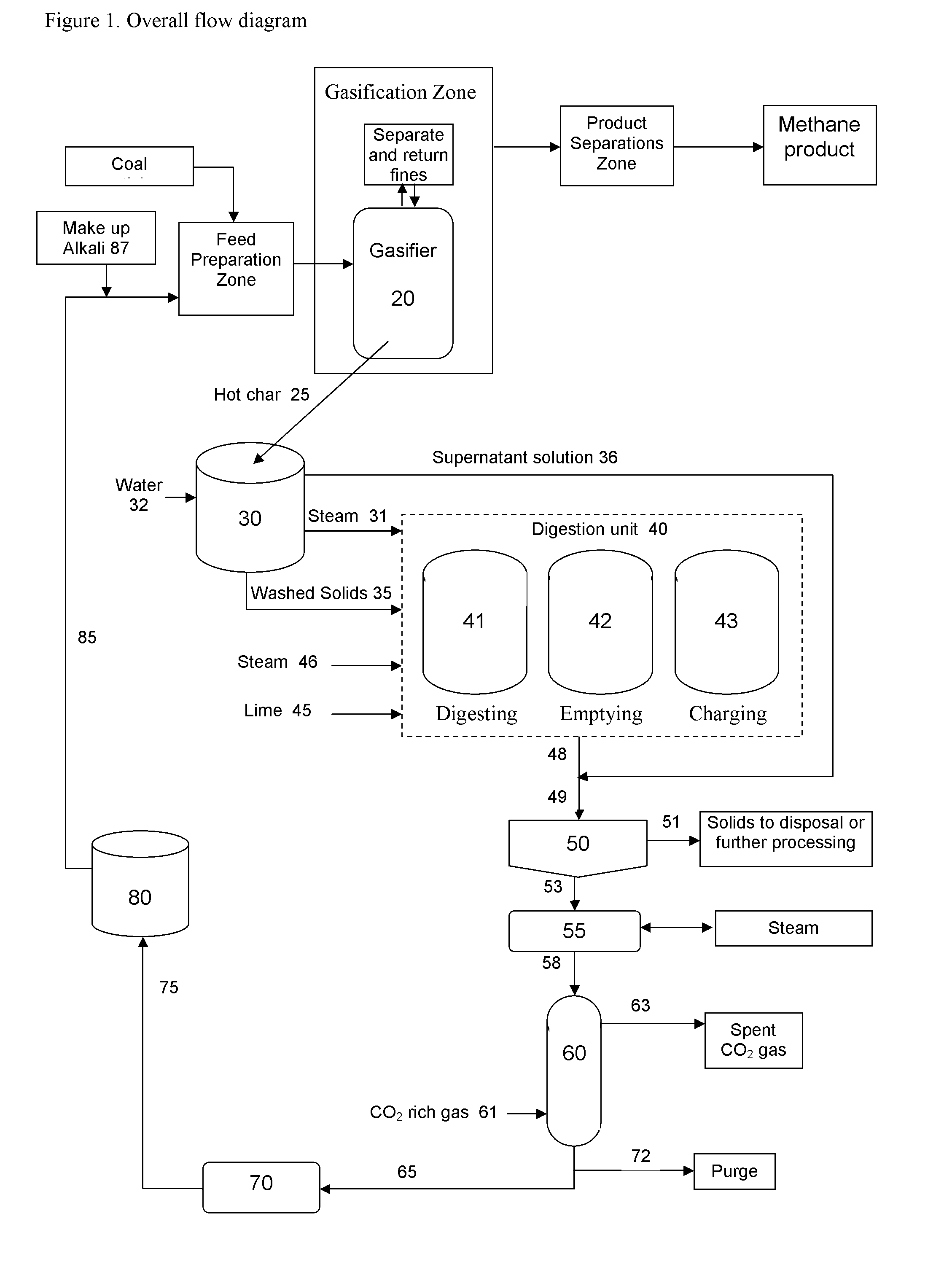
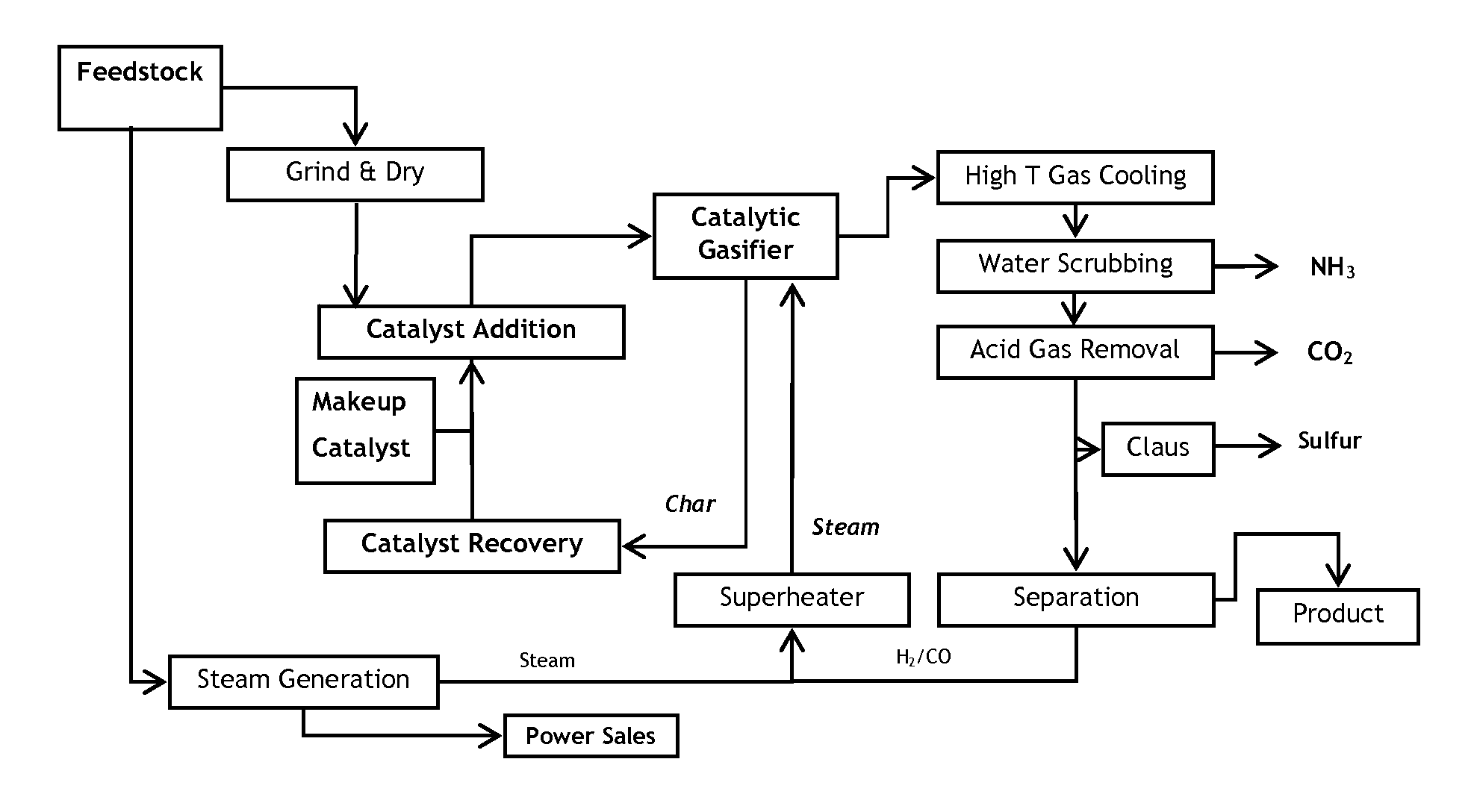
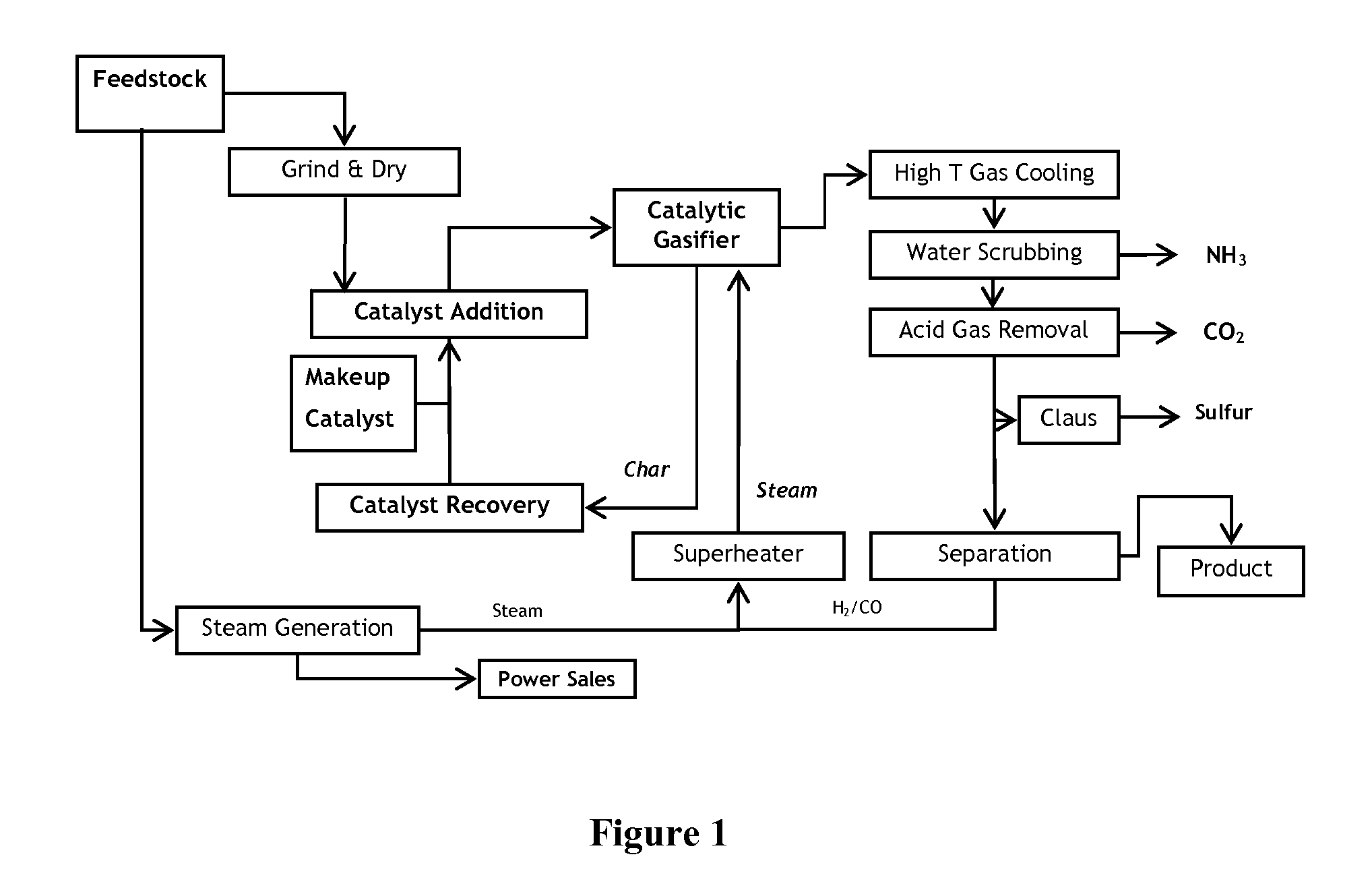
Catalytic steam gasification process with recovery and recycle of alkali metal compounds
ActiveUS20070277437A1Promote leachingLarge specific surface areaHydrogenGas modification by gas mixingCompound aAlkali metal
A high efficiency gasification process for converting carbonaceous solids to methane and apparatus for its practice are described. The process includes reacting steam and carbonaceous solids comprising ash in the presence of alkali metal catalyst in a gasification reactor to produce combustible gases and char particles comprising ash and alkali metal catalyst constituents, treating a stream of such char particles in an alkali metal catalyst recovery system to recover the catalyst constituents as alkali metal compounds, and recycling such recovered compounds. Within the alkali metal catalyst recovery system the process includes quenching the stream of char particles with water whereby such particles are cooled and fractured, dissolving soluble alkali metal catalyst constituents from the fractured solids to form a first alkali metal catalyst solution and washed solids, optionally reacting the washed solids in alkaline solution to form a second alkali metal catalyst solution, upgrading said first and optional second alkali solution to recover the alkali metal catalyst constituents as said alkali metal compounds.
Owner:SURE CHAMPION INVESTMENT LTD
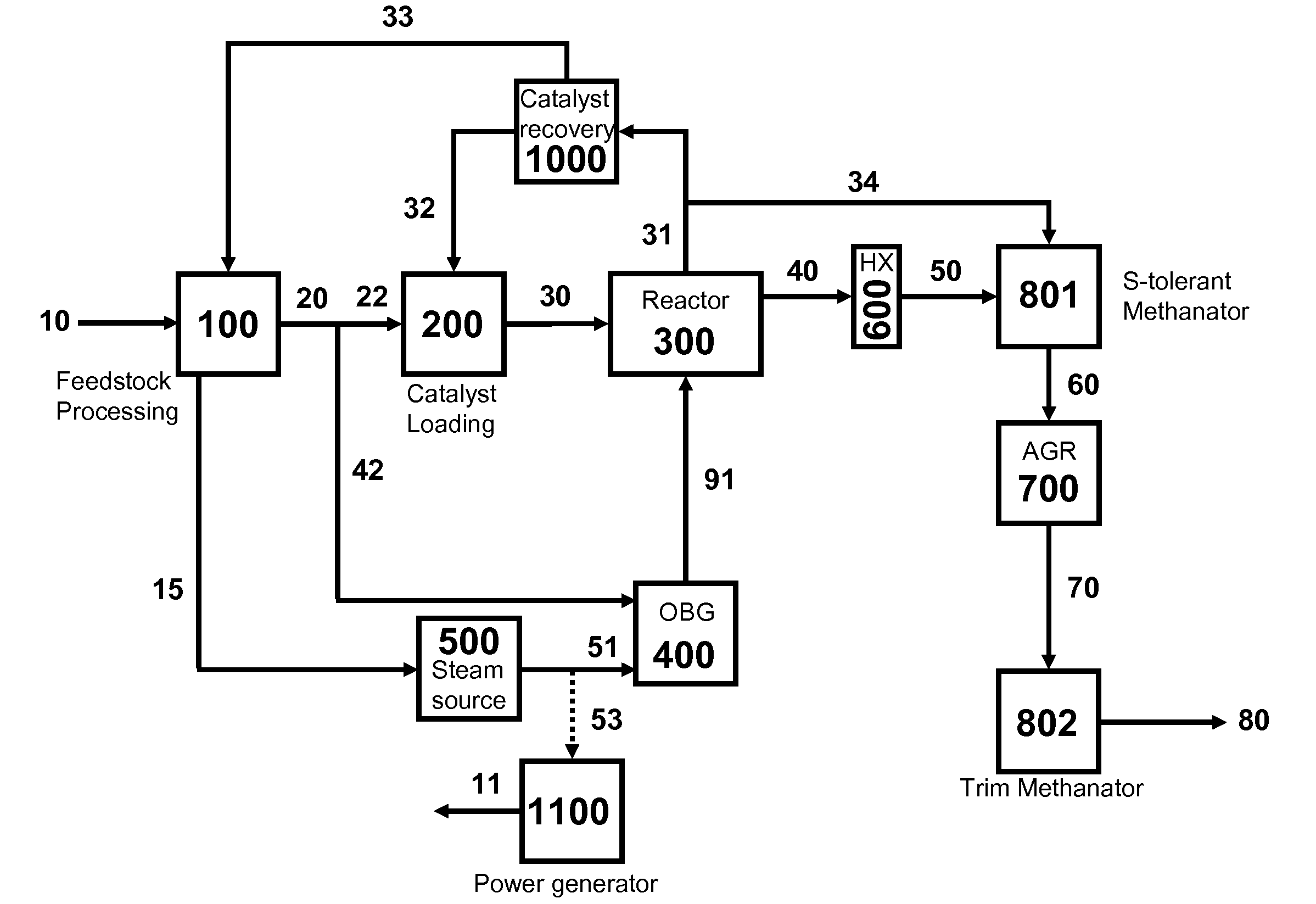
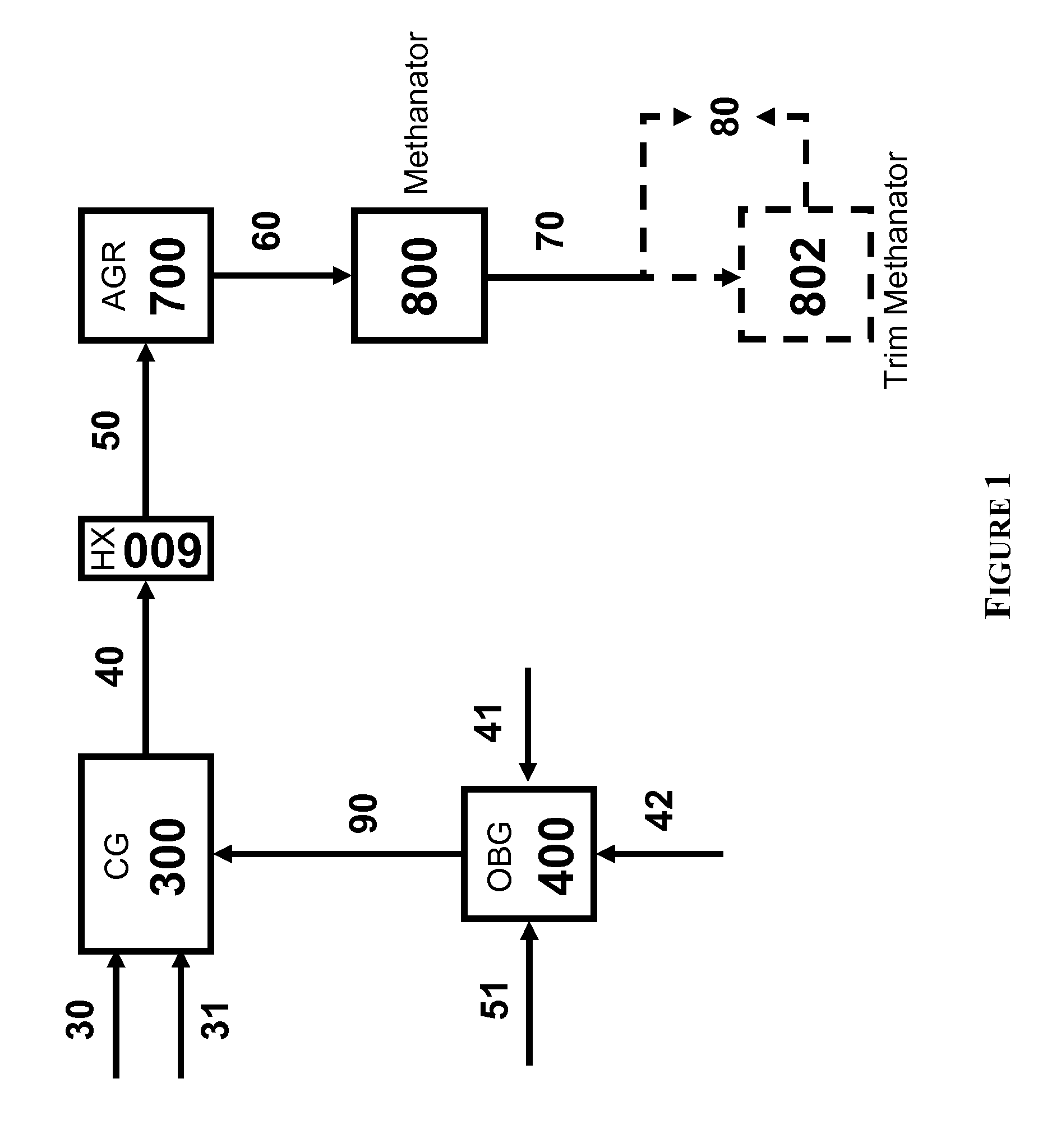
Processes for Gasification of a Carbonaceous Feedstock
The present invention relates to processes and continuous processes for preparing gaseous products, and in particular, methane via the catalytic gasification of carbonaceous feedstocks in the presence of steam. In one aspect of the invention, the processes comprise at least partially combusting a first carbonaceous feedstock with an oxygen-rich gas stream in an oxygen-blown gasifier, under suitable temperature and pressure, to generate a first gas stream comprising hydrogen, carbon monoxide and superheated steam; and reacting a second carbonaceous feedstock and the first gas stream in a catalytic gasifier in the presence of a gasification catalyst under suitable temperature and pressure to form a second gas stream comprising a plurality of gaseous products comprising methane, carbon dioxide, hydrogen, carbon monoxide and hydrogen sulfide. The processes can comprise using at least one catalytic methanator to convert carbon monoxide and hydrogen in the gaseous products to methane and in certain embodiments do not recycle carbon monoxide or hydrogen to the gasifier.
