Production of pseudotyped recombinant AAV virions
a technology of recombinant aav and virions, which is applied in the field of molecular biology, gene therapy, microbiology and virology, can solve the problems of inefficient methods, insufficient large-scale production of pseudotyped recombinant virions, and the preference of particular tissue of interest for delivery of conventional aav, etc., to achieve efficient, large-scale production of pseudotyped virions, and high purification and concentration of virion
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Materials and Methods
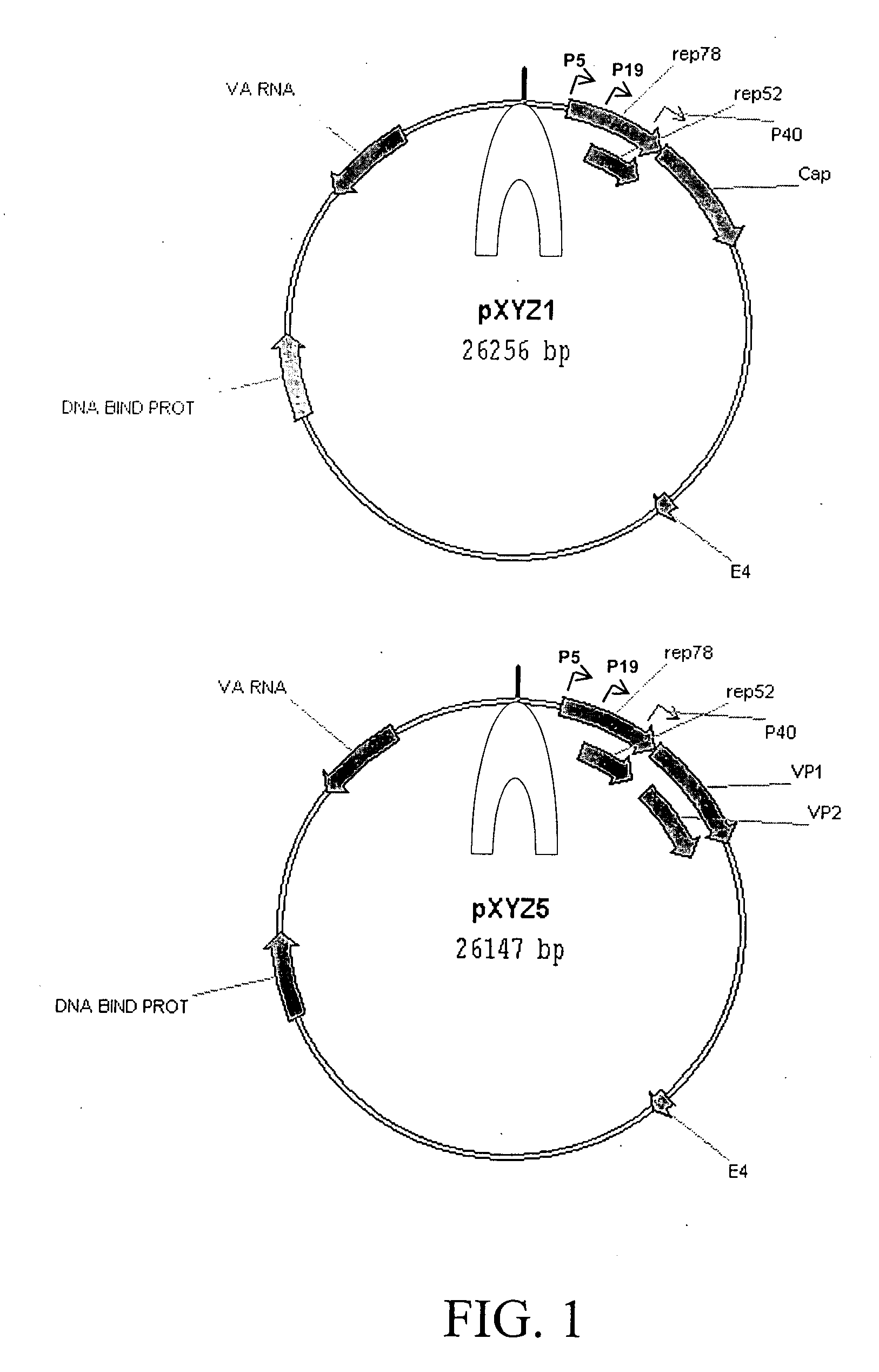
[0060] AAV helper plasmids were constructed by combining the ORF coding for the AAV2 Rep proteins and the ORF coding for capsid proteins of serotypes 1 and 5. The pACG2R1C helper plasmid was constructed by substituting the AAV1 cap ORF for AAV2 cap ORF in pACG2 (Li et al., J. Virol. 71:5236-5243, 1997) and a similar approach was applied to the pACG2R5C plasmid. Rep2cap1 and rep2cap5 helper cassettes were then subcloned into an Ad helper plasmid, pXYZ, constructed from pAdEasy. To construct pXYZ, several Ad genes (penton, core protein, hexon, and Ad DNA polymerase) were disrupted and the left hand end of the Ad genome was removed to eliminate the possibility of generating infectious Ad and Ad structural proteins (some of which are cytotoxic). The resultant plasmids pXYZ1 and pXYZ5 (FIG. 1) were used to pseudotype AAV2-ITR-containing expression cassettes into AAV1 and AAV5 capsids, respectively.
