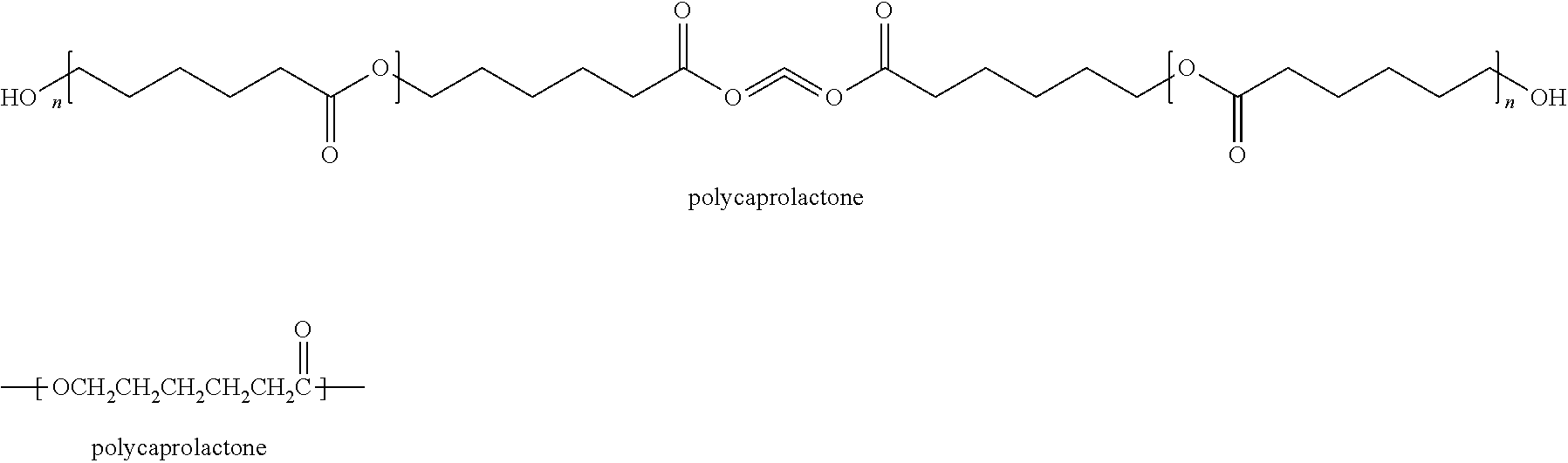
Implantable liposome embedded matrix composition, uses thereof, and polycaprolactone particles as scaffolds for tissue regeneration
a technology of embedded matrix composition and polycaprolactone, which is applied in the direction of phosphorous compound active ingredients, prosthesis, immunological disorders, etc., can solve the problems of changing the concentration of compound in the matrix and affecting the bioactivity of the compound
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Localized Differentiation of Endogenous Monocytes into an Angiogenic Phenotype
[0201]a. Preparation of Liposomes Containing IL-4 and IL-10 and Liposomes Containing Adenosine.
Reagents
Dioleoyl Phosphatidyl Choline
Saline (0.9% NaCl)
[0202]Human recombinant IL-4 and IL-10 are used together to activate the angiogenic phenotype in monocytes.
[0203]Adenosine can be used alone or in combination with IL-4 and IL-10 to activate the angiogenic phenotype in monocytes.
Method of Producing Liposomes Containing IL-4 and IL-10 for Targeting of Monocytes
Under Aseptic Conditions:
[0204]1. Dry 0.5 μmole of dioleoyl phosphatidyl choline under nitrogen in a disposable glass tube.
2. Evacuate in dessicator under vacuum for 30 minutes.
3. Add saline to required volume and scrape the sides of the glass tube to dislodge the lipid.
4. Add IL-4 and IL-10 1 μg / μl of lipid used, and, optionally adenosine 200 μM.
5. Vortex for 30 seconds. Sonicate twice in a bath sonicator at 7° C. for 1 min. This makes multilamellar ves...
example 2
Presentation of Cancer Cell Antigens to Monocytes / Dendritic Cells in the Vicinity of a Tumor
[0208]a. Preparation of Patient Specific Tumor Antigens
[0209]Tumor cells are taken by biopsy from a patient. The cells are lysed mechanically or by using a surface active material such as triton-X100. If surface antigens from the patient's cancer cells are required, cells can be incubated in a hypotonic buffer such as distilled water to remove cell content, and protein can then be extracted from the membranes. Protein extract from the cells is filtered to remove surface active materials and resuspended in saline solution. The protein extract from the cells is used for loading into liposomes. Proteins are loaded inside the liposomes to be phagocytosed by monocytes infiltrating the implanted matrix.
b. Preparation of the Liposomes Containing Tumor Antigens
Reagents
Dioleoyl Phosