Intradermal injection device
a technology of intradermal injection and injection tube, which is applied in the direction of intravenous device, infusion needle, infusion syringe, etc., can solve the problems of insufficient immune response, difficult to perform intradermal injection, and difficult to perform above-described technique, etc., and achieve good interface and limit leakage
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
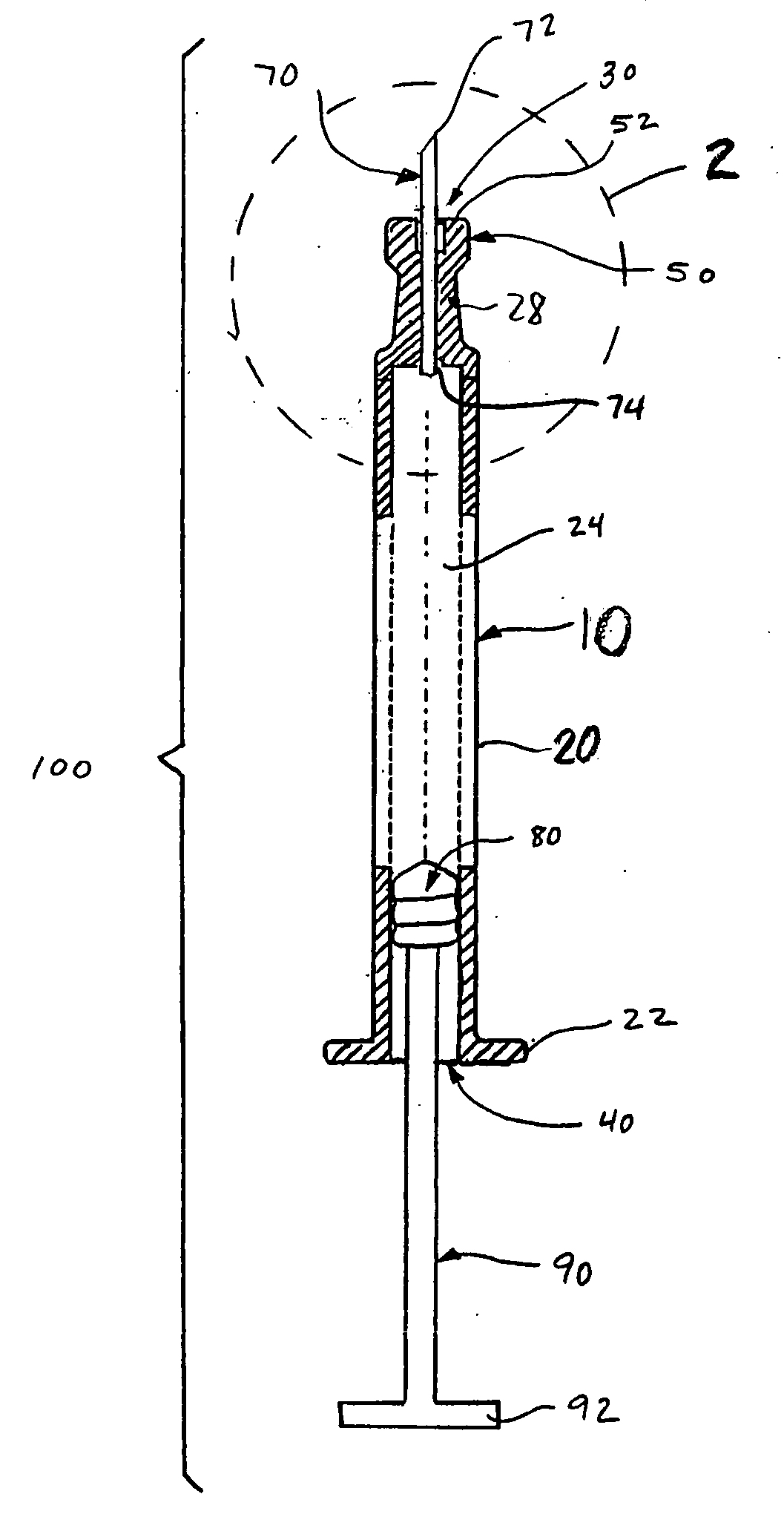
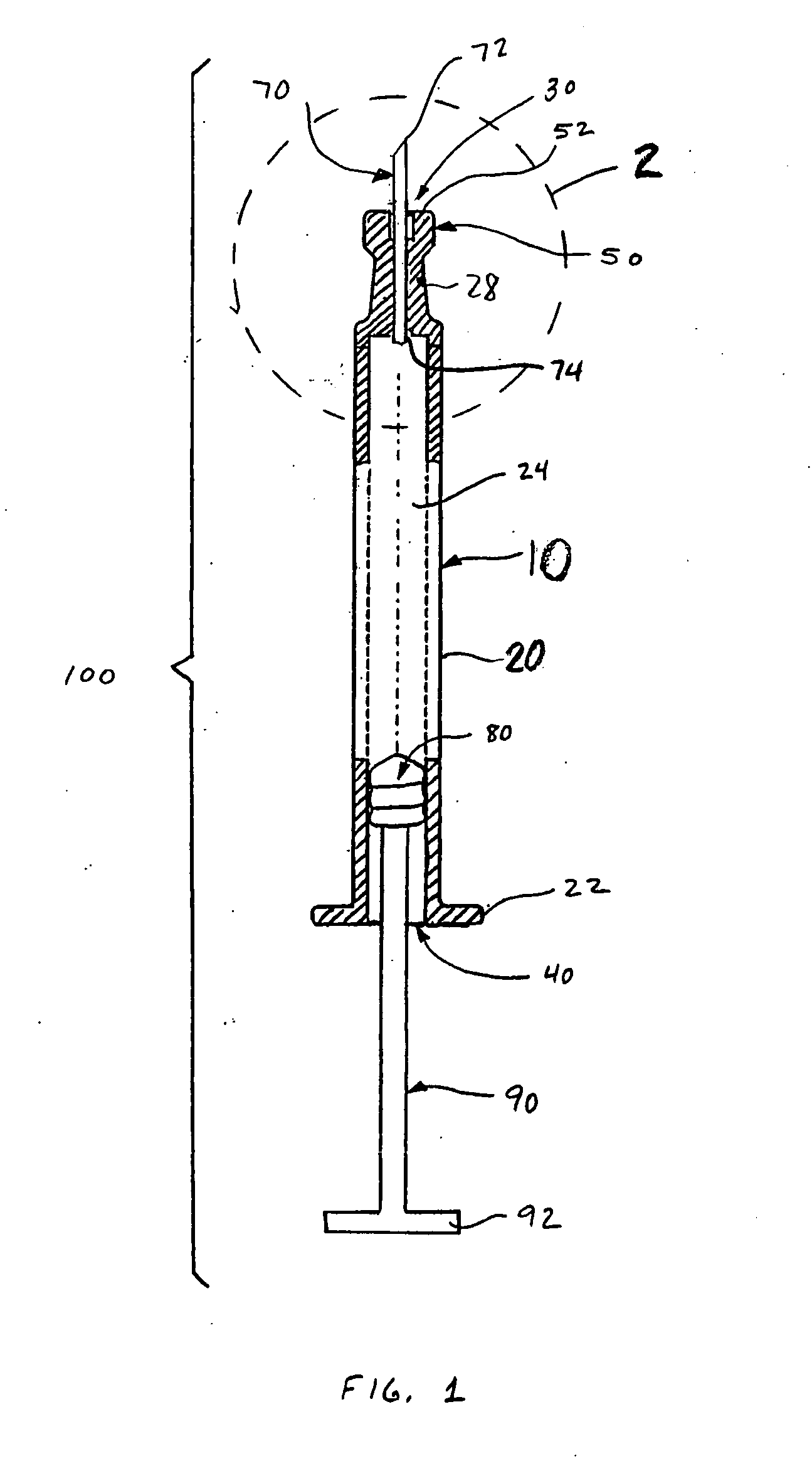
