Kit for predicting, preventing and treating myopia
A kit and myopia technology, applied in the biological field, to achieve the effect of good diagnosis and treatment, improve acceptance, and reduce the burden of treatment
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0024] Embodiment 1 clinical experiment
[0025] 1 method
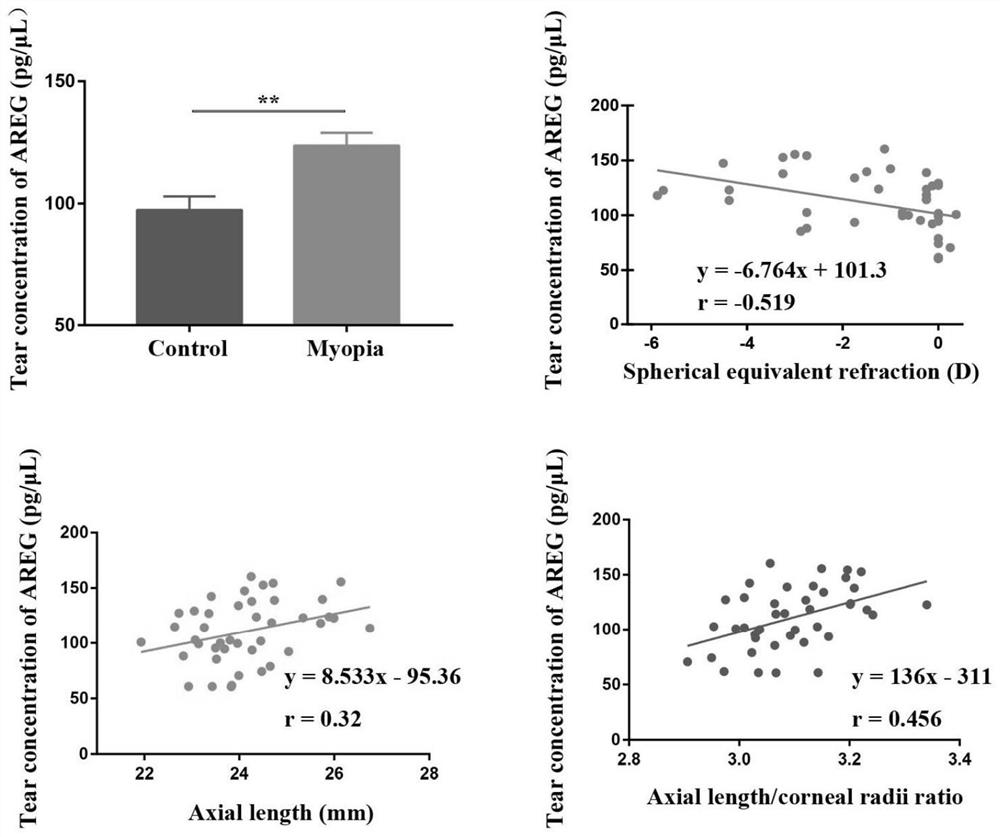
[0026] The clinical subjects recruited into the group were subjected to subjective refraction with a comprehensive refractometer and axial length measurement with an IOL-master, and the tears were collected with Schirmer test paper (collection time was 5 minutes) and then quickly immersed in 500 μL of protein lysate (BC3710; Beijing Suolaibao Technology Co., Ltd., China) EP tube, and stored at -20 ° C, followed by ELISA method to detect the concentration of AREG protein in each sample.
[0027] Inclusion criteria: age 18-40 years old, corrected visual acuity 1.0, anisometropia ≤ 1.50D
[0028] Exclusion criteria: systemic diseases, long-term medication history, eye surgery history, ocular surface diseases, eye inflammation, laser surgery, etc.
[0029] Grouping: myopia group (equivalent spherical lens -0.5 to -6.0D)
[0030] Emmetropic group (equivalent spherical lens +0.5 to -0.5D)
[0031] Inspection method: Mea...
Embodiment 2
[0042] Embodiment 2 animal experiments
[0043] 1 method
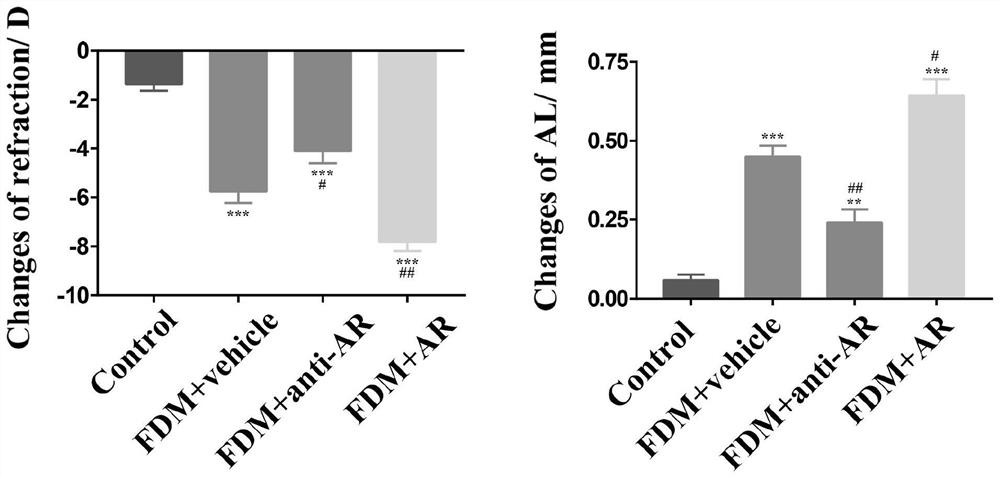
[0044] 2-week-old male tricolor guinea pigs were divided into normal group and myopia group. In the myopia group, guinea pigs in the myopia group were covered with the right eye to induce form-deprivation myopia (FDM). Starting from the third week of the cover, the AREG antibody (FDM+ anti-AR), AREG exogenous protein (FDM+AR), PBS (FDM+vehicle), once a week, continuous injection for 2 weeks, the diopter and axial length of each group were measured before and after the experiment.
[0045] FDM model induction method: 2-week-old male three-color guinea pigs were raised in a natural light environment. The guinea pigs wore a headgear made of translucent balloons to cover the right eye. Movements such as head rotation are not affected.
[0046] Measurement of biological parameters: After instilling tropicamide eye drops 4 times at intervals of 5 minutes, the guinea pigs were subjected to a strip photoretinoscopy (YZ24; Suzh...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More