Owner:SURE CHAMPION INVESTMENT LTD
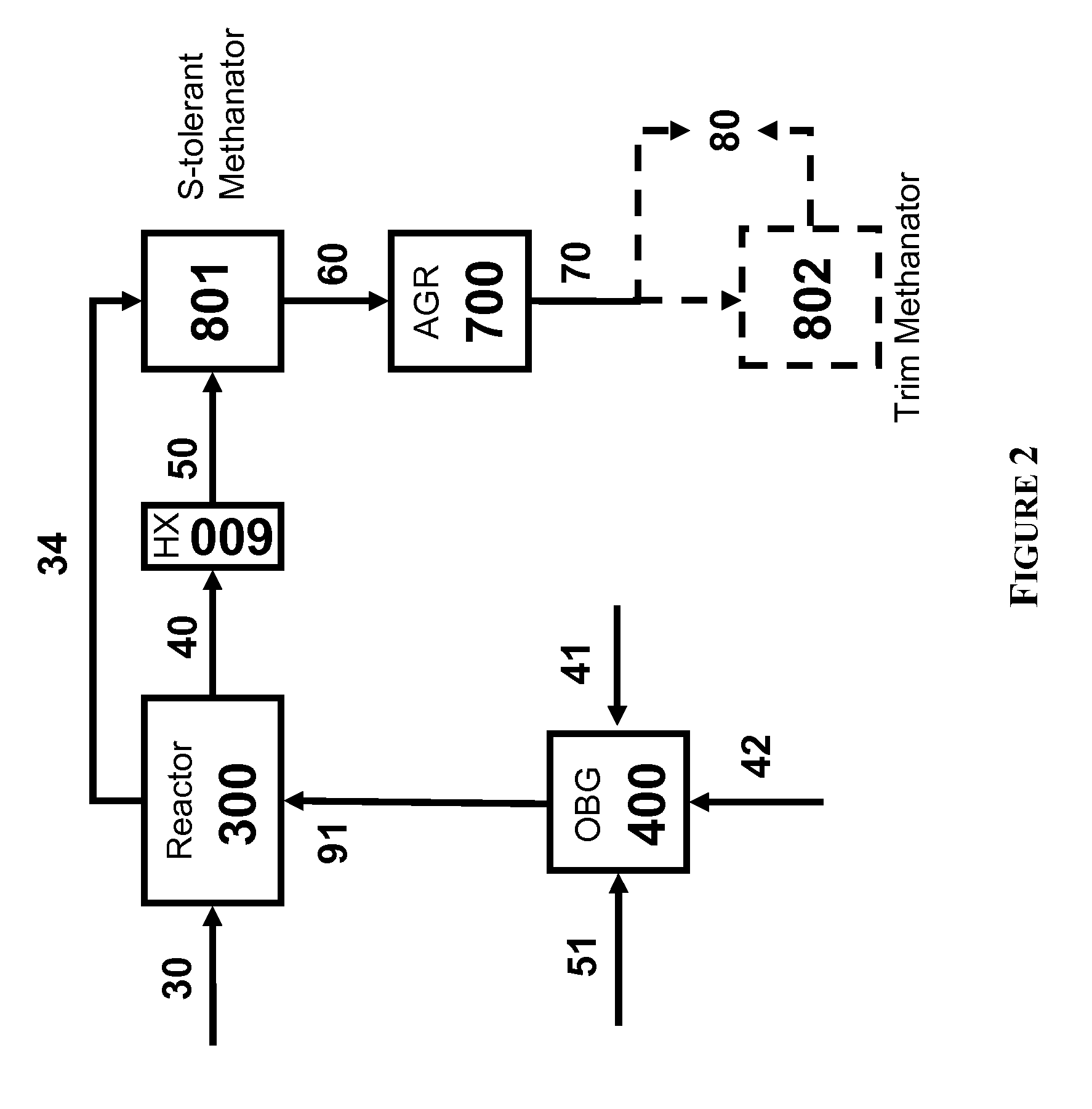
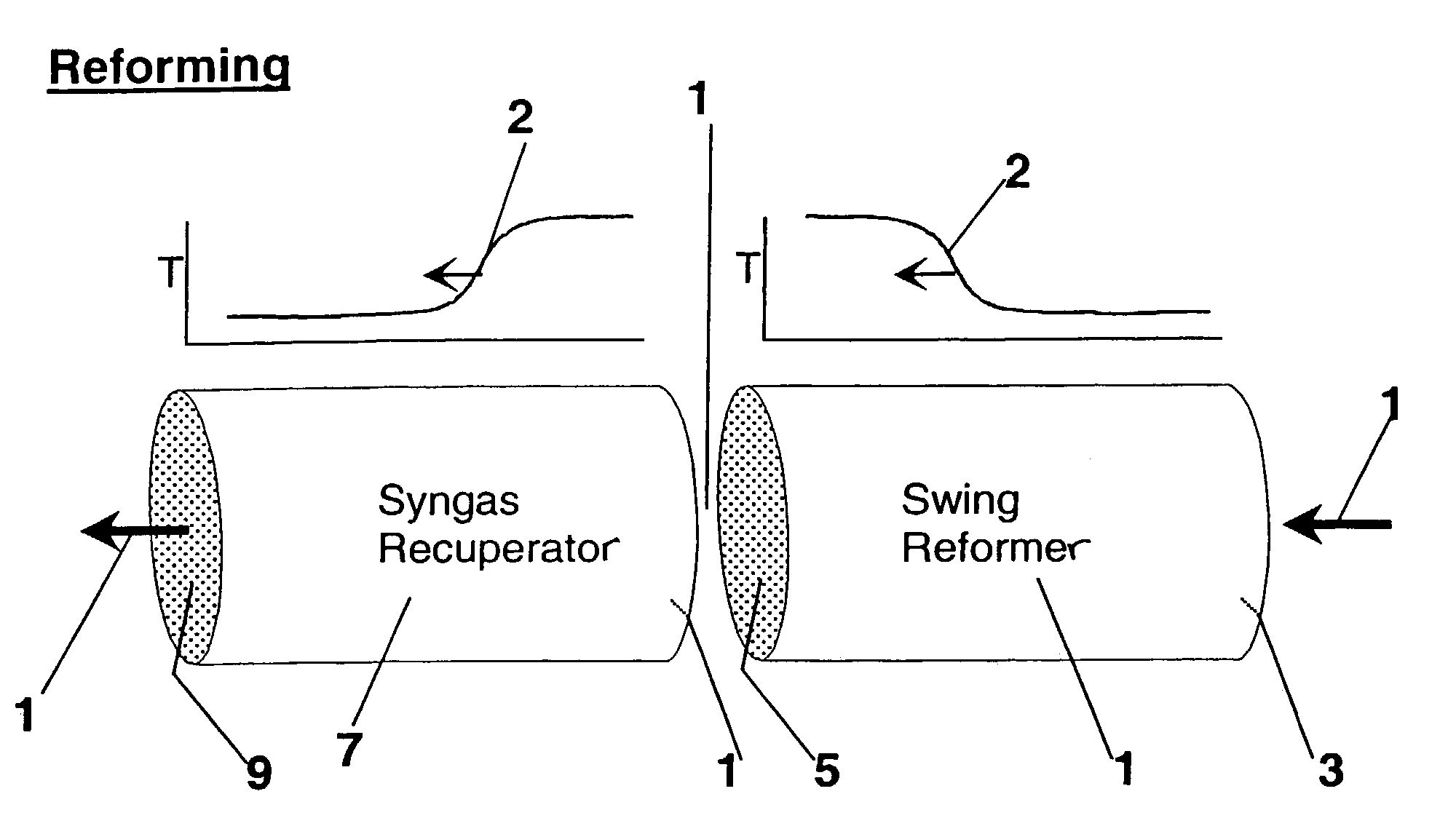
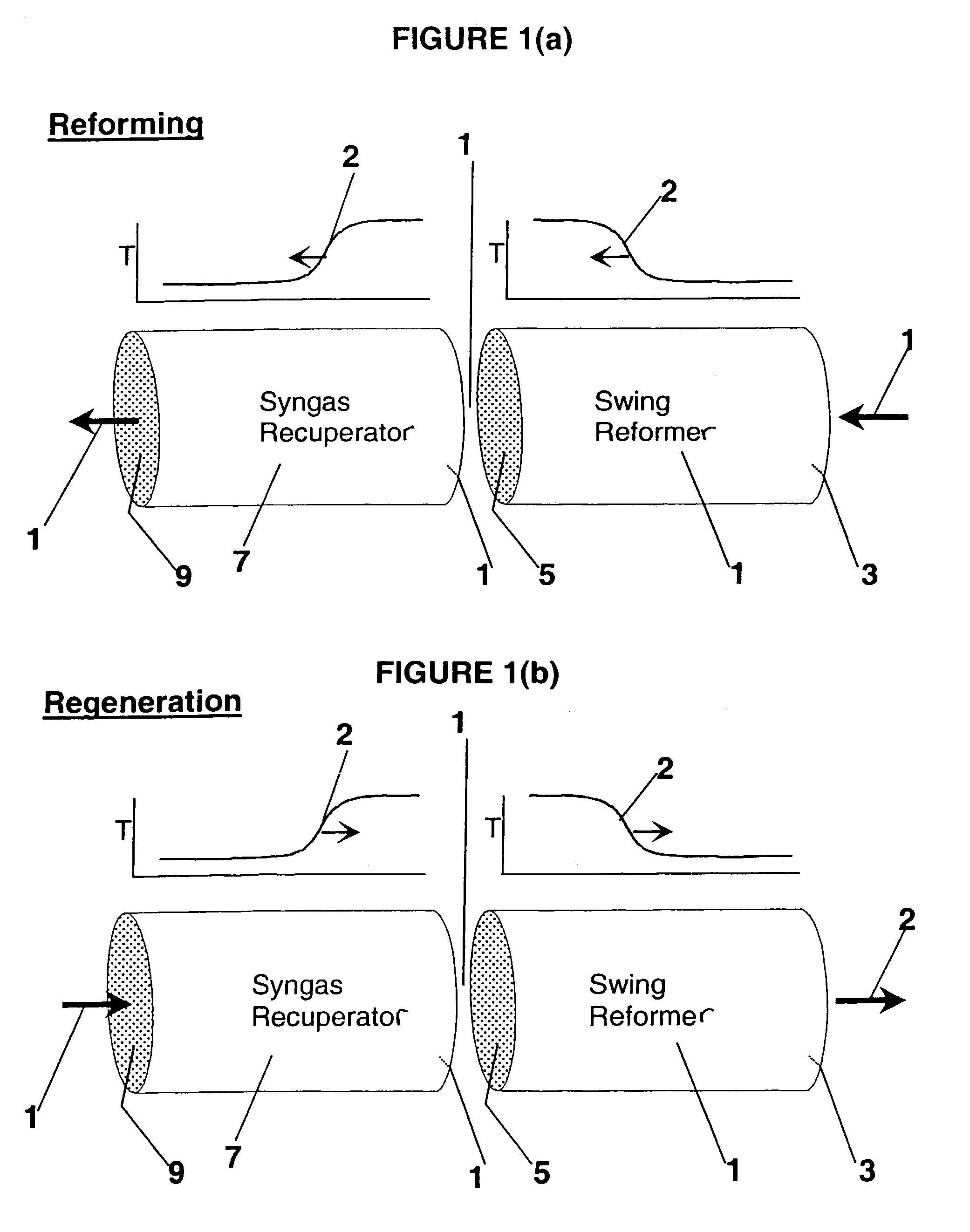
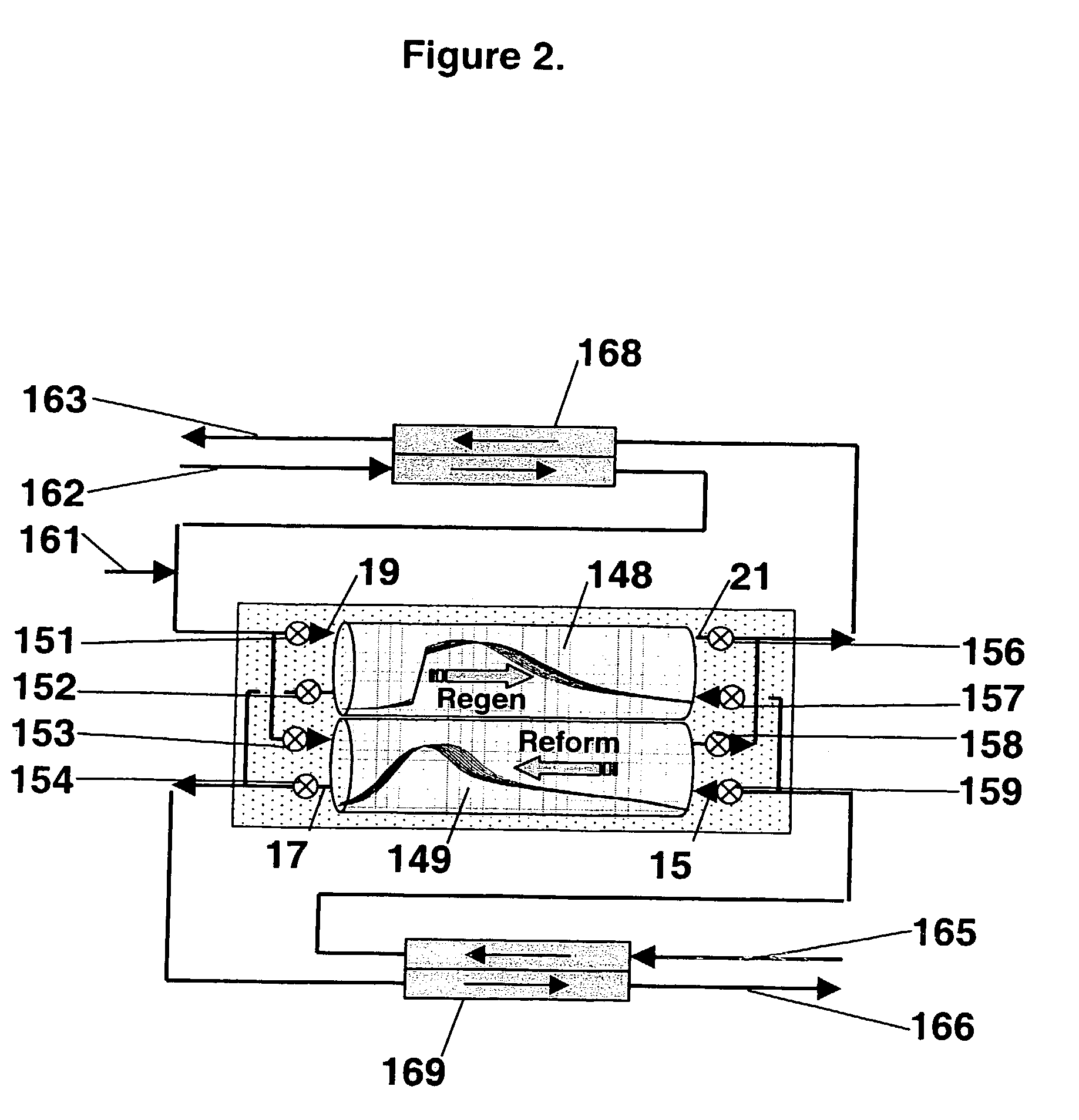
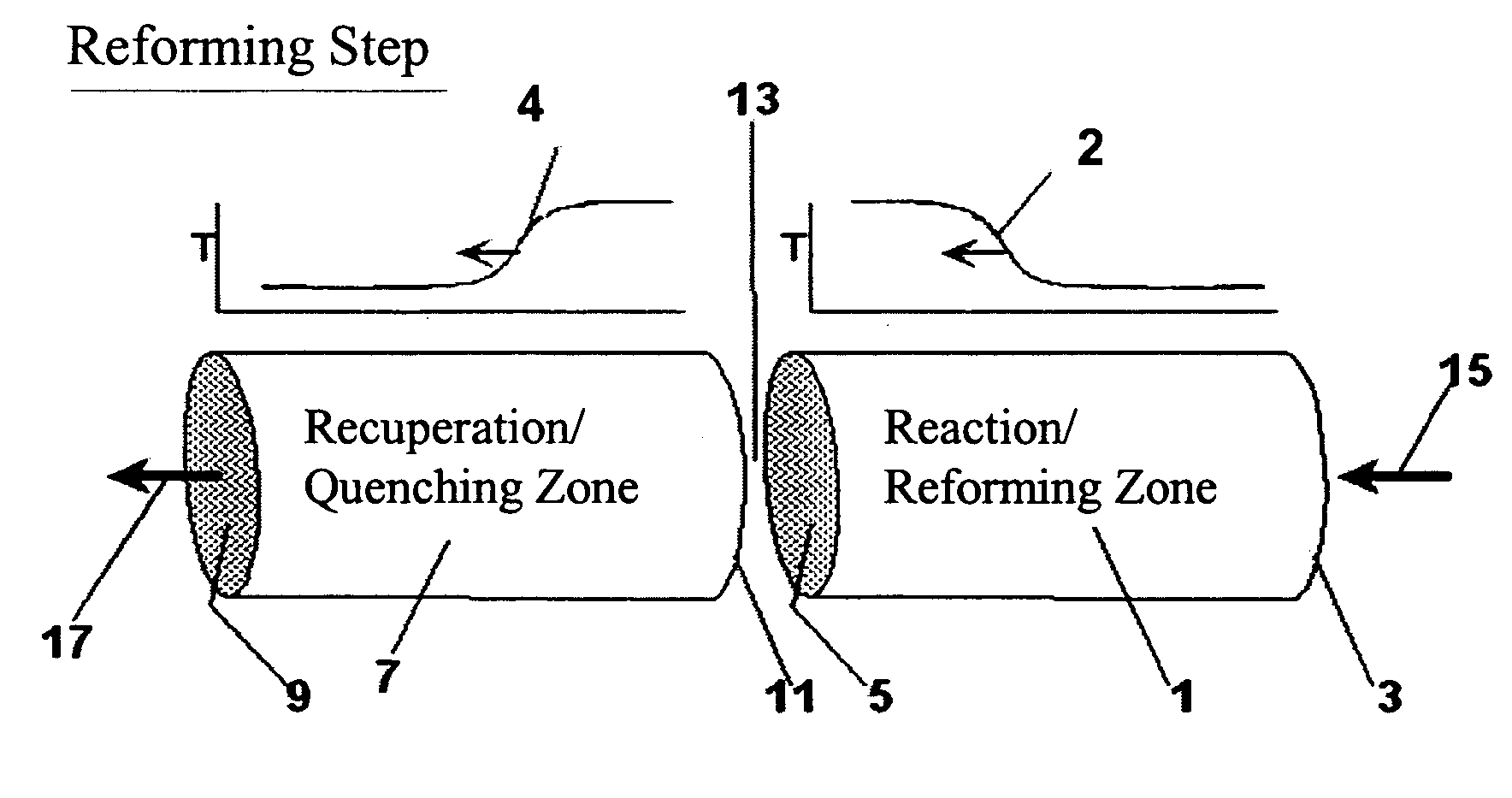
Pressure swing reforming
Synthesis gas is produced though a cyclic method where the first step of the cycle includes reforming a hydrocarbon feed over a catalyst to synthesis gas in a first zone of a bed and the second step reheats this first zone. A hydrocarbon feed is introduced to a bed along with CO2 and optionally steam where it is reformed into synthesis gas. The synthesis gas is collected at a second zone of the bed and an oxygen-containing gas is then introduced to this second zone of the bed and combusted with a fuel, thereby reheating the first zone to sufficient reforming temperatures. Additionally, a non-combusting gas can also be introduced to the second zone to move heat from the second zone to the first zone.