Construction of pXYZ1 and pXYZ5 Helper Plasmids
[0061] pXYZ Ad helpe...
example 2
Purification of rAAV1 and 5 Virions
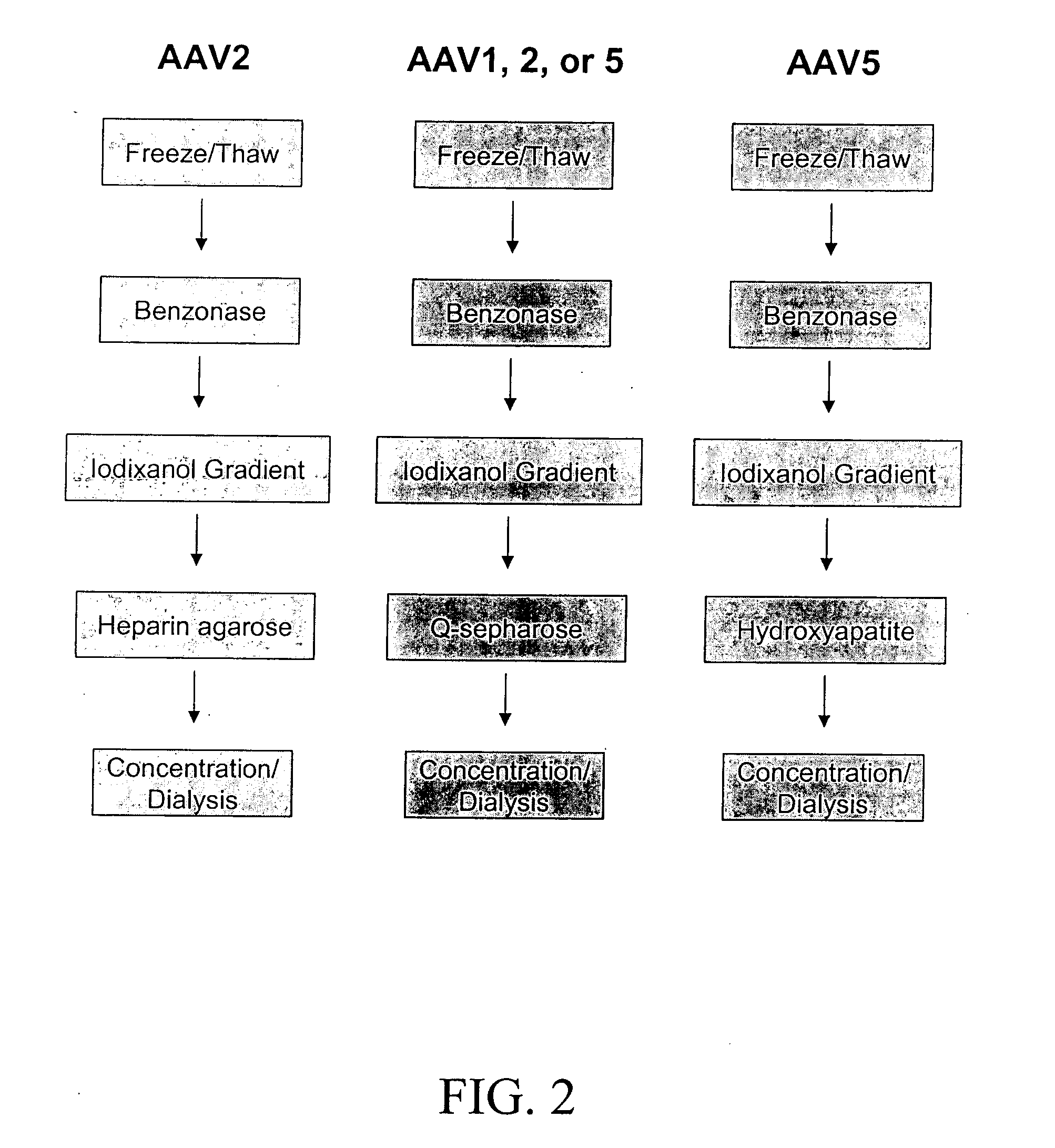
[0076] Because AAV1 and AAV5 both lack significant binding to the heparin affinity resin used to purify rAAV2 virions, purification protocols were developed that use density gradient centrifugation followed by anion exchange or hydroxyapatite chromatography.
