Gentiana urnula formula granule, preparation method and detection method thereof
A technology for formula granules and gentian wunu, applied in the field of medicine, can solve the problems of troublesome decoction methods, inability to effectively control the efficacy of drugs, and non-decoction as required.
Inactive Publication Date: 2019-08-02
青海普兰特药业有限公司
View PDF3 Cites 1 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0004] The traditional decoction method of Wunu gentian decoction pieces is extremely troublesome. Patients do not know much about the amount of water added, soaking time, heat, decoction time, decoction first and then decoction, etc., resulting in not decocting according to the requirements
The traditional method of decocting traditional Chinese medicine with water cannot effectively control the efficacy of the medicine, which leads to the inability to guarantee the safety of the medicine
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0063] Example 1: 400 g of dry paste powder of gentian gentian, 0.8 g of dextrin, and 1 g of polyethylene glycol.
Embodiment 2
[0064] Example 2: 550 g of dry paste powder of gentian gentian, 1.1 g of dextrin, and 1.7 g of polyethylene glycol.
Embodiment 3
[0065] Example 3: 500 g of dry cream powder of gentian gentian, 1 g of dextrin, and 1.5 g of polyethylene glycol.
[0066] The formulations of Examples 1-3 are prepared according to any preparation method of Examples 4-6, and tested according to any detection method of Examples 7-13 after being made.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
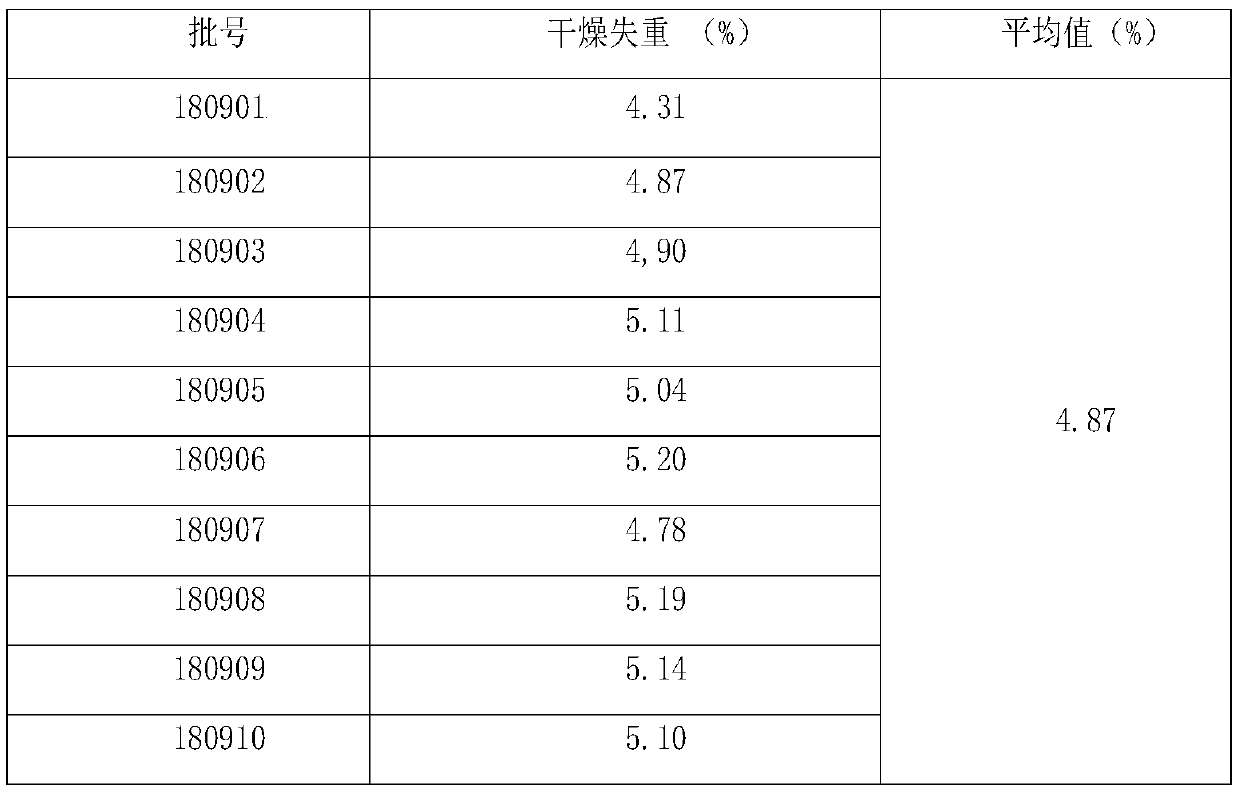
The invention relates to a gentiana urnula formula granule, a preparation method and a detection method. The formula granule is composed of dry gentiana urnula paste powder, dextrin and polyethylene glycol. The gentiana urnula formula granule is prepared from a single medicine of gentiana urnula through extracting, concentrating, drying and pelletizing; treatment based on syndrome differentiationby a physician can be facilitated, the dosage is increased or decreased based on the syndrome; the detecting content is to identify gentiana urnula glycoside a through the thin layer chromatography, and detect the content of the gentiana urnula glycoside a in the formula granule through the liquid chromatography. By adopting the method, under different thin layer plates, temperature and humidity conditions, the spots on chromatograms are clear, the separation is in accord with the requirements, the Rf value is moderate, and better identification chromatography can be obtained under different conditions; a verification test shows that the method provided by the invention is good in reproducibility and durability, the liquid chromatography is adopted to detect the content of the gentiana urnula glycoside a, the result is accurate and reliable; the method is simple, good in repeatability, and can be used for effectively controlling the quality of the gentiana urnula formula granule.
Description
technical field [0001] The invention relates to the field of medicine, in particular to a preparation method and a quality detection method of Wunu Gentian formula granules. Background technique [0002] Wunu Gentian (scientific name: Gentiana urnula H.Smith): a perennial herb of Gentianaceae, 4-6 cm high, with well-developed stolons. Fibrous roots are numerous, slightly fleshy, pale yellow. Branches numerous, sparsely clustered, erect, very low, with shortened internodes. The leaves are densely arranged in imbricate tiles, the base is black-brown residual leaves, the middle is yellow-brown dead leaves, the upper part is green or lavender fresh leaves, fan-shaped and truncated, flowers are solitary, and 2-3 clusters are rarely clustered on the top of branches. Base surrounded by upper leaves; no pedicel; calyx tube membranous, lobes green or purple, leaf-shaped, same shape as leaves, capsule exposed, ovate-lanceolate, 1.5-1.8 cm long, apex acute, base obtuse , slender sta...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): A61K36/515A61K9/16A61K47/10A61K47/36G01N30/02G01N30/06G01N30/54G01N30/74G01N30/90G01N30/94
CPCA61K9/1641A61K9/1652A61K36/515A61K2236/331A61K2236/39A61K2236/51G01N30/02G01N30/06G01N30/54G01N30/74G01N30/90G01N30/94G01N2030/027G01N2030/062
Inventor 余梅郭静刘红昝占全陈钟
Owner 青海普兰特药业有限公司