Owner:EXXON RES & ENG CO
Hybrid catalyst system for exhaust emissions reduction
InactiveUS20060010857A1Improve efficiencySpeed up the conversion processHydrogenGas treatmentExhaust fumesEngineering
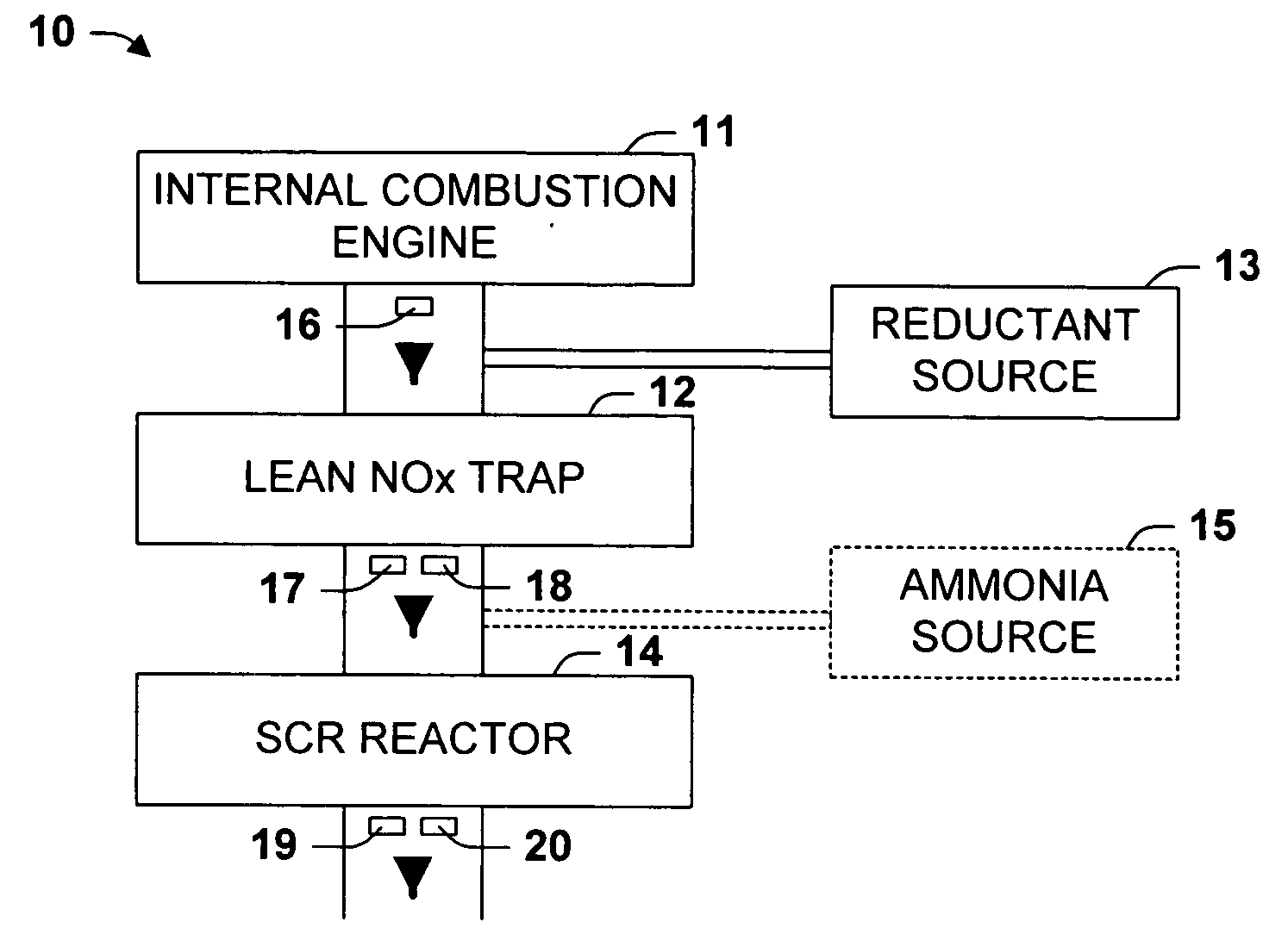
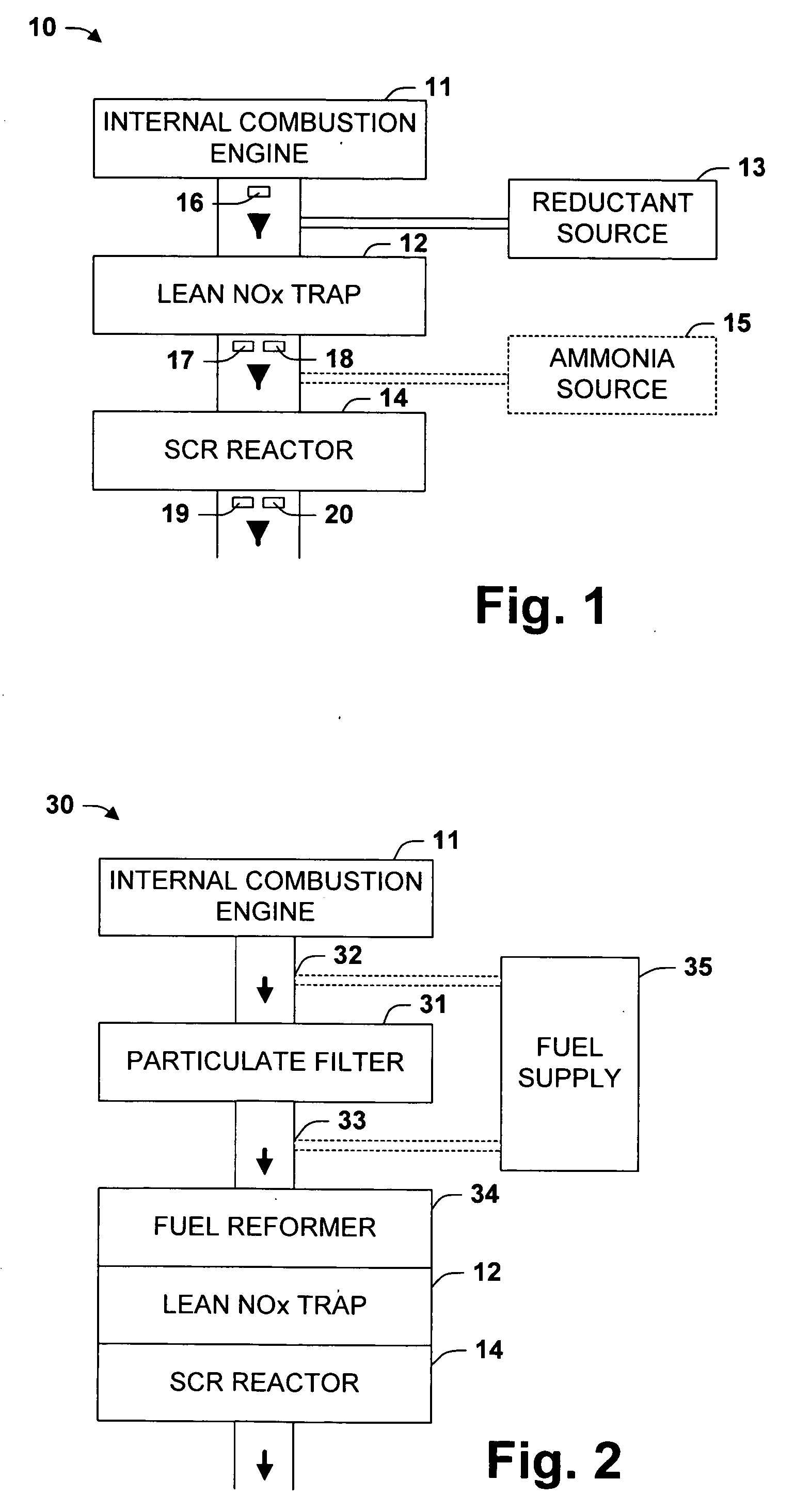
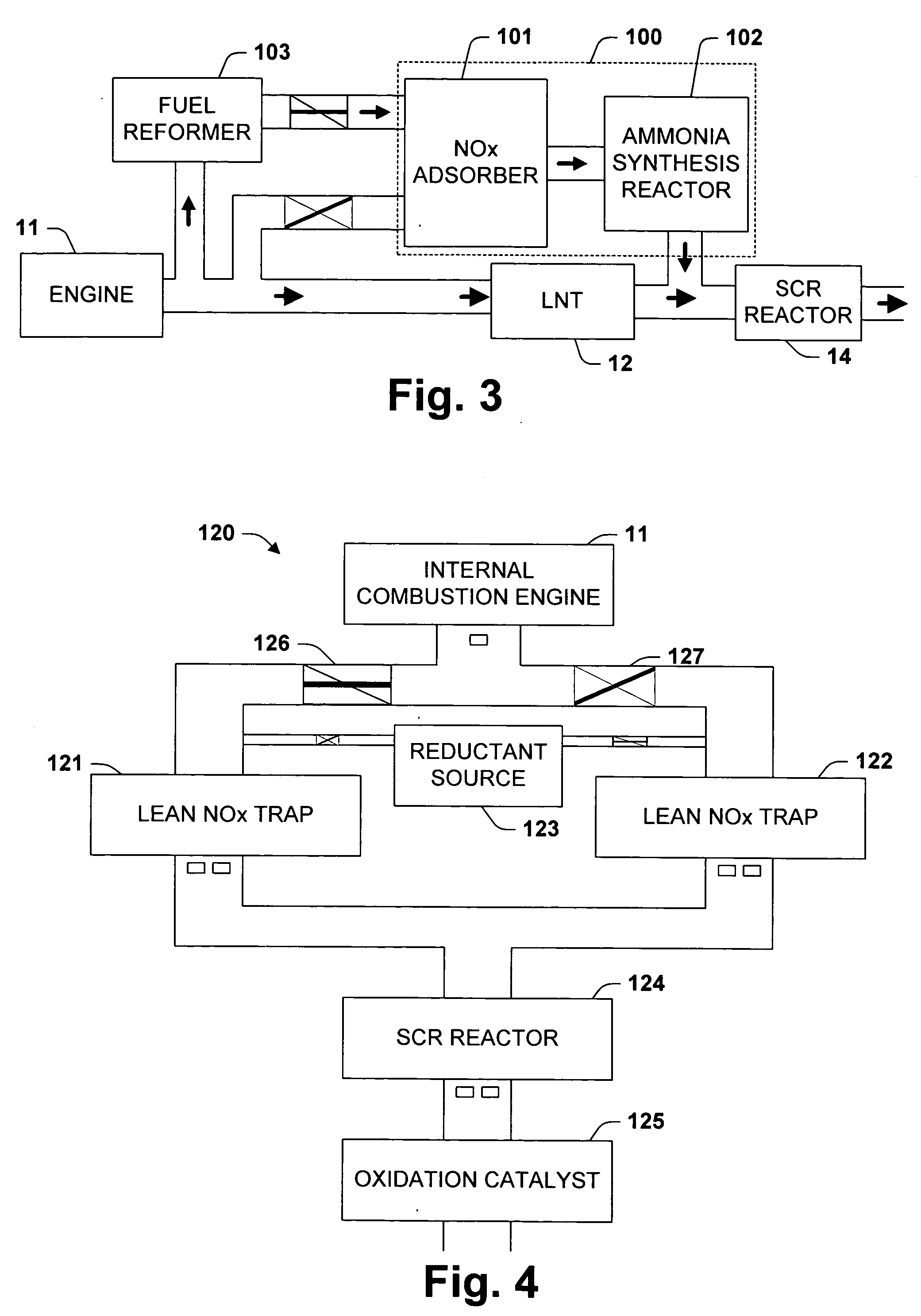
One aspect of the invention relates an exhaust treatment system having an SCR reactor following a NOx adsorber. Syn gas is used to regenerate the NOx adsorber. Another aspect relates to an LNT / SCR provided with an ammonia source separate from the LNT. A further aspect relates to a system comprising first and second LNTs and one or more SCRs downstream of the LNTs. A still further aspect relates to a device comprising first and second NOx adsorbers contained in a single housing. Another aspect relates to coating a surface of a moving part in an exhaust system with an oxidation catalyst to mitigate fouling. Additional aspects of the invention relate to strategies for controlling one or more of the time to initiate a regeneration cycle, the time to terminate a regeneration cycle, and the reductant injection rate during regeneration of LNT / SCR exhaust treatment systems.
Owner:INT ENGINE INTPROP CO LLC
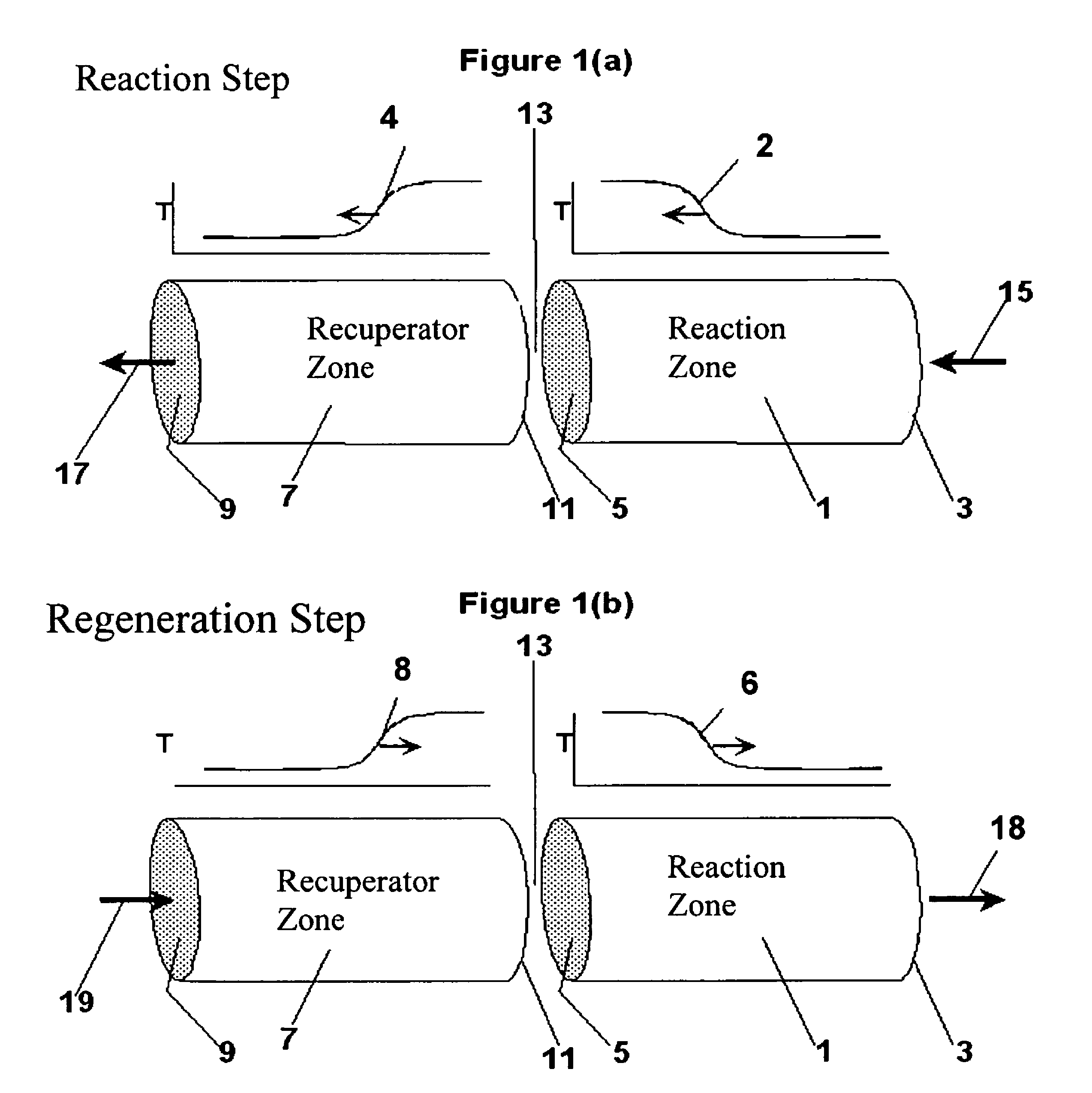
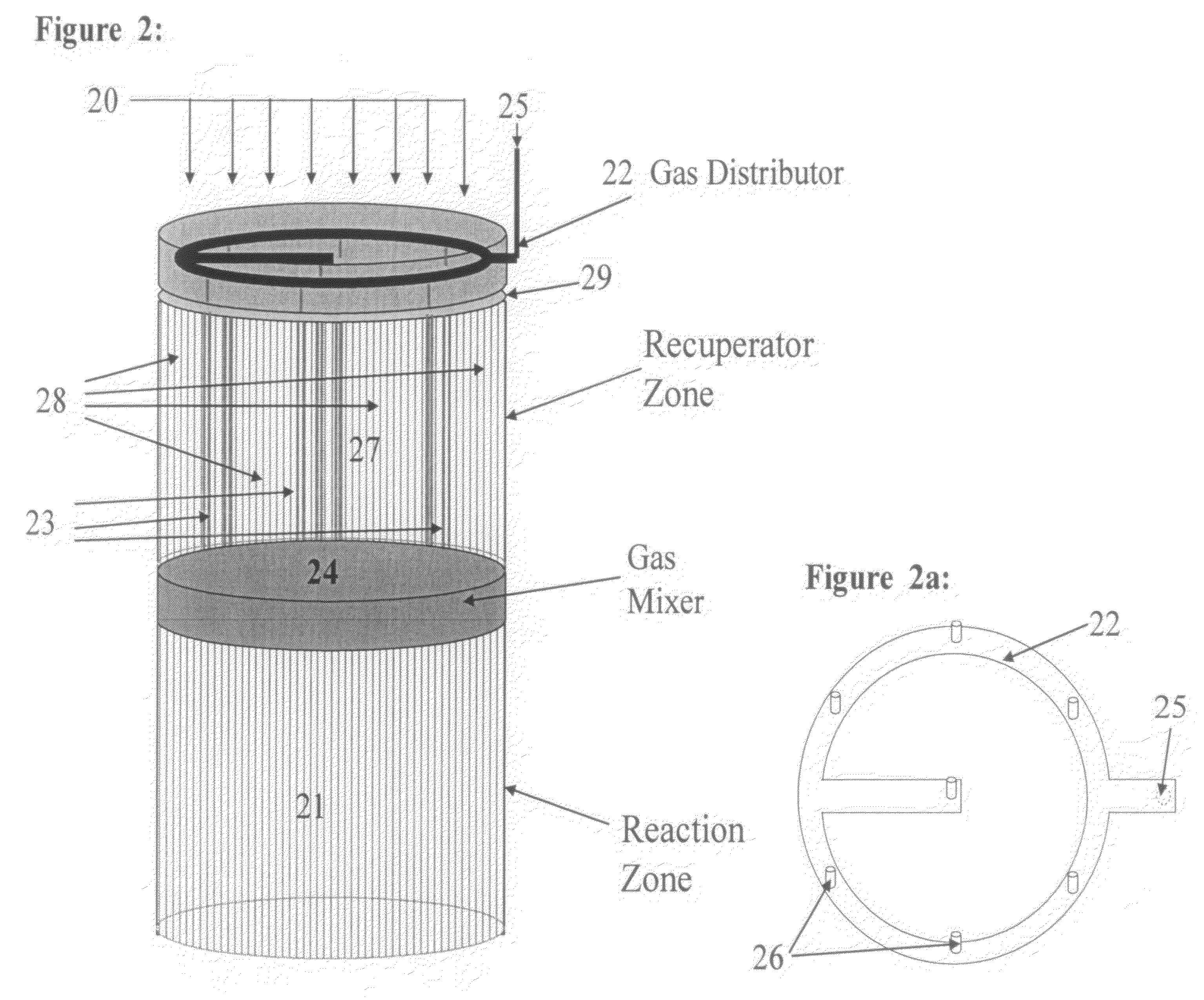
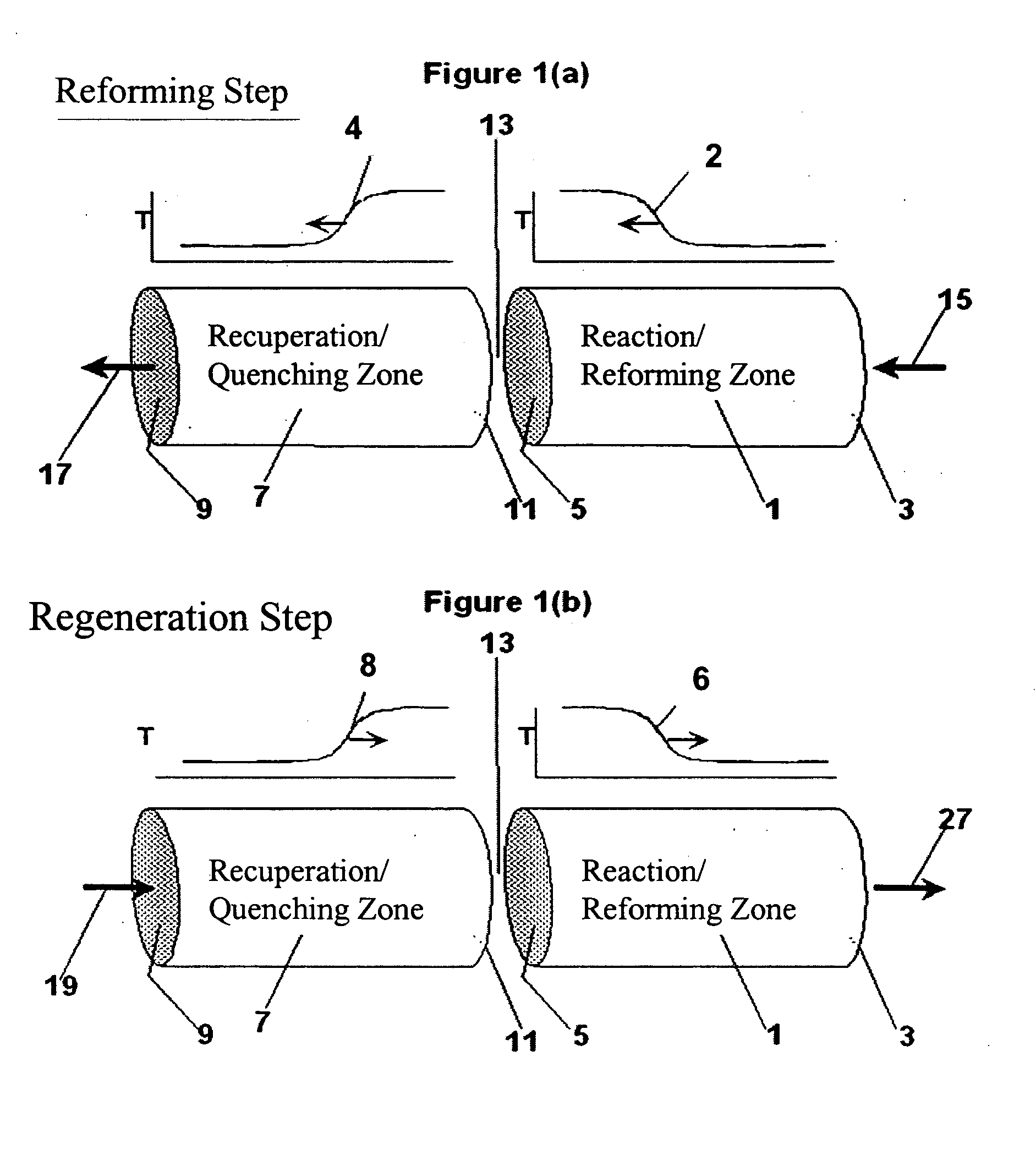
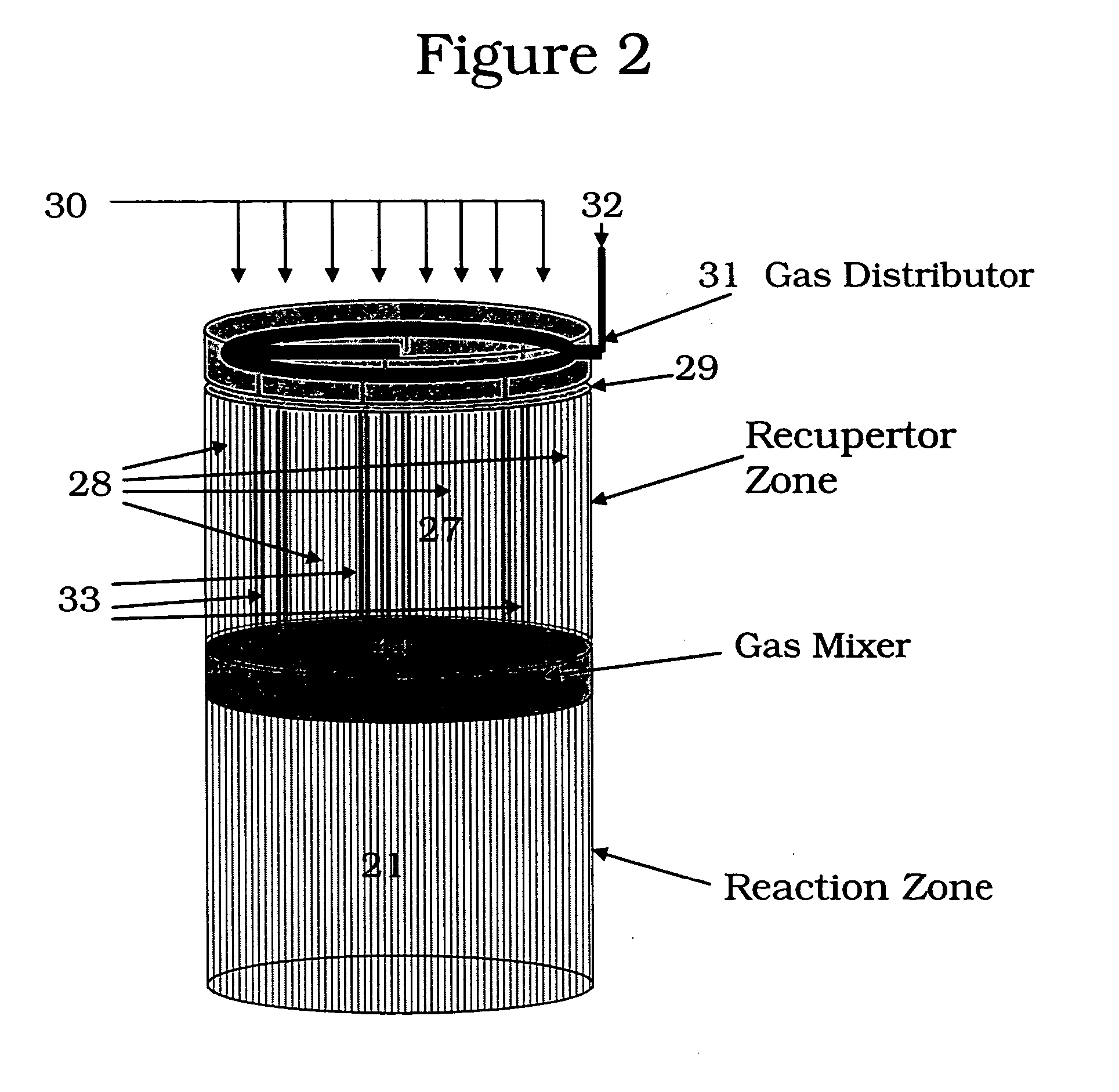
Controlled combustion for regenerative reactors with mixer/flow distributor
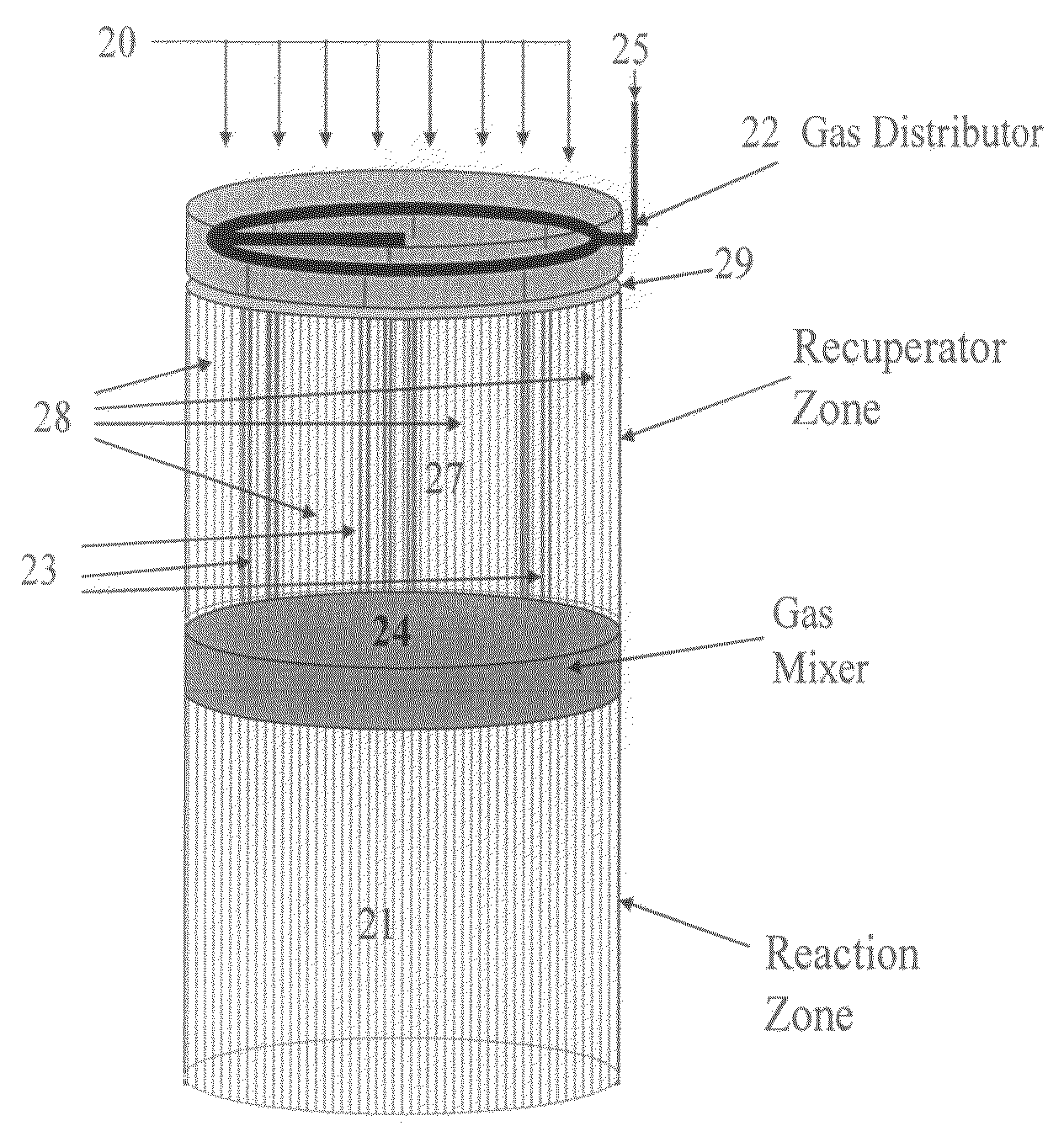
ActiveUS7815873B2Efficient transferEvenly distributedHydrocarbon by dehydrogenationFlow mixersReactor systemCombustion
The overall efficiency of a regenerative bed reverse flow reactor system is increased where the location of the exothermic reaction used for regeneration is suitably controlled. The present invention provides a method and apparatus for controlling the combustion to improve the thermal efficiency of bed regeneration in a cyclic reaction / regeneration processes. The process for thermal regeneration of a regenerative reactor bed entails(a) supplying the first reactant through a first channel means in a first regenerative bed and supplying at least a second reactant through a second channel means in the first regenerative bed,(b) combining said first and second reactants by a gas mixing means situated at an exit of the first regenerative bed and reacting the combined gas to produce a heated reaction product,(c) passing the heated reaction product through a second regenerative bed thereby transferring heat from the reaction product to the second regenerative bed.
Owner:EXXON RES & ENG CO
Methane conversion to higher hydrocarbons
ActiveUS20070191664A1Efficient preparationValid conversionGenerators with non-automatic water feedFlow mixersReactor systemReagent