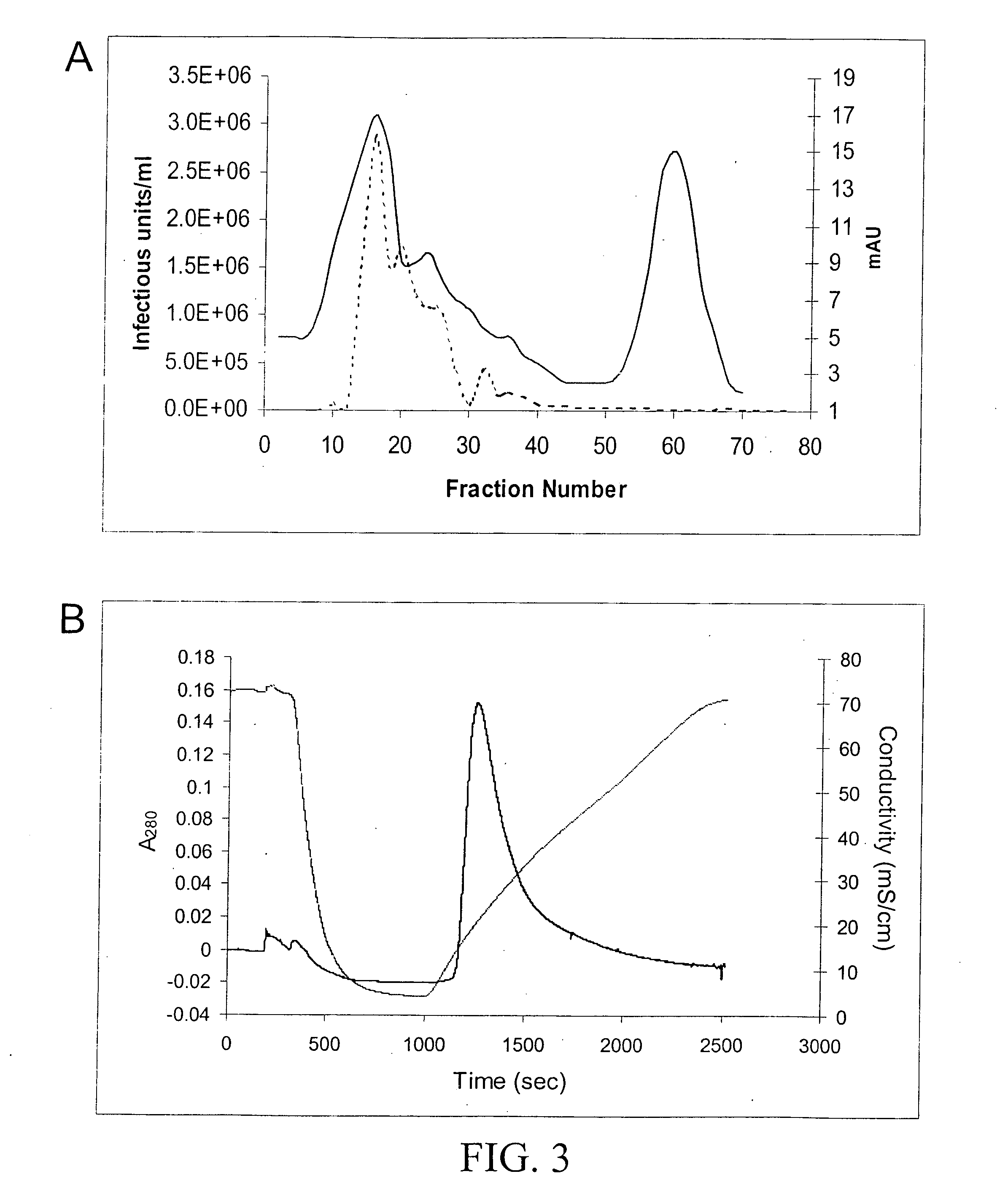
[0077] Following density gradient centrifugation, rAAV virions were purified by column chromatography. Three column resins were used: heparin-agarose, Q-sepharose, and hydroxyapatite. AAV2 virions bound heparin-agarose (FIGS. 6A and B), AAV5 virions bound hydroxyapatite, and AAV1, 2, and 5 virions bound Q-Sepaharose (FIGS. 3A and 4). rAAV2 virions eluted from heparin with 0.35M NaCl and rAAV5 virions eluted from hydroxyapatite with 0.2 M phosphate. AAV1, 2, and 5 eluted from Q-Sepharose in 0.5 M NaCl. As shown in FIG. 6A, virions produced were 99% pure with the three capsid proteins at the proper ratio of ˜1:1:20 for VP1:VP2:VP3. A combination of iodixanol step gradient followed by either affi...
example 3
ICA For rAAV and rcAAV
[0079] The infectious titer of rAAV was determined by measuring the ability of the virus to infect C12 cells expressing AAV2 rep and cap ORFs, unpackage, and replicate (FIG. 5A). In this assay, rep-cap expressing C12 cells were infected with serial dilutions of rAAV. To score the infecting viral particle it was amplified through viral DNA replication, whereupon the number of viral genomes reached several thousand per cell. This amplification was achieved by co-infecting the cell with a saturating amount of Ad5 to initiate rep and cap gene expression required for AAV DNA replication. The cells were then incubated for 40 hours, harvested, and transferred onto a nylon membrane and lysed. The immobilized viral DNA was hybridized with a transgene-specific probe and the cells infected with rAAV particles were scored as black dots following autoradiography (FIG. 5A).
[0080] WT AAV may contaminate vector preparations, and rcAAV may be formed during the production of r...
PUM
| Property | Measurement | Unit |
|---|---|---|
| pH | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More