Sustained release formulations of opioid and nonopioid analgesics
a non-opioid and formulation technology, applied in the field of monoeximic solid dosage forms, can solve the problems of inefficient and complicated manufacturing processes, and achieve the effect of improving the ability to treat pain and simple formulation or composition
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Preparation of Extended Release Monoeximic Dosage Forms Containing Acetaminophen (APAP) and Hydrocodone Bitartrate Hemipentahydrate (HBH) at Lab Scale
[0135] This example shows the preparation of single unit monoeximic matrix dosage forms.
[0136] Tablets weighing about 800 mg were prepared using wet granulation using a laboratory high shear mixer (Key International Inc.). APAP dense powder was dry mixed with HBH, and excipents except for APAP DC-90, silicon dioxide and magnesium stearate for 1.0 minute (150 rpm) followed by granulating with Eudragit L-30D aqueous dispersion at low speed (200 rpm) for approximately 2.0 minutes in the high shear mixer. The wet granules were then dried in an oven at 50° C. overnight and passed through a 20-mesh screen. The granulation was blended with APAP DC-90 and silicon dioxide for 3.0 minutes, and magnesium stearate for an additional 2.0 minutes at 25 rpm in a V-blender. The powder blend was subsequently compressed into tablets using a hydraulic C...
example 2
Preparation of Sustained Release Monoeximic Dosage Forms Containing APAP and HBH at Lab Scale
[0139] This following example shows the preparation of multi-unit monoeximic matrix dosage forms.
[0140] Tablets weighing 192-200 mg were each prepared using wet granulation or direct compression. For wet granulation, APAP dense powder was dry mixed with HBH, lactose and proportional amount of hydroxypropyl methylcellulose (HPMC) (listed in Table 3 below as Methocel K100LV and Methocel K4M) for 2-3 minutes (150 rpm) followed by granulation with an Eudragit L-30D aqueous dispersion at low speed (200 rpm) for approximately 2 minutes in a laboratory high shear mixer (Key International Inc.). The wet granules were then dried in an oven at 50° C. overnight and passed through a 20-mesh screen. The granulation was blended with other excipients including the remainder of HPMC, hydroxylpropyl cellulose (HPC) (listed in Table 3 below as Kluecel EXF), APAP DC-90 and silicon dioxide for 3 minutes and t...