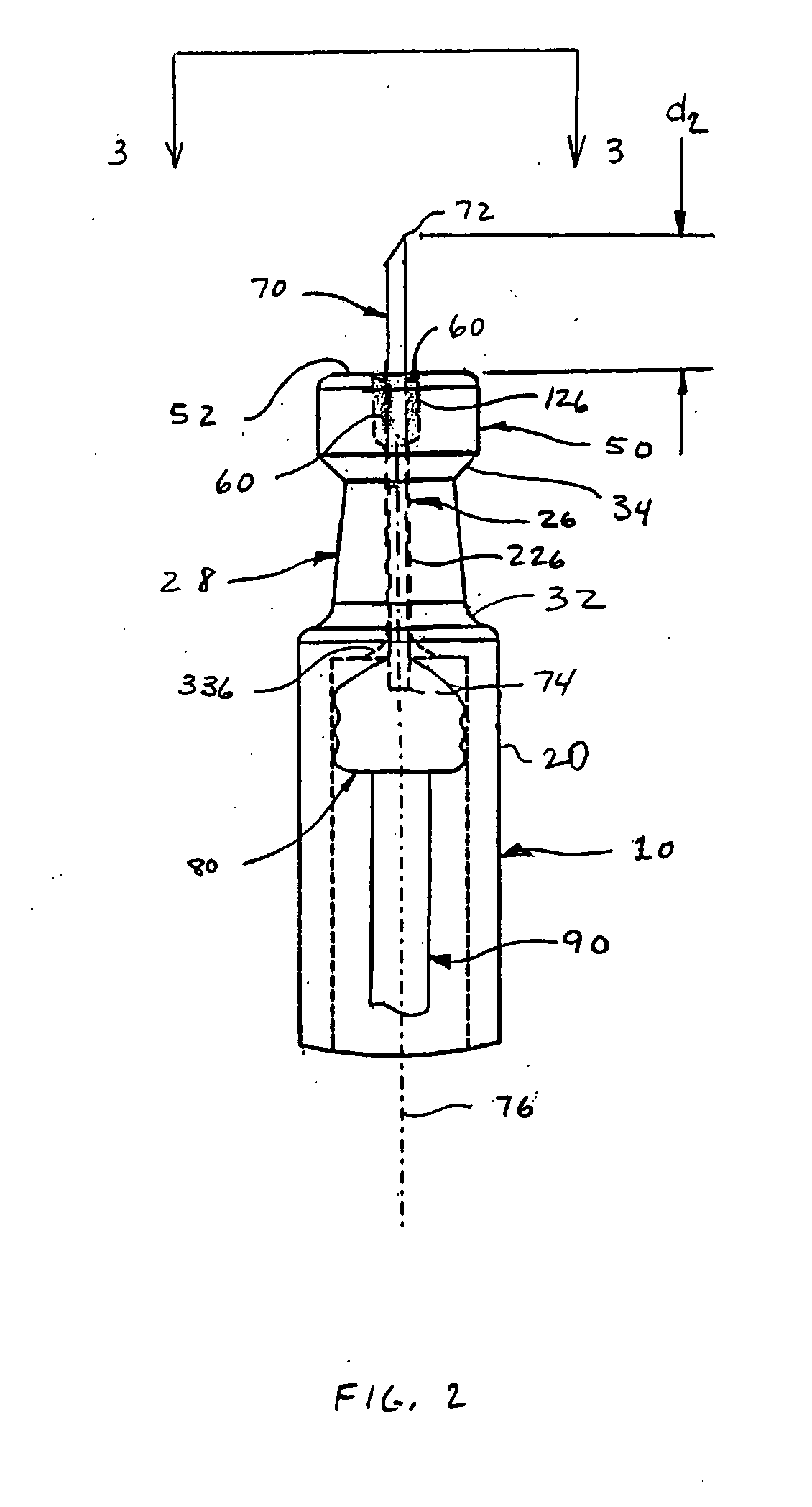
[0021]FIGS. 1-3 depict an intradermal injection device 100 constructed in accordance with an embodiment of the present invention. The injection device 100 is preferably a syringe comprised of a body 10 having a barrel 20, an open distal end 40 with a flange 22, a proximal end 30, and a reservoir 24 defined therebetween. A drug substance may be placed in the reservoir 24 before the injection device 100 is provided to the end user. The injection device 100 also includes a plunger 80 slidingly and sealingly provided within the reservoir 24, and a plunger rod 90 secured to the plunger 80 to facilitate movement of the plunger 80 within the reservoir 24 to effect expulsion of the drug substance therefrom.
[0022] The body 10 narrows near the proximal end 30 to form a neck 28 that supports a limiter 50 defined at the proximal end 30 of the body 10. The neck 28 is preferably tapered, particularly to converge in a distal to proximal direction. A first transition 32 may be provided to accommod...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More