The present invention provides a process for the manufacture of acetylene and other higher hydrocarbons from methane feed using a reverse-flow reactor system, wherein the reactor system includes (i) a first reactor and (ii) a second reactor, the first and second reactors oriented in a series relationship with respect to each other, the process comprising supplying each of first and second reactant through separate channels in the first reactor bed of a reverse-flow reactor such that both of the first and second reactants serve to quench the first reactor bed, without the first and second reactants substantially reacting with each other until reaching the core of the reactor system.
Owner:EXXONMOBIL CHEM PAT INC
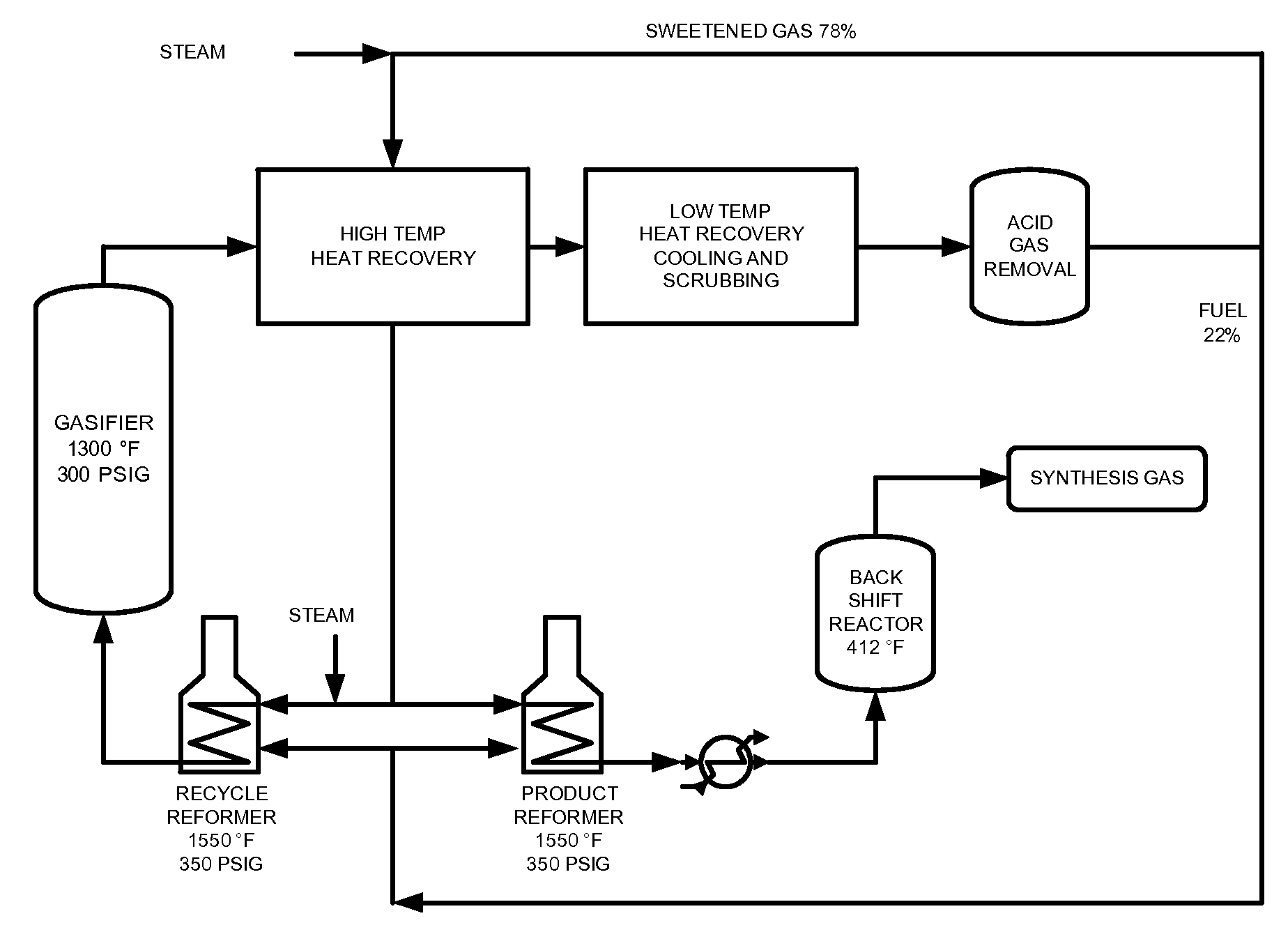
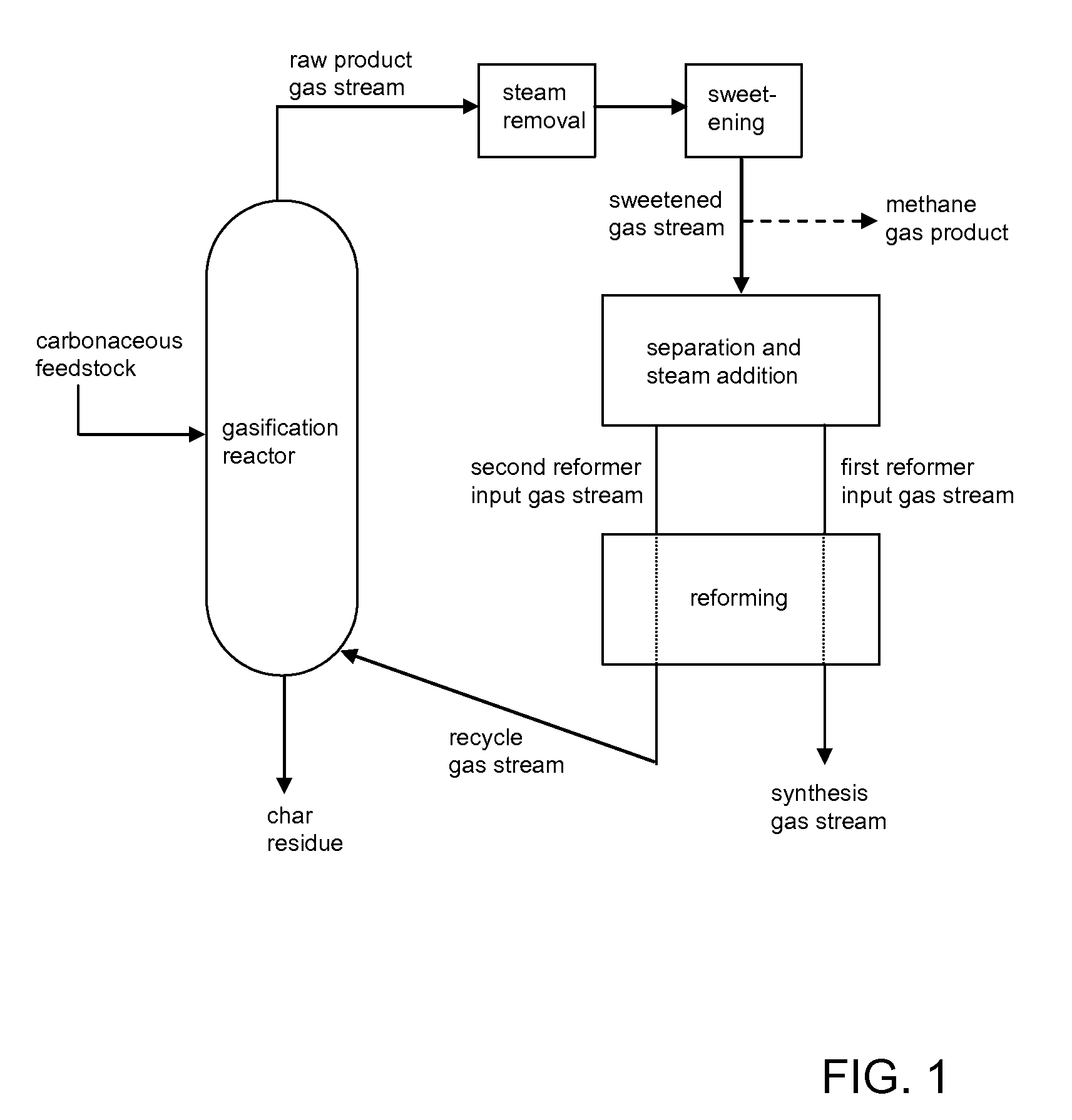
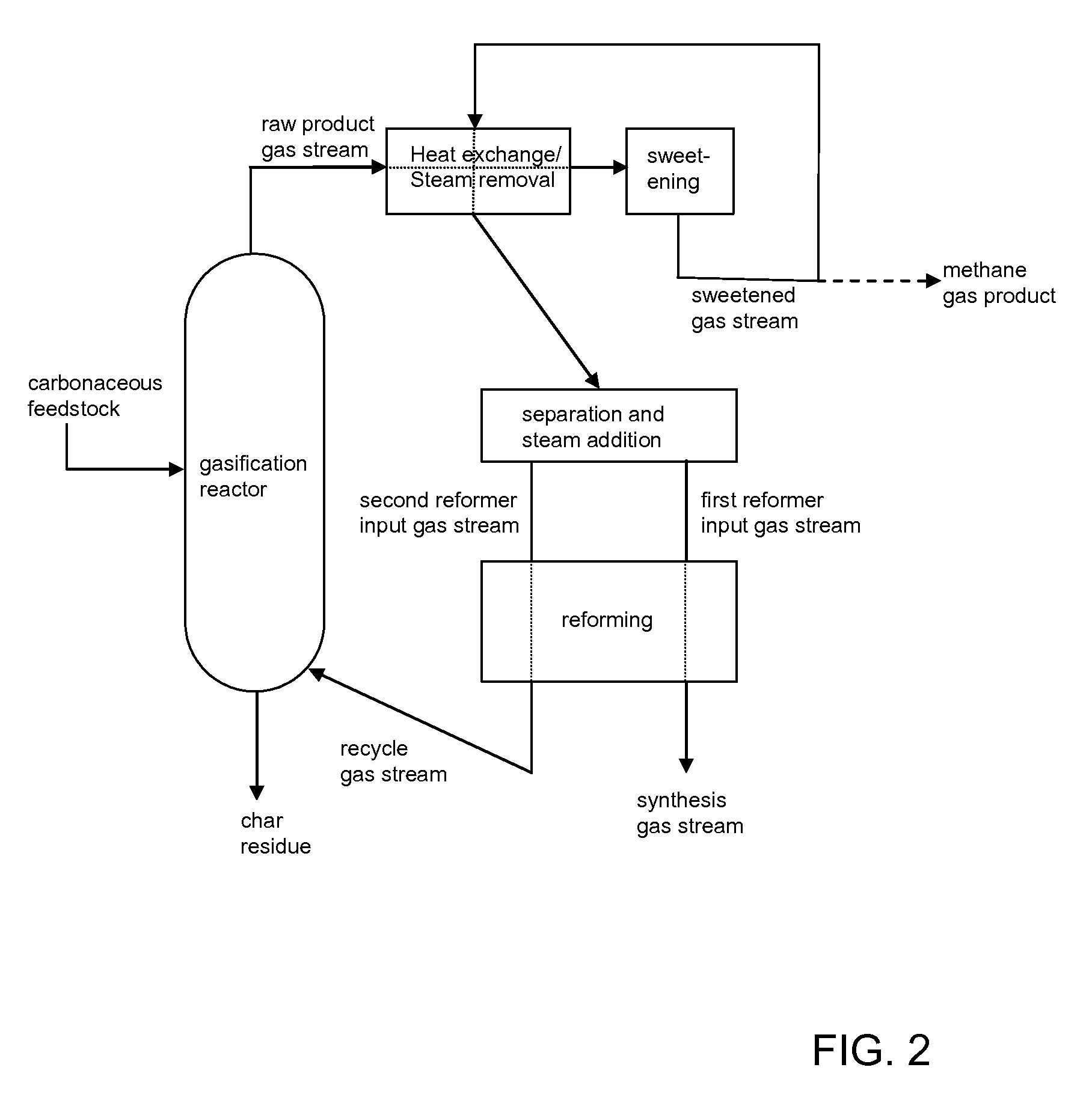
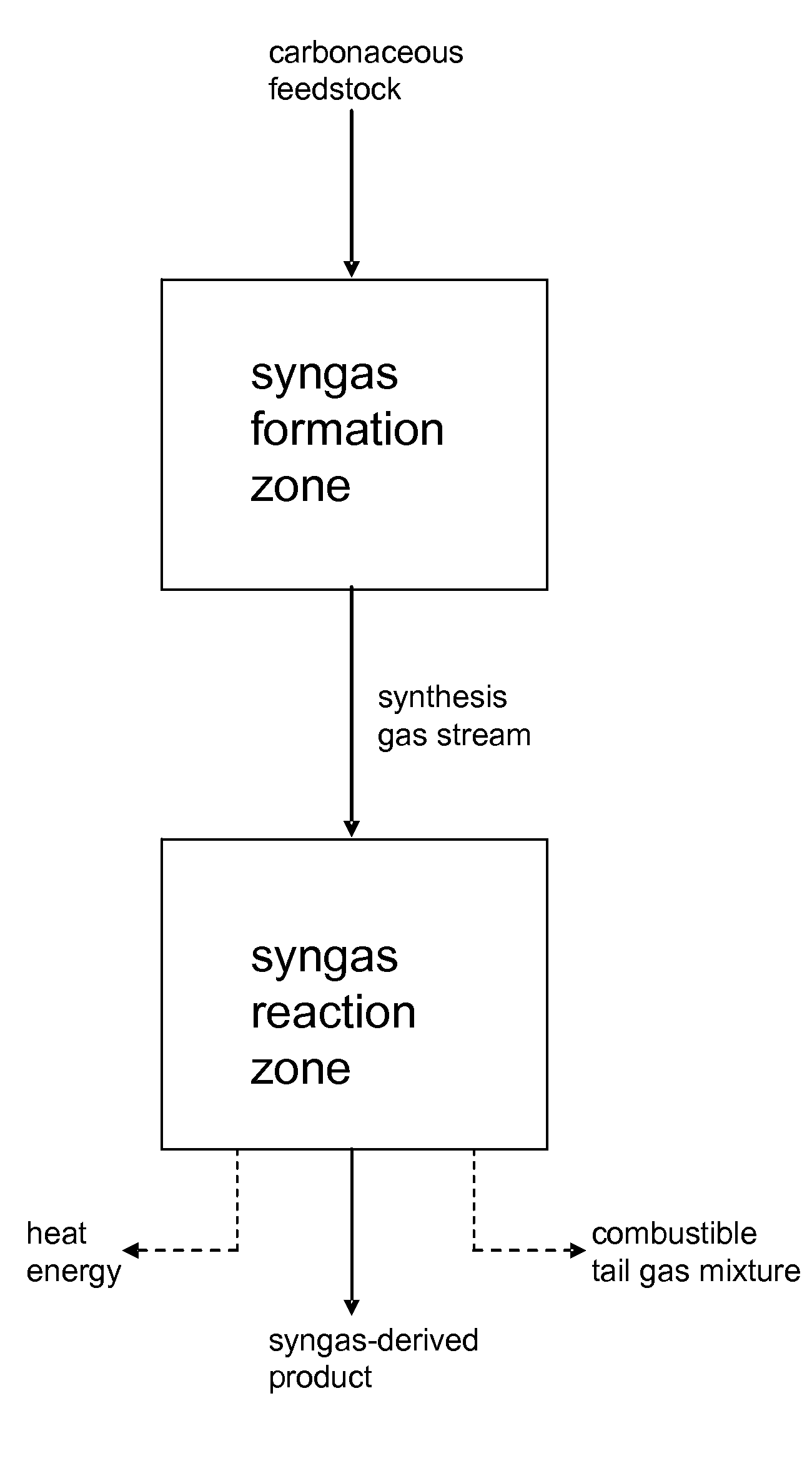
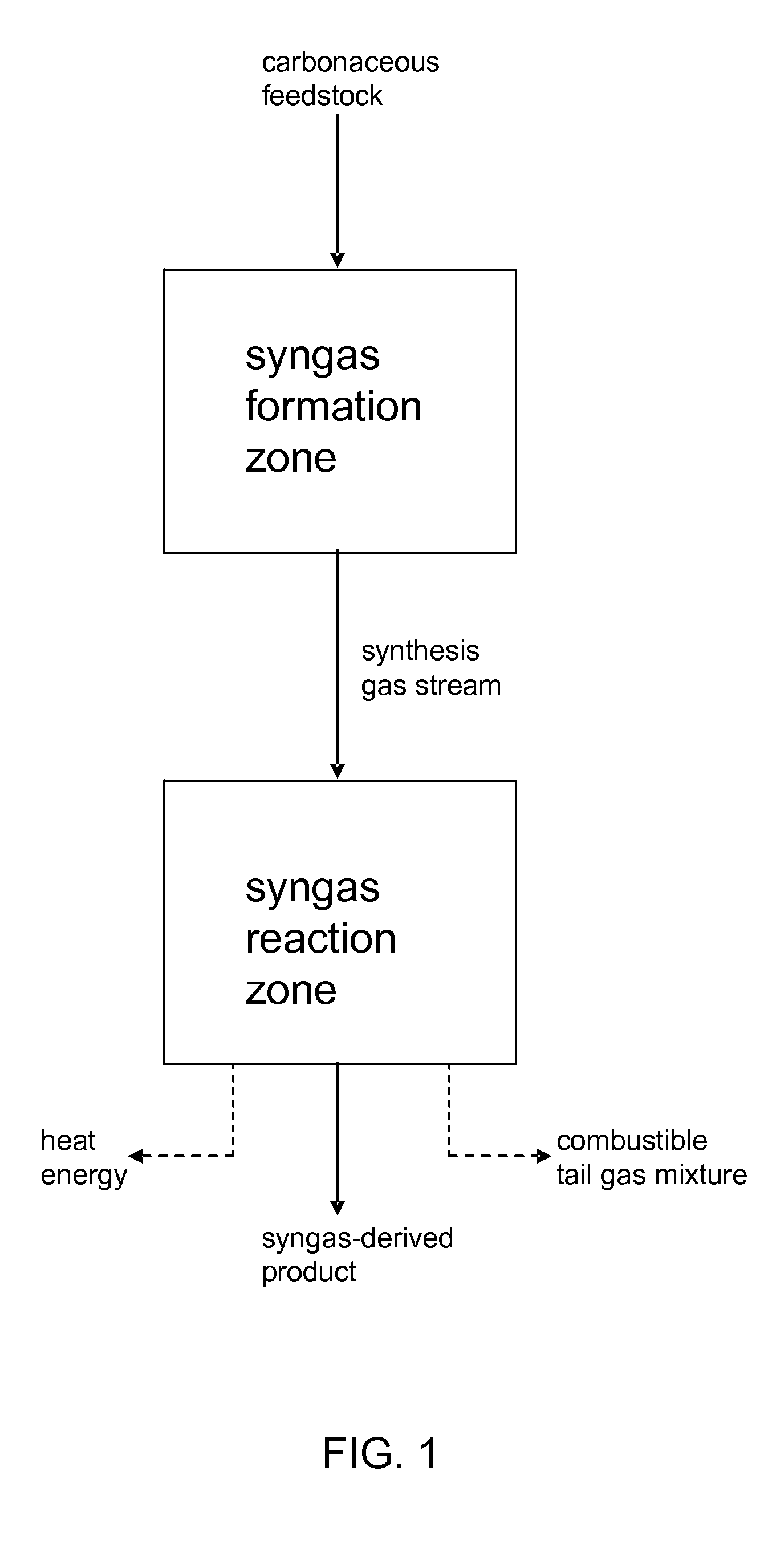
Processes for Making Synthesis Gas and Syngas-Derived Products
The present invention provides processes for making synthesis gas and processes for making syngas-derived products. For example, one aspect of the present invention provides a process for making a synthesis gas stream comprising hydrogen and carbon monoxide, the process comprising (a) providing a carbonaceous feedstock; (b) reacting the carbonaceous feedstock in a gasification reactor in the presence of steam and a gasification catalyst under suitable temperature and pressure to form a raw product gas stream comprising a plurality of gases comprising methane, hydrogen and carbon monoxide; (c) removing steam from and sweetening the raw product gas stream to form a sweetened gas stream; (d) separating and adding steam to the sweetened gas stream to form a first reformer input gas stream having a first steam / methane ratio; and a second reformer input stream having a second steam / methane ratio, in which the first steam / methane ratio is smaller than the second steam / methane ratio; (e) reforming the second reformer input stream to form a recycle gas stream comprising steam, carbon monoxide and hydrogen; (f) introducing the recycle gas stream to the gasification reactor; and (g) reforming the first reformer input stream to form the synthesis gas stream.
Owner:SURE CHAMPION INVESTMENT LTD
Conversion of petroleum residua to methane
InactiveUS6955695B2Eliminate needReduce usageThermal non-catalytic crackingElectrolysis componentsParticulatesGas phase
This invention discloses improvements on previous inventions for catalytic conversion of coal and steam to methane. The disclosed improvements permit conversion of petroleum residua or heavy crude petroleum to methane and carbon dioxide such that nearly all of the heating value of the converted hydrocarbons is recovered as heating value of the product methane. The liquid feed is distributed over a fluidized solid particulate catalyst containing alkali metal and carbon as petroleum coke at elevated temperature and pressure from the lower stage and transported to the upper stage of a two-stage reactor. Particulate solids containing carbon and alkali metal are circulated between the two stages. Superheated steam and recycled hydrogen and carbon monoxide are fed to the lower stage, fluidizing the particulate solids and gasifying some of the carbon. The gas phase from the lower stage passes through the upper stage, completing the reaction of the gas phase.