example 3
Preparation of Extended Release Monoeximic Dosage Forms Containing APAP and HBH at Pilot Plant Scale
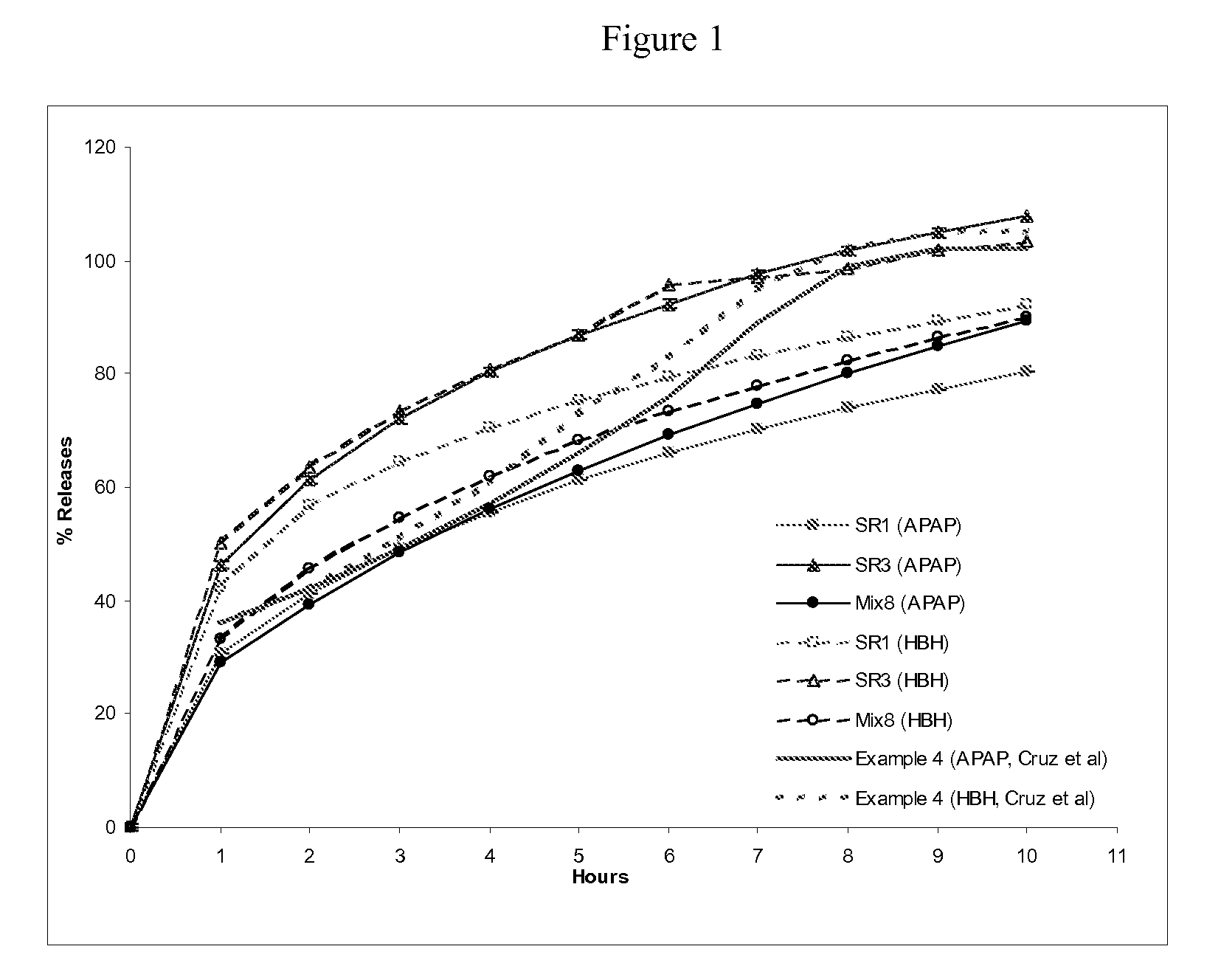
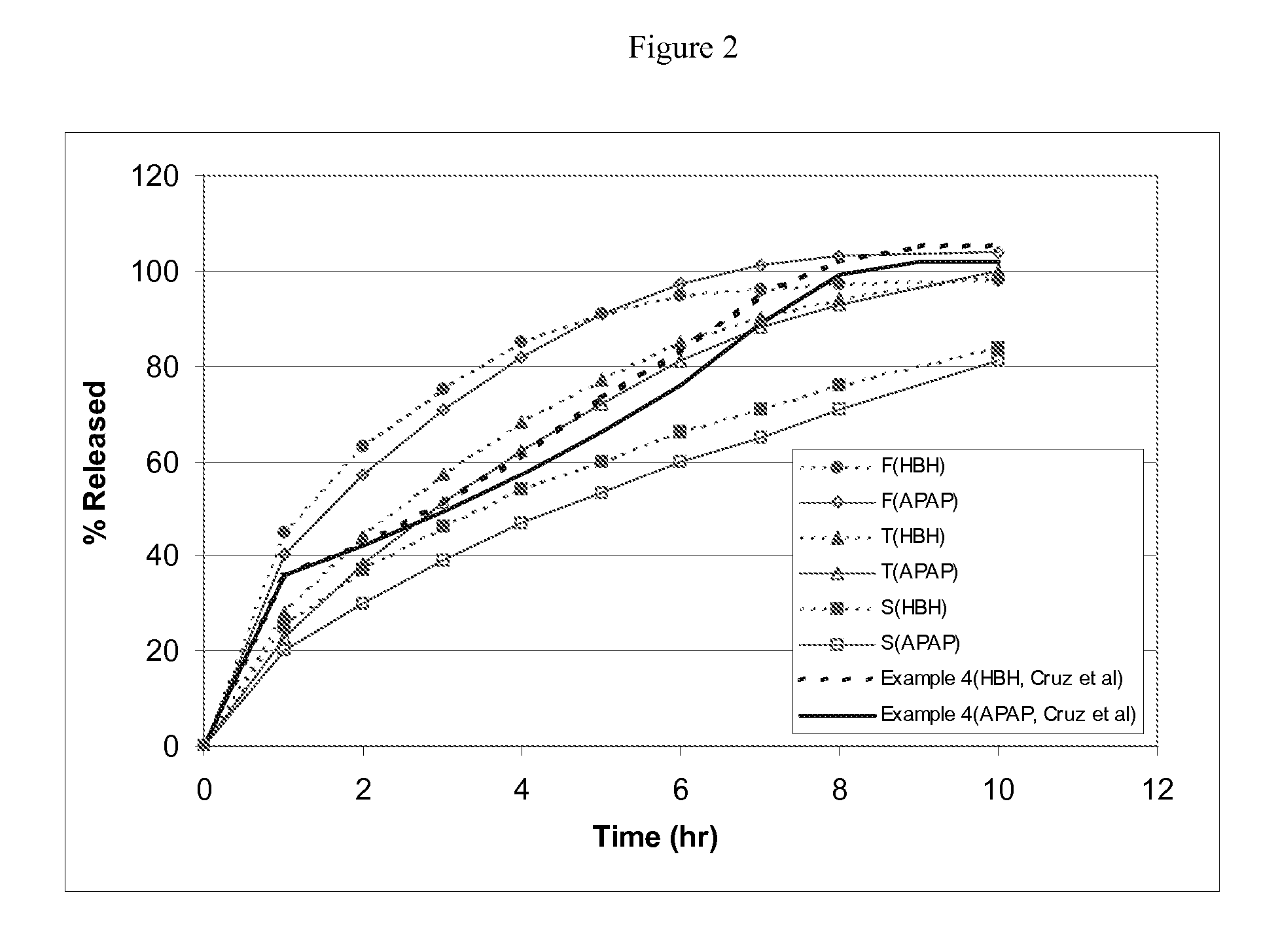
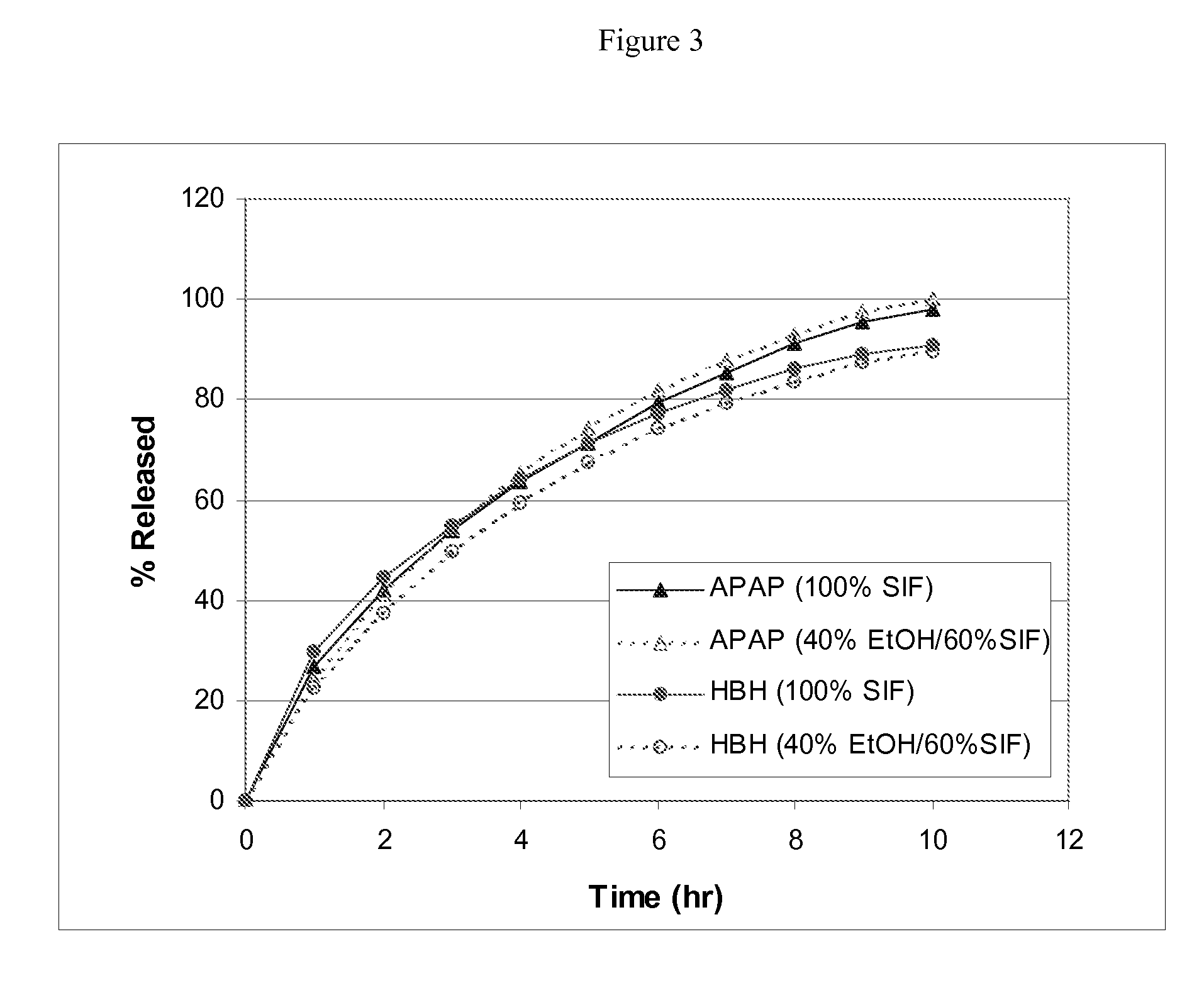
[0144] This example shows (1) multi-unit monoeximic matrix dosage forms designed for bioavailibility evaluations in humans; and (2) the resistance of the monoeximic matrix dosage forms to dose dumping from these dosage forms after the consumption of alcoholic beverages.
[0145] The dosage form used in this Example to evaluate human biovailability and that used to assess the impact of alchoholic solutions on the dosage forms differed from each other by 1% Eudragit L-30D-55 by weight.
[0146] Three (3) formulations that were designed for the bioavailability study in healthy subjects are provided in Table 5 below. Tablets weighing about 192.5-200 mg each were prepared using wet granulation. APAP dense powder was dry mixed with HBH, lactose (for Formulation F only) and approximately 8% hydroxypropyl methylcellulose (HPMC, Methocel K100 LV for Formulations F and T, Methocel K4M for Formulat...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More