Owner:PETRO2020
Coal Compositions for Catalytic Gasification
Particulate compositions are described comprising an intimate mixture of a coal and a gasification catalyst. The particulate compositions are gasified in the presence of steam to yield a plurality of gases including methane and at least one or more of hydrogen, carbon monoxide, carbon dioxide, hydrogen sulfide, ammonia and other higher hydrocarbons. Processes are also provided for the preparation of the particulate compositions and converting the particulate composition into a plurality of gaseous products.
Owner:SURE CHAMPION INVESTMENT LTD
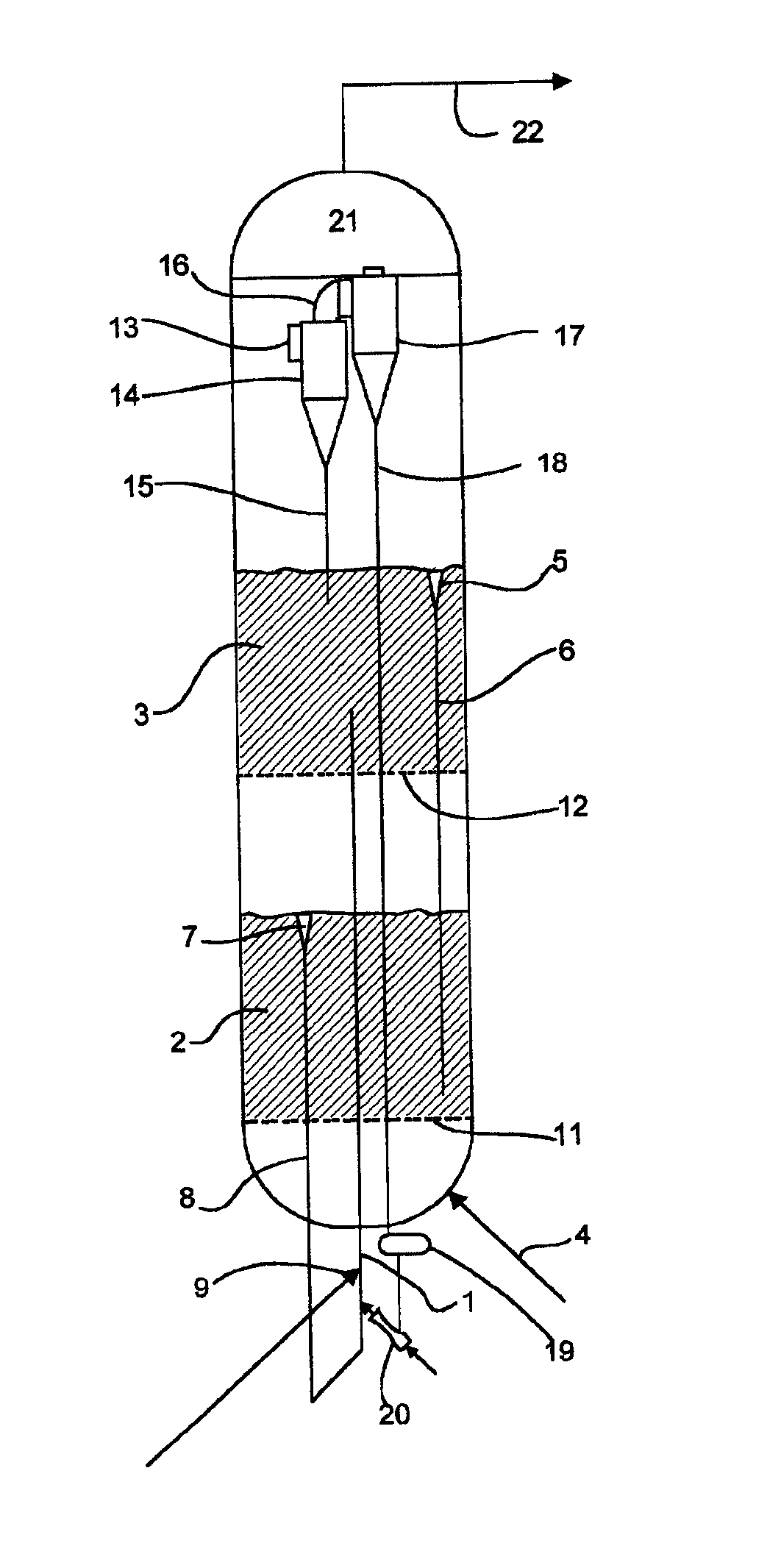
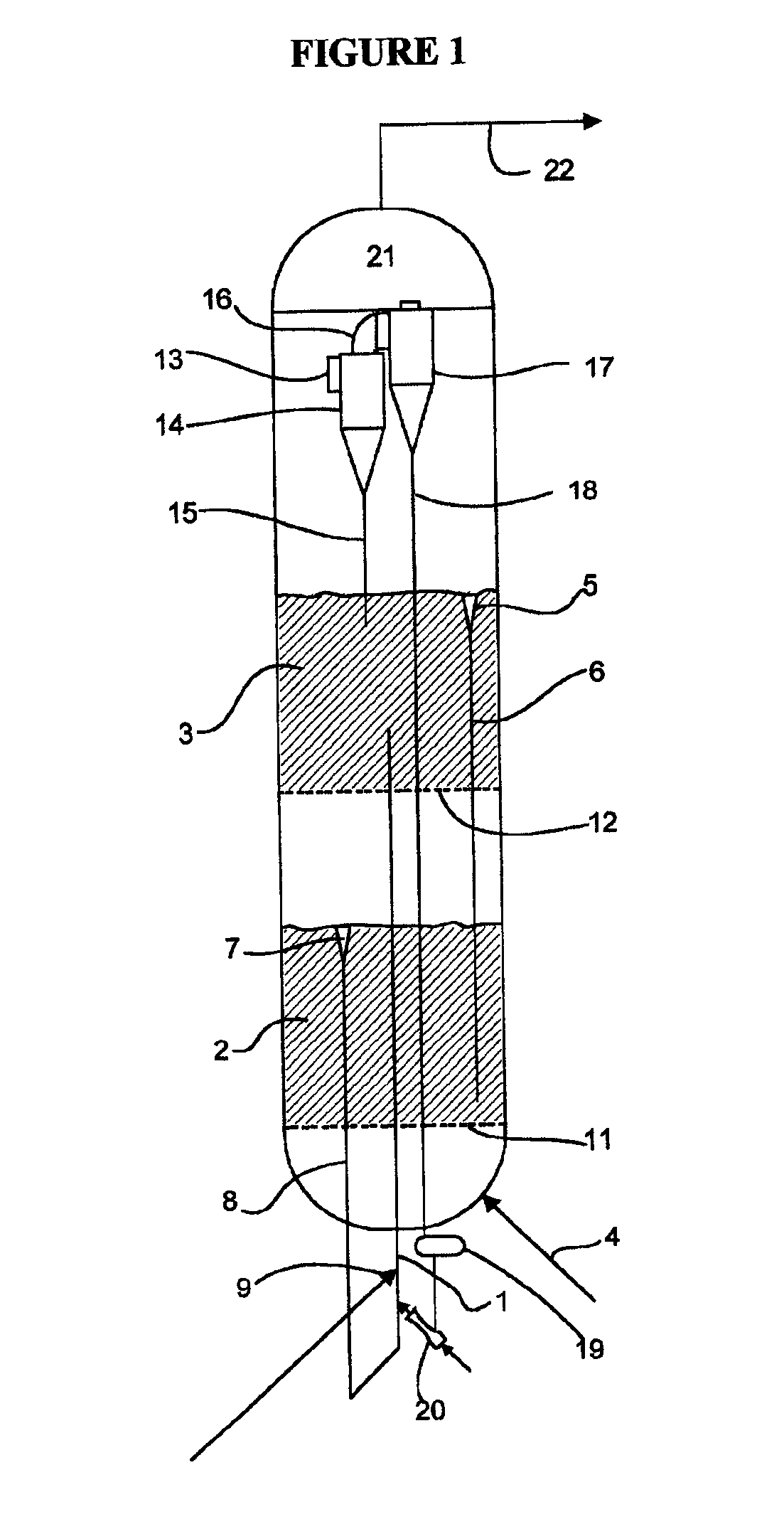
Processes for Making Syngas-Derived Products
The present invention provides processes for making syngas-derived products. For example, one aspect of the present invention provides a process for making a syngas-derived product, the process comprising (a) providing a carbonaceous feedstock; (b) converting the carbonaceous feedstock in a syngas formation zone at least in part to a synthesis gas stream comprising hydrogen and carbon monoxide; (c) conveying the synthesis gas stream to a syngas reaction zone; (d) reacting the synthesis gas stream in the syngas reaction zone to form the syngas-derived product and heat energy, a combustible tail gas mixture, or both; (e) recovering the syngas-derived product; and (f) recovering the heat energy formed from the reaction of the synthesis gas stream, burning the combustible tail gas mixture to form heat energy, or both.
Owner:SURE CHAMPION INVESTMENT LTD
Plant and a method for increased oil recovery
Owner:DEN NORSKE STATS OLJESELSKAP AS
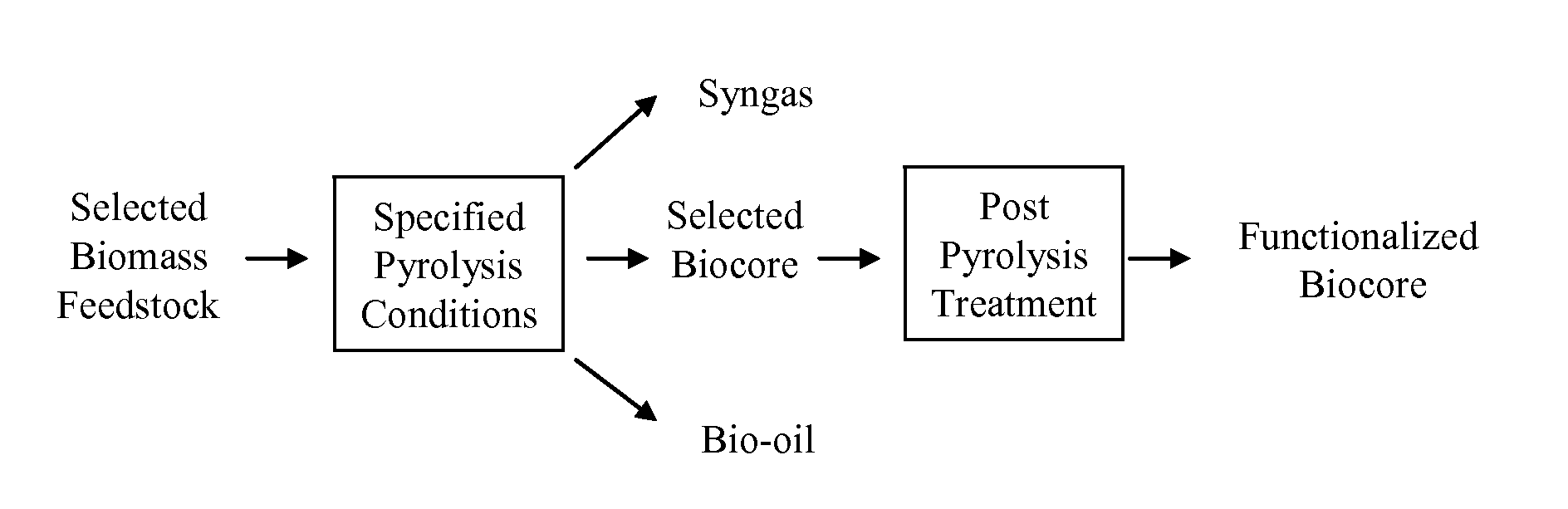
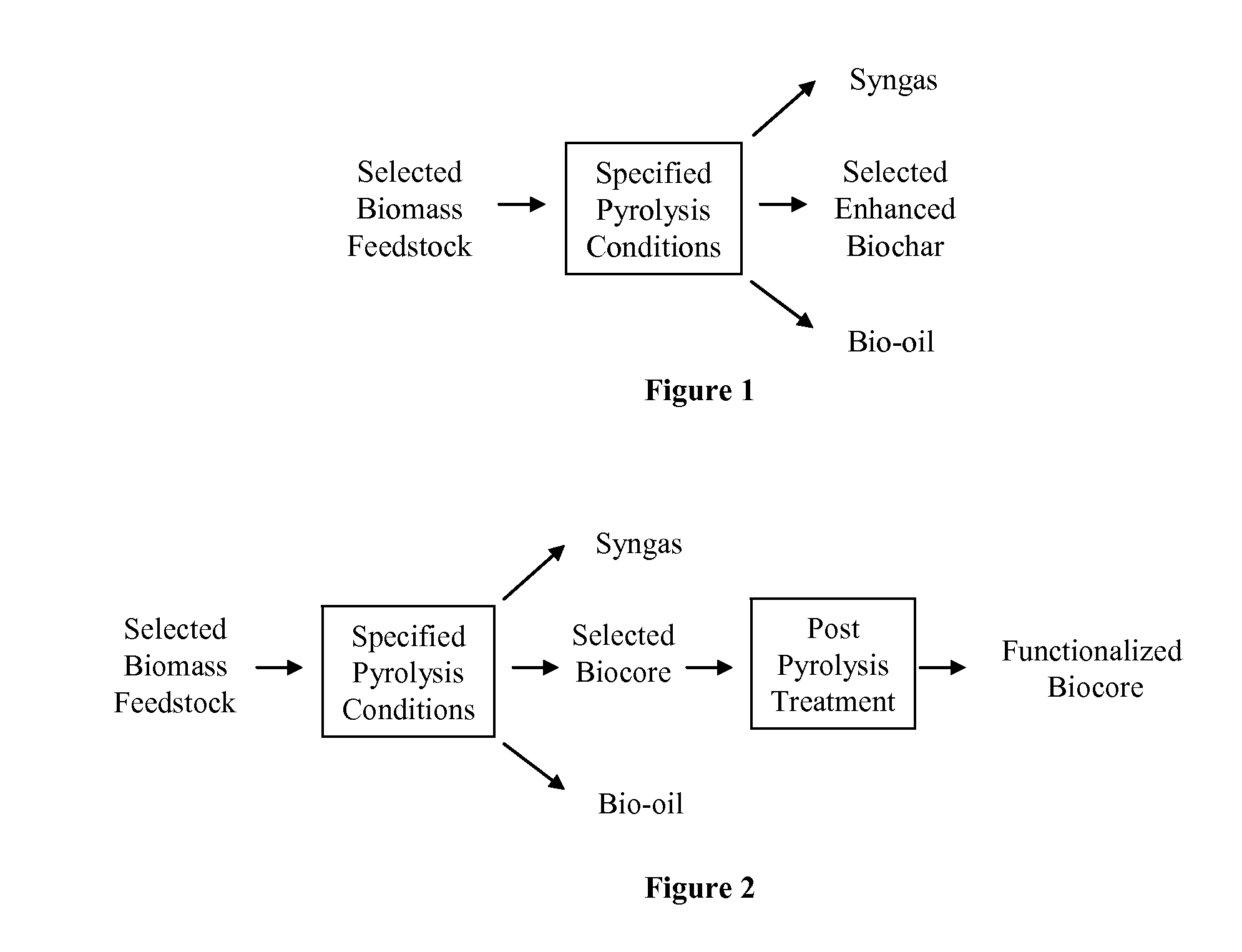
Biochar
ActiveUS8361186B1Enhanced and functionalizedCalcareous fertilisersMagnesium fertilisersForming gasBiological activation
The invention provides for methods, devices, and systems for pyrolyzing biomass. A pyrolysis unit can be used for the pyrolysis of biomass to form gas, liquid, and solid products. The biomass materials can be selected such that an enhanced biochar is formed after pyrolysis. The biomass can be pyrolyzed under specified conditions such that a selected biochar core is formed. The pyrolysis process can form a stable biochar core that is inert and / or resistant to degradation. The biochar or biochar core can be functionalized to form a functionalized biochar or functionalized biochar core. Functionalization can include post-pyrolysis treatments such as supplementation with microbes or physical transformations including annealing and / or activation.
Owner:FULL CIRCLE BIOCHAR
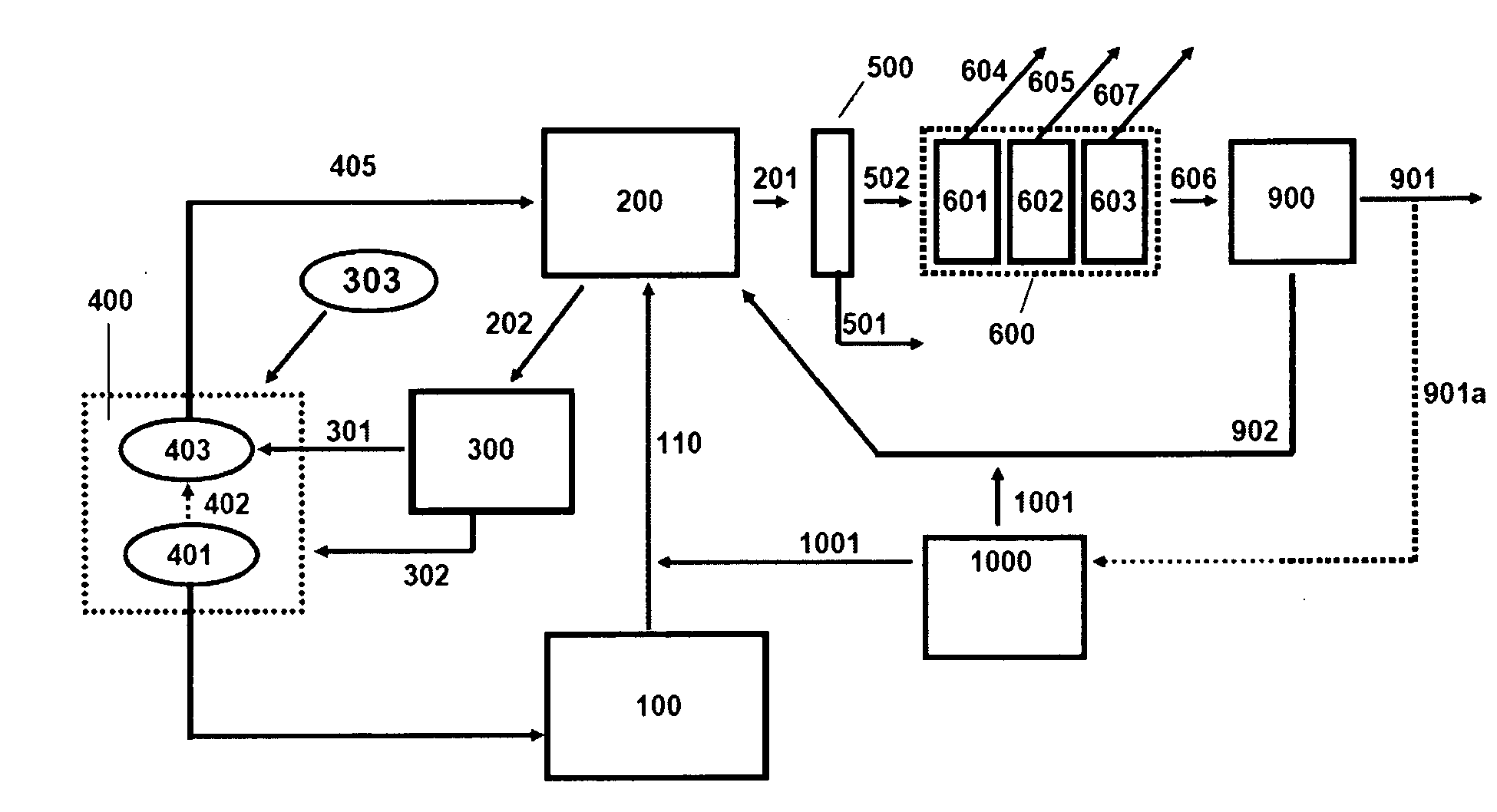
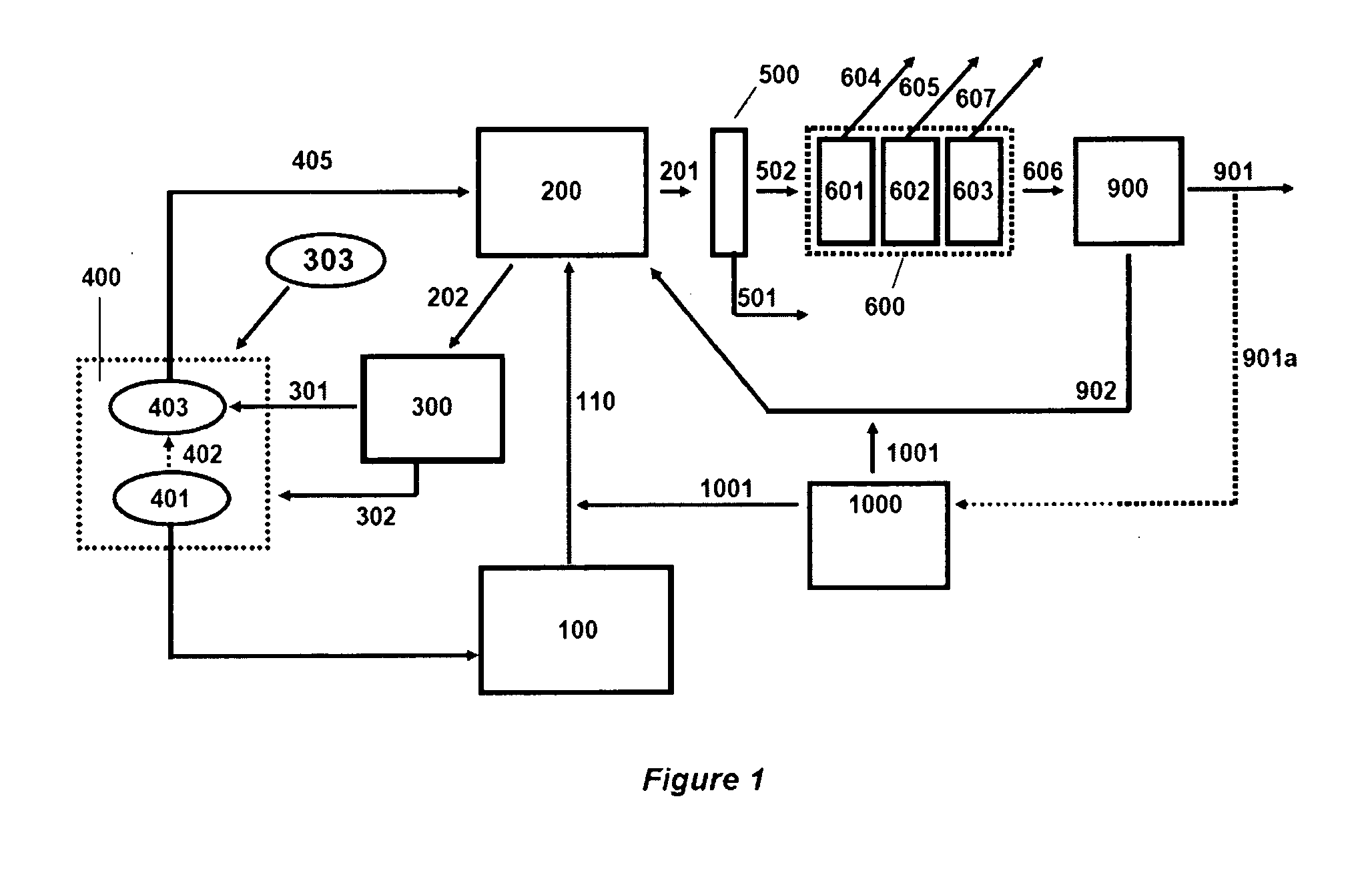
Co-Feed of Biomass as Source of Makeup Catalysts for Catalytic Coal Gasification
Continuous processes are provided for converting a carbonaceous feedstock comprising biomass containing alkali metal, non-biomass components, and at least one gasification catalyst including an alkali metal recovered from solid char, into a plurality of gaseous products including methane and at least one or more of hydrogen, carbon monoxide, and other higher hydrocarbons.
Owner:SURE CHAMPION INVESTMENT LTD
Continuous Process for Converting Carbonaceous Feedstock into Gaseous Products
Continuous processes for converting a carbonaceous feedstock into a plurality of gaseous products are described. The continuous processes include, among other steps, recovering a substantial portion of alkali metal from the solid char that results from the gasification of a carbonaceous feedstock. The alkali metal is recovered as an alkali metal carbonate. A gasification catalyst for a subsequent gasification step may comprise the recovered alkali metal carbonate and a makeup amount of alkali metal hydroxide.
Owner:SURE CHAMPION INVESTMENT LTD
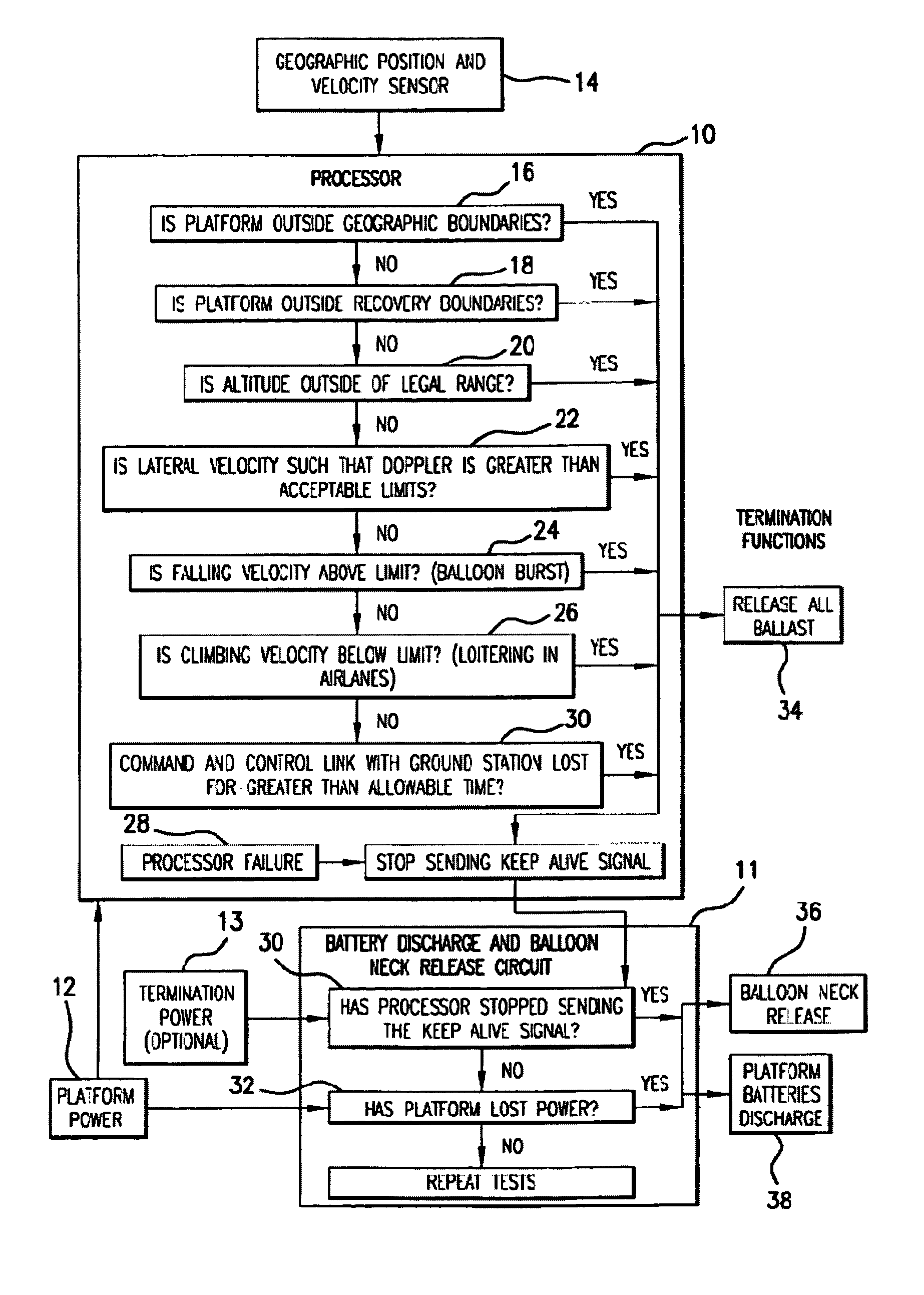
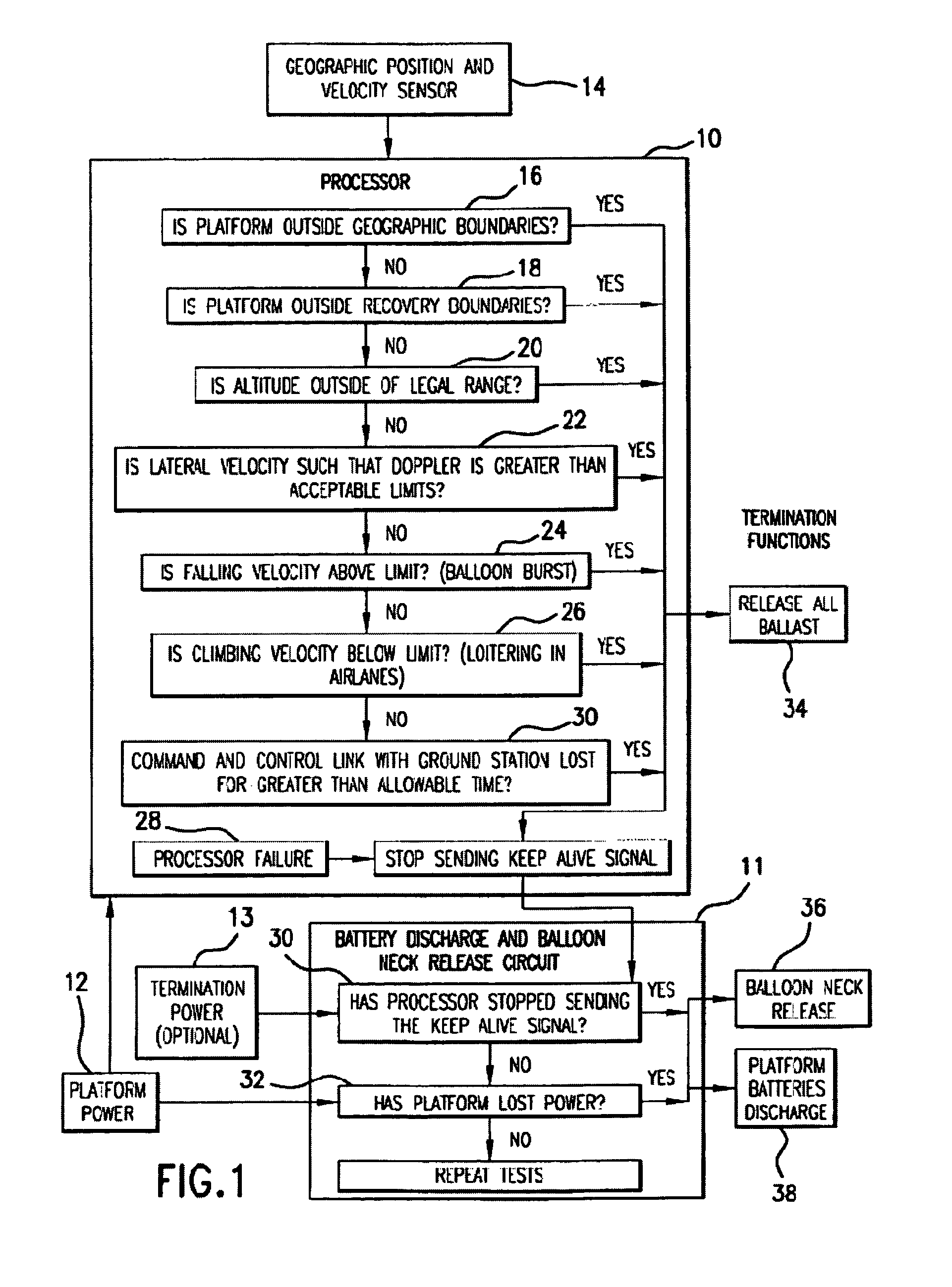
Systems and applications of lighter-than-air (LTA) platforms
InactiveUS7356390B2Large costLarge deploymentDigital data processing detailsPosition fixationGeolocationEngineering
Innovative new methods in connection with lighter-than-air (LTA) free floating platforms, of facilitating legal transmitter operation, platform flight termination when appropriate, environmentally acceptable landing, and recovery of these devices are provided. The new systems and methods relate to rise rate control, geo-location from a LTA platform including landed payload and ground-based vehicle locations, and steerable recovery systems.
Owner:SPACE DATA CORP
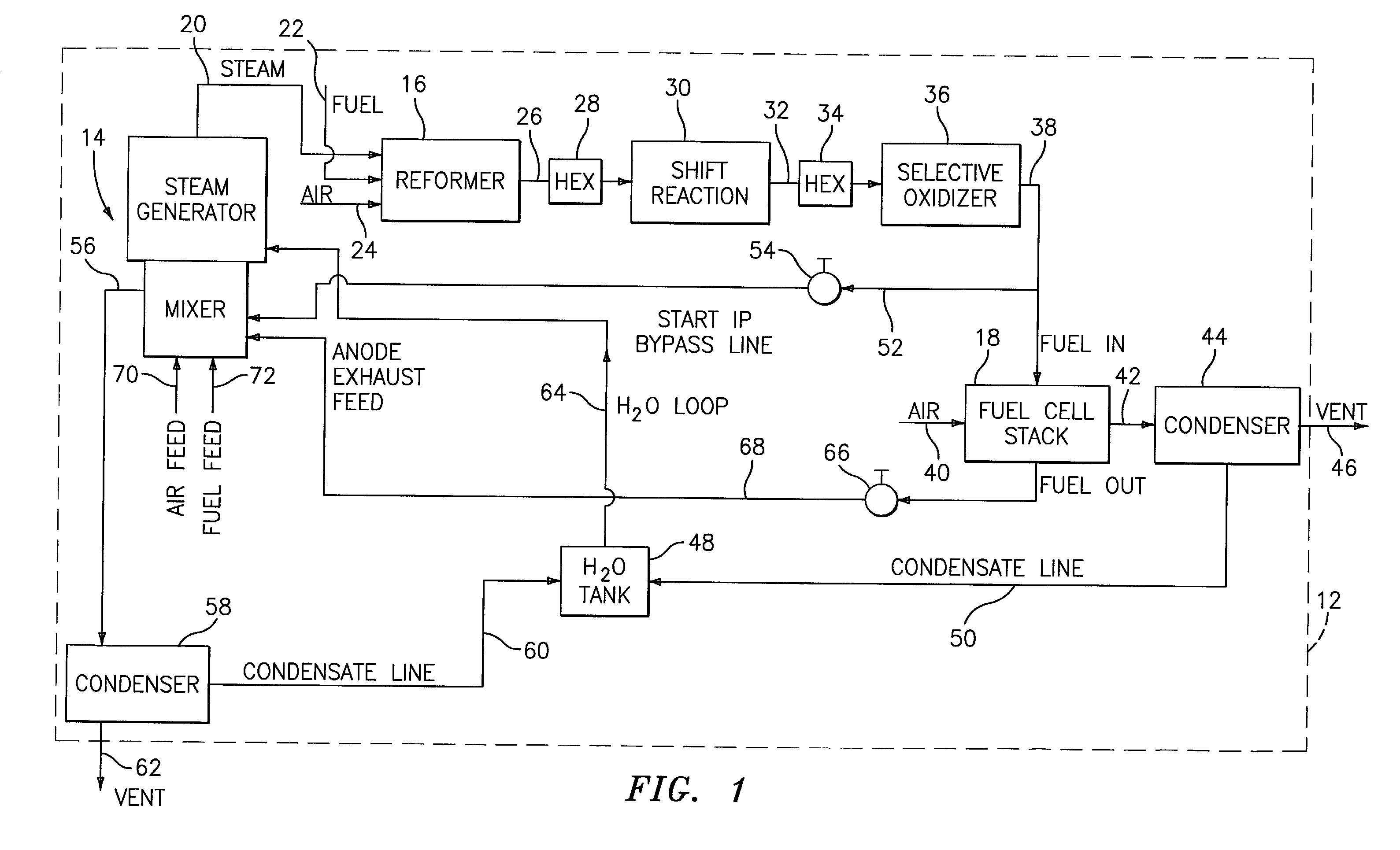
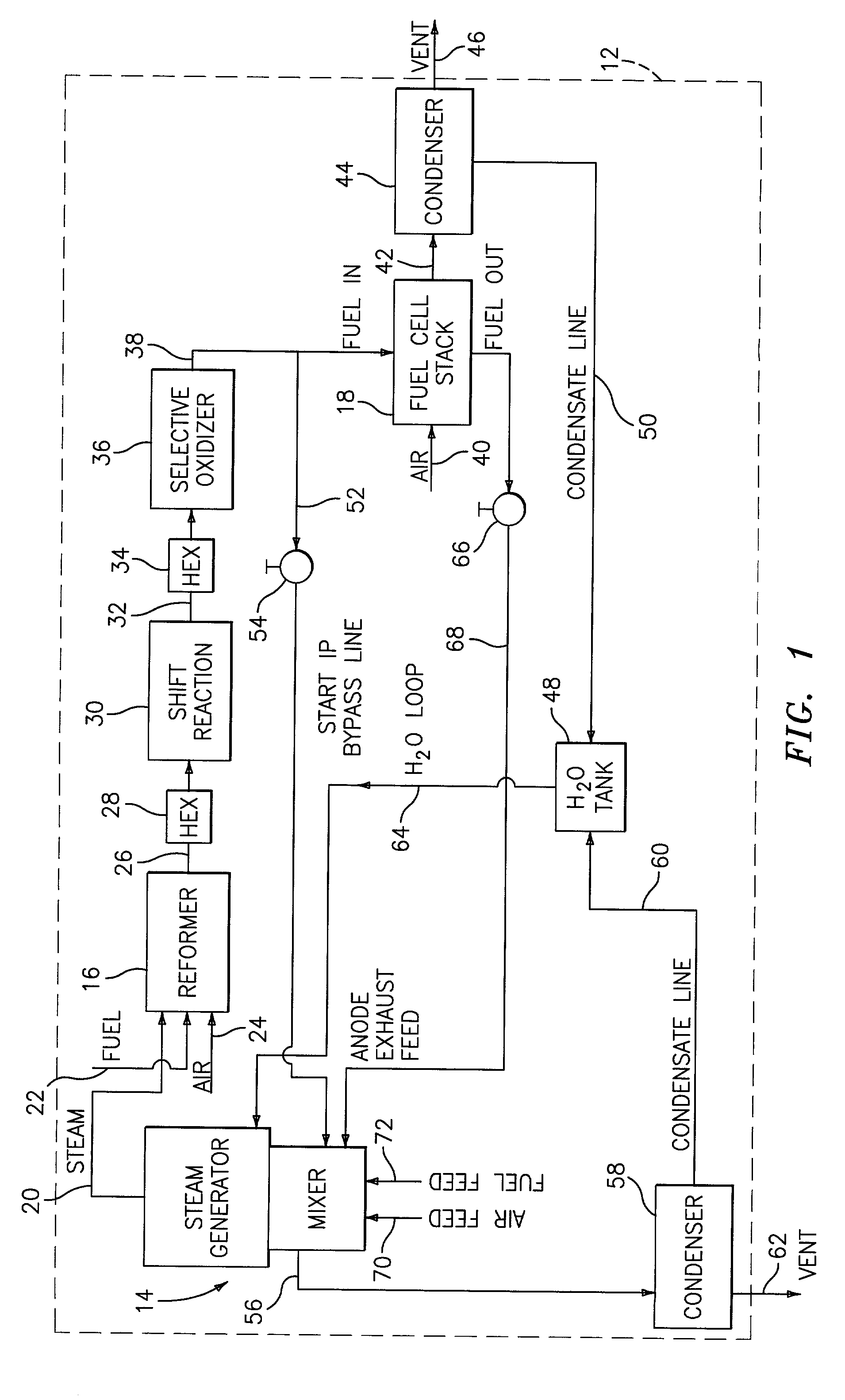
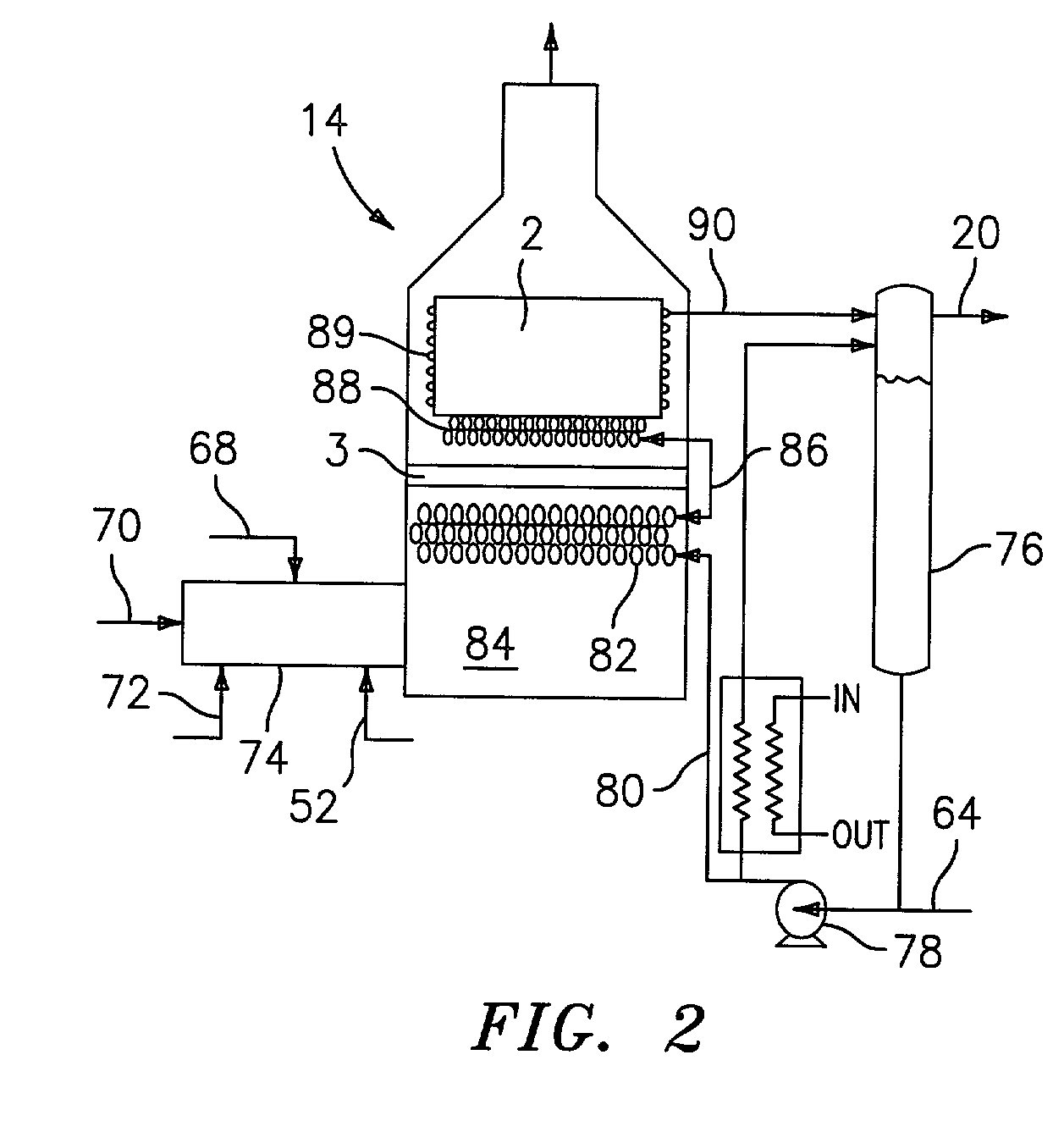
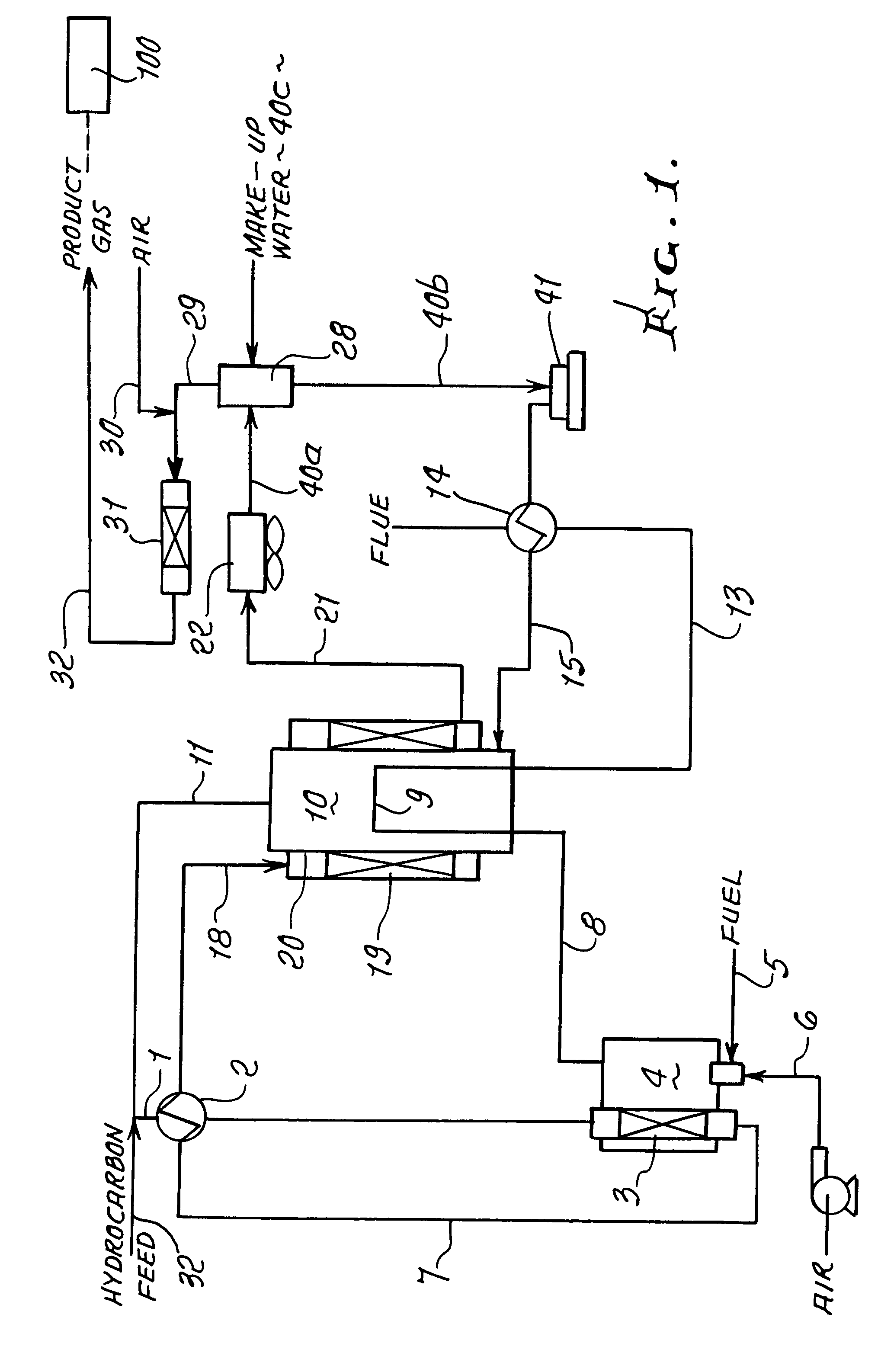
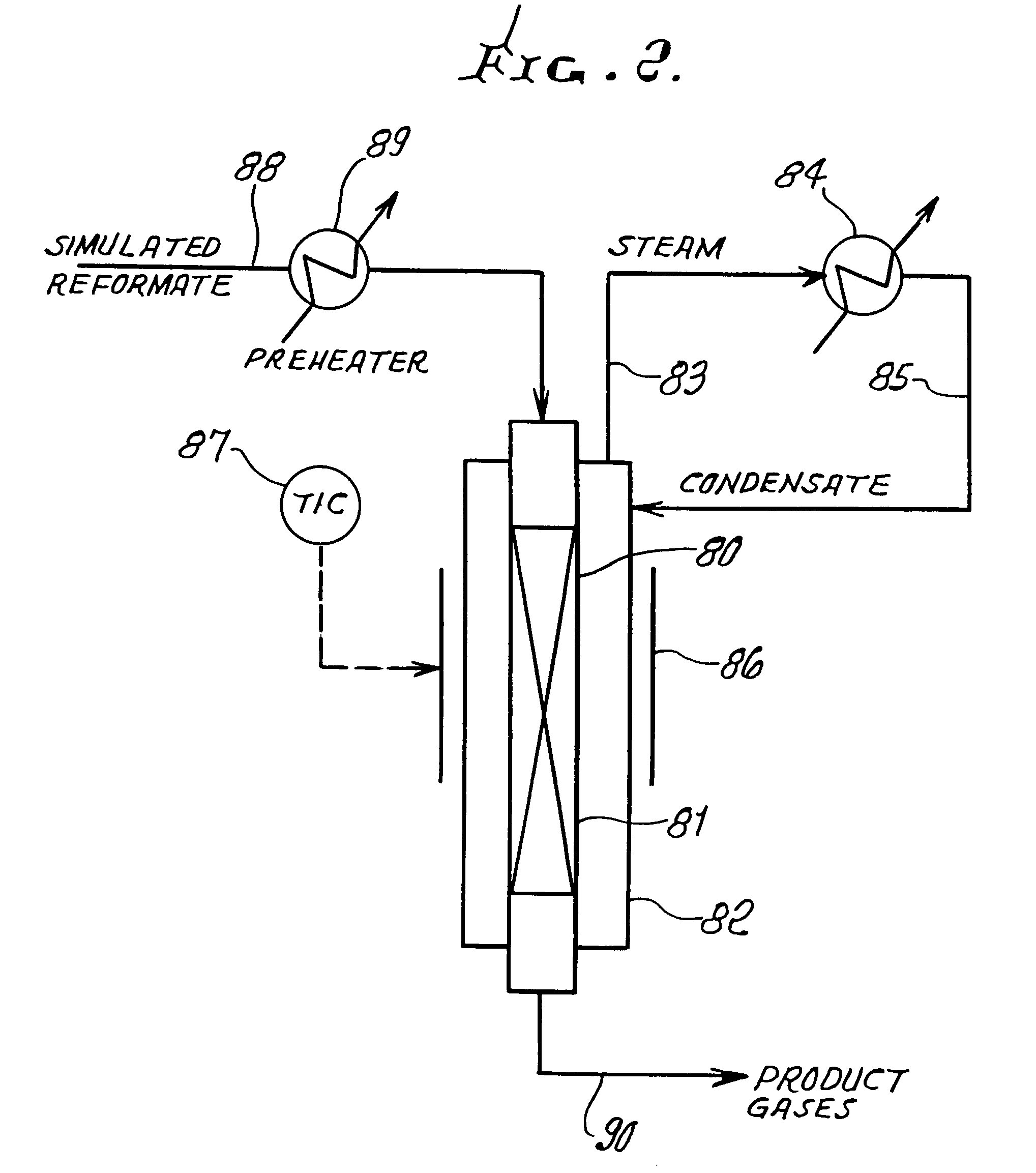
Steam generator for a PEM fuel cell power plant
A burner assembly includes a catalyzed burner for combusting an anode exhaust stream from a polymer electrolyte membrane (PEM) fuel cell power plant. The catalysts coated onto the burner can be platinum, rhodium, or mixtures thereof. The burner includes open cells which are formed by a lattice, which cells communicate with each other throughout the entire catalyzed burner. Heat produced by combustion of hydrogen in the anode exhaust stream is used to produce steam for use in a steam reformer in the PEM fuel cell assembly. The catalyzed burner has a high surface area wherein about 70-90% of the volume of the burner is preferably open cells, and the burner has a low pressure drop of about two to three inches water from the anode exhaust stream inlet to the anode exhaust stream outlet . The burner assembly operates at essentially ambient pressure and at a temperature of up to about 1,700° F. (646° C.). The burner assembly can combust anode exhaust during normal operation of the fuel cell assembly. The burner assembly also includes an adjunct burner which can combust gasoline or anode bypass gas (the latter of which is a reformed fuel gas which is tapped off of the fuel cell stack fuel inlet line) during startup of the fuel cell power plant. Once start up of the fuel cell power plant is achieved, the burner assembly will need only combustion of the anode exhaust by the catalytic burner to produce steam for the reformer.
Owner:BALLARD POWER SYSTEMS
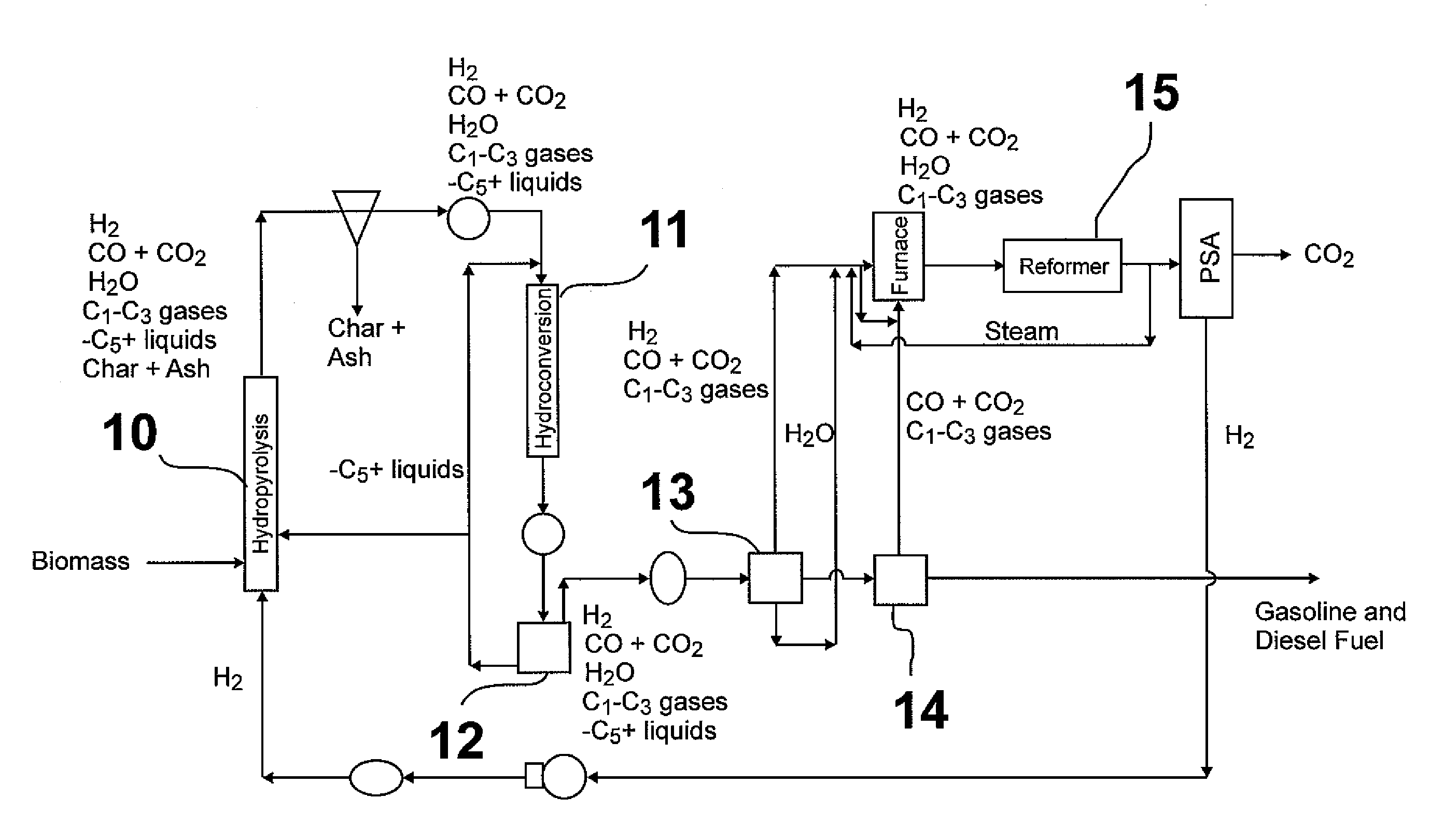
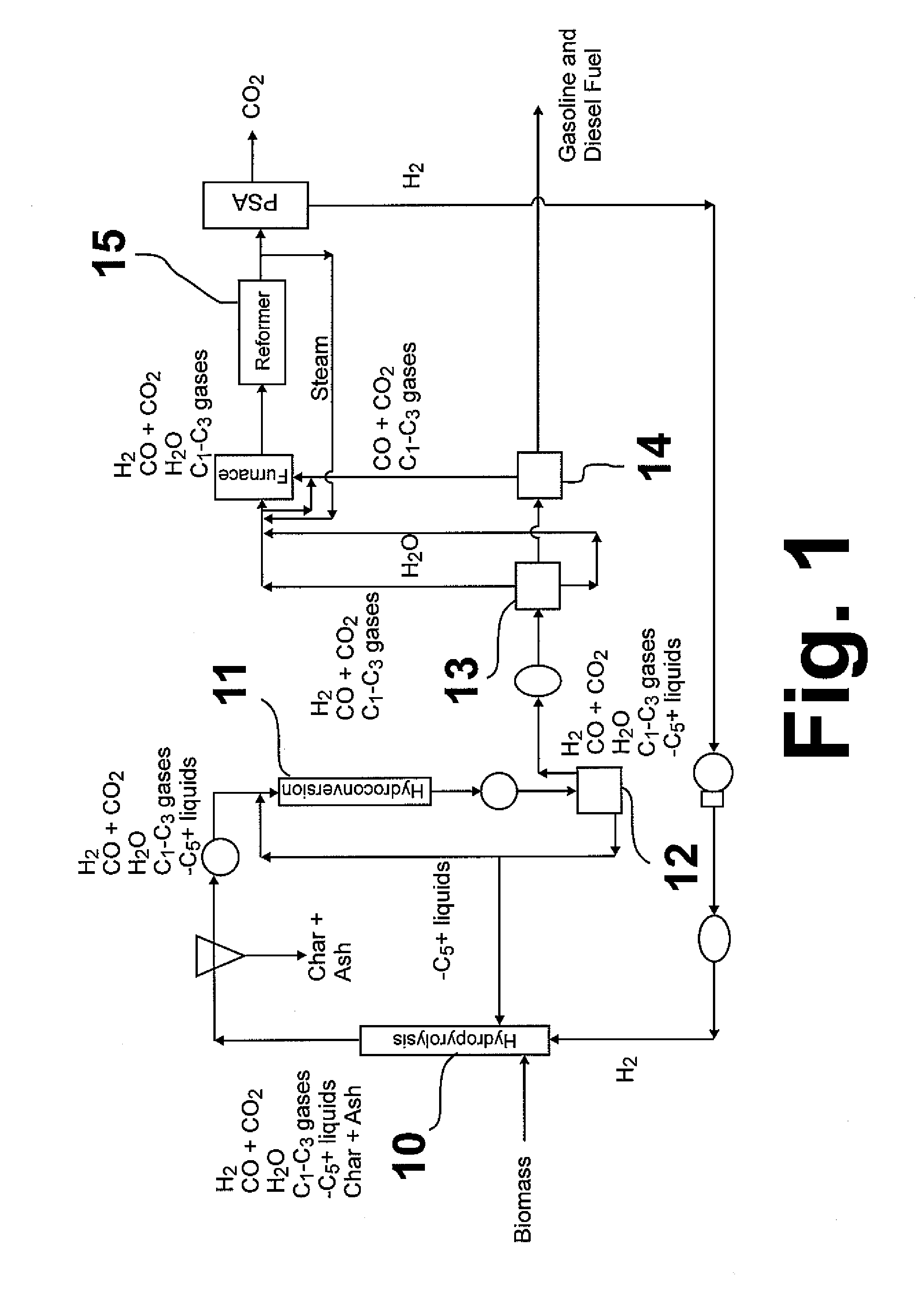
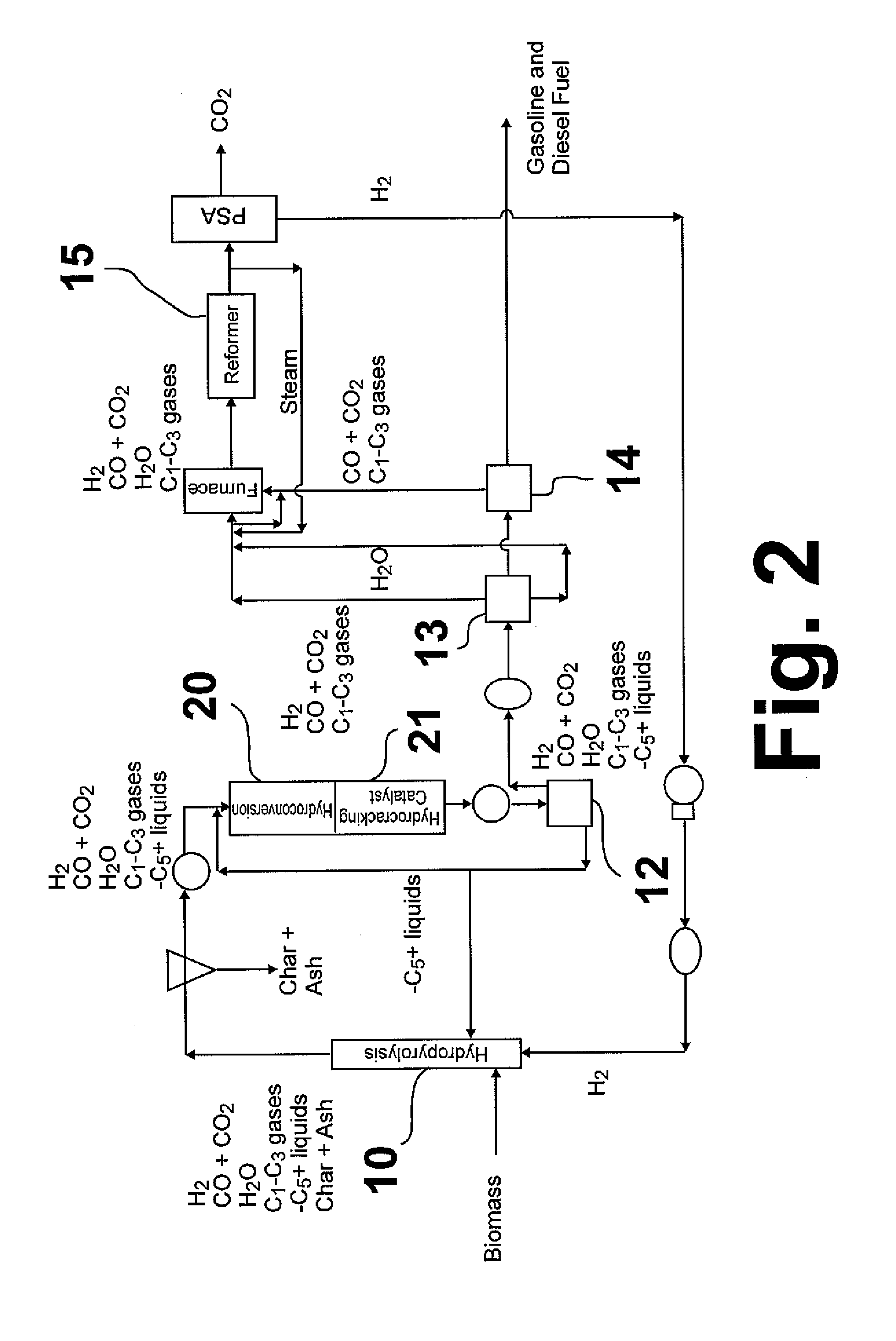
Hydropyrolysis of biomass for producing high quality liquid fuels
A self-sustaining process for producing high quality liquid fuels from biomass in which the biomass is hydropyrolyzed in a reactor vessel containing molecular hydrogen and a deoxygenating catalyst, producing a partially deoxygenated hydropyrolysis liquid, which is hydrogenated using a hydroconversion catalyst, producing a substantially fully deoxygenated hydrocarbon liquid and a gaseous mixture comprising CO and light hydrocarbon gases (C1-C3). The gaseous mixture is reformed in a steam reformer, producing reformed molecular hydrogen, which is then introduced into the reactor vessel for hydropyrolizing the biomass. The deoxygenated hydrocarbon liquid product is further separated to produce diesel fuel, gasoline, or blending components for gasoline and diesel fuel.
Owner:GAS TECH INST
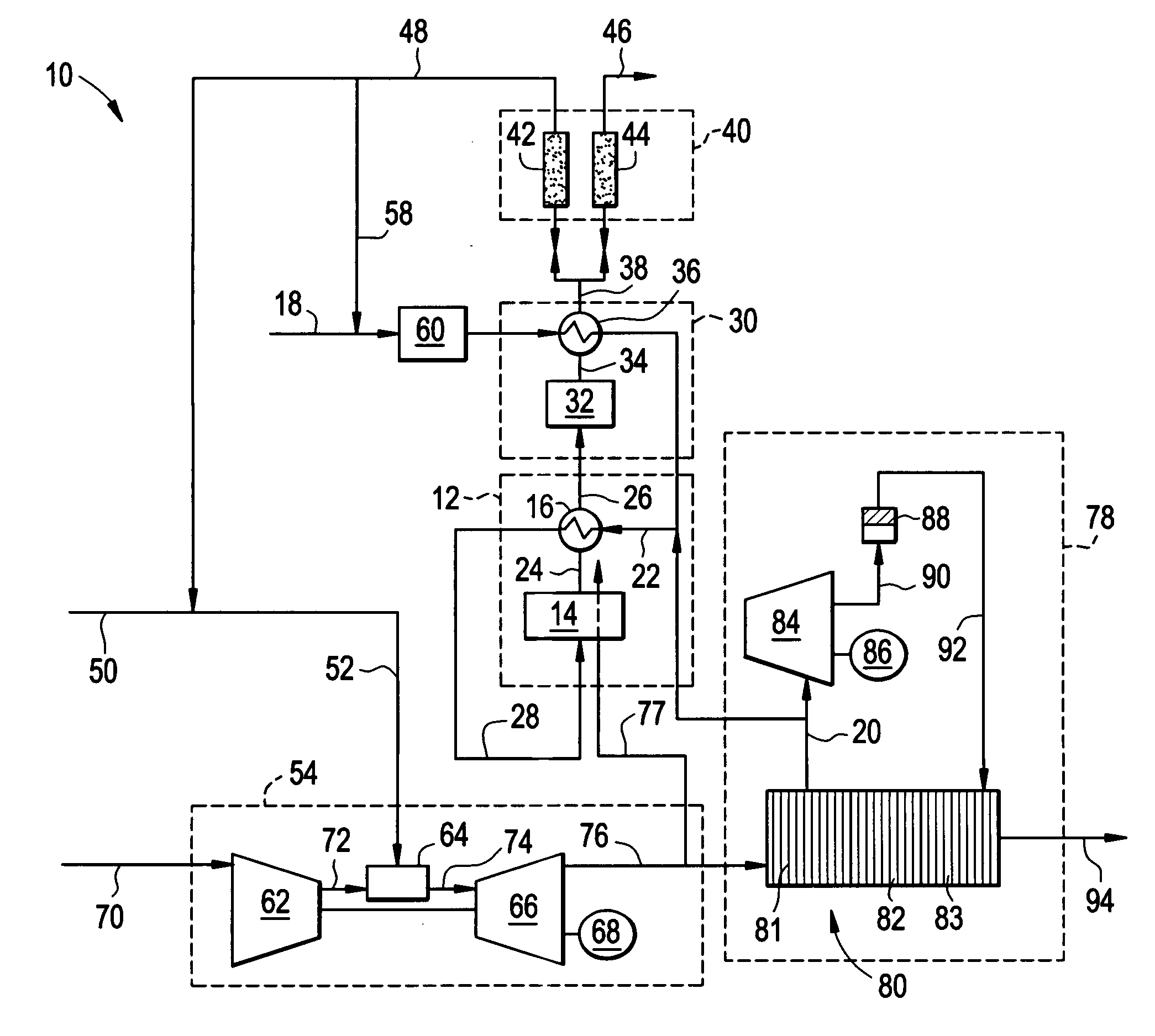
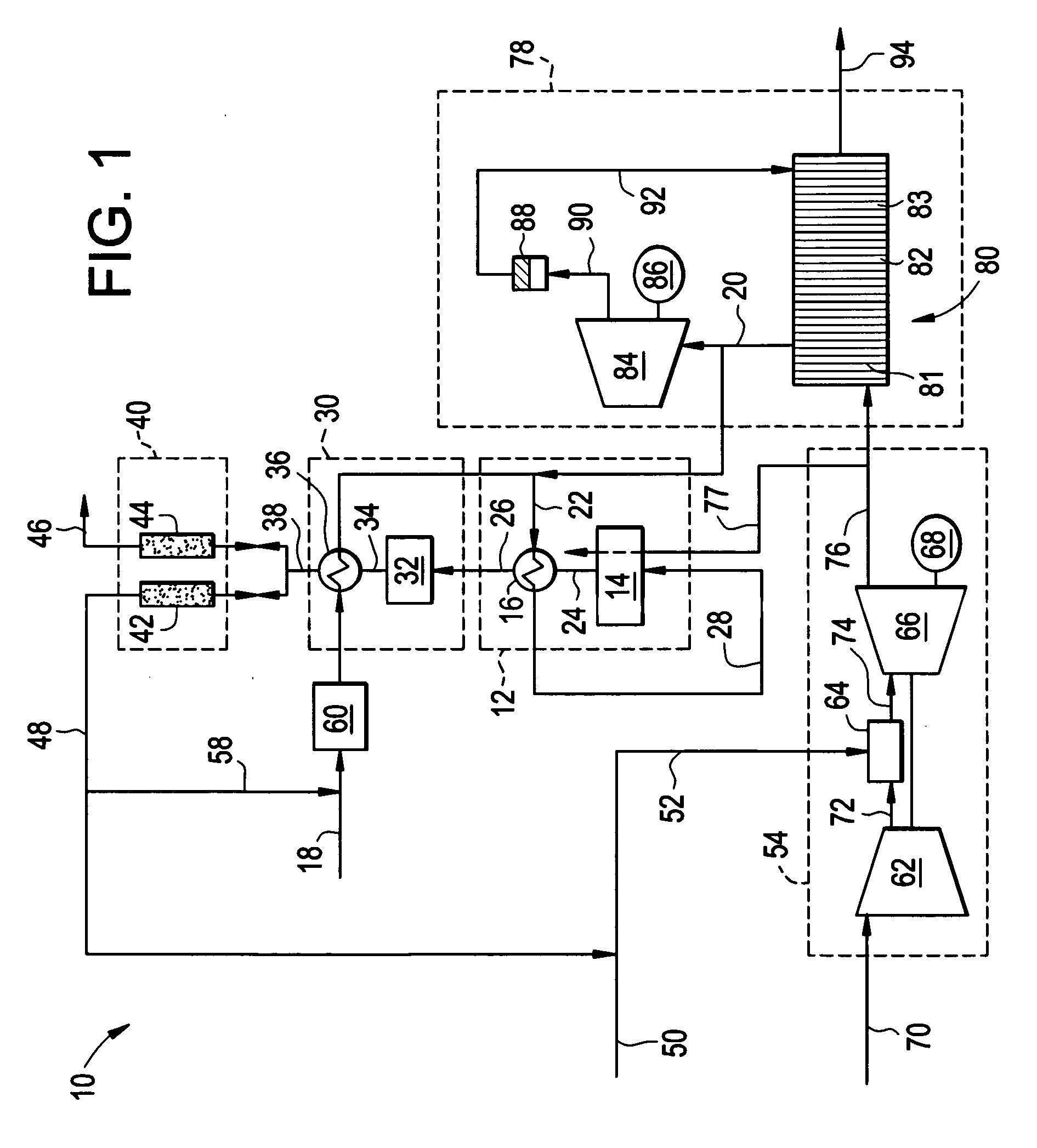
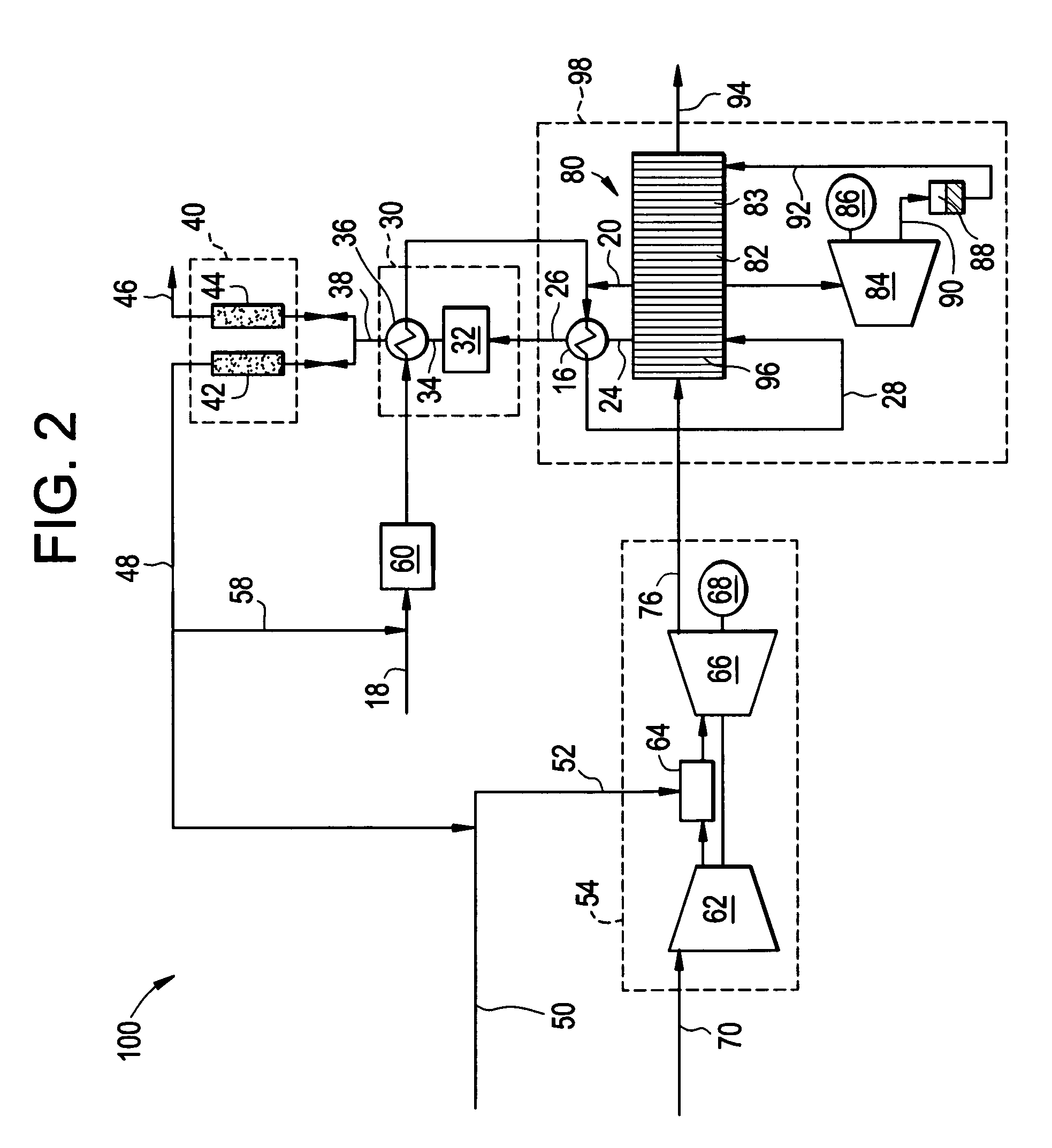
Reforming system for combined cycle plant with partial CO2 capture
A combined cycle system includes, a pre-steam-methane-reformer operating at a temperature of less than about 800 degrees Celsius to reform a mixed fuel stream to generate a first reformate stream, a water-gas-shift reactor to convert carbon monoxide in the first reformate stream to carbon dioxide and form a second reformate stream, a carbon dioxide removal unit for removing carbon dioxide from the second reformate stream and form a carbon dioxide stream and a third reformate stream; wherein less than about 50 percent of the carbon contained in the mixed fuel stream is recovered as carbon dioxide by the removal unit, a gas turbine unit for generating power and an exhaust stream, and a steam generator unit configured to receive the exhaust stream, wherein the heat of the exhaust stream is transferred to a water stream to generate the steam for the mixed fuel stream and for a steam turbine.
Owner:GENERAL ELECTRIC CO
Thermally-integrated low temperature water-gas shift reactor apparatus and process
InactiveUS7074373B1Reduced toleranceExtended temperature rangeHydrogenPhysical/chemical process catalystsWater-gas shift reactionProduct gas
A thermally-integrated lower temperature water-gas shift reactor apparatus for converting carbon monoxide in the presence steam comprises a catalyst bed that is disposed within an outer region surrounding a waste heat recovery steam generator operating at a selected pressure corresponding to the optimum temperature for conducting the catalytic water-gas shift reaction and a process for useful recovery of the exothermic heat of reaction to generate steam that is used in a process for the conversion of hydrocarbon feedstock into useful gases such as hydrogen.
Owner:HARVEST ENERGY TECH
Popular searches
Hydrogen/synthetic gas production Gas generation devices Chemical/physical/physico-chemical processes Liquid-gas reaction processes Ammonia compounds Combustion using gaseous and pulverulent fuel Continuous combustion chamber Combustion using liquid and pulverulent fuel Organic compound preparation Oxygen compounds preparation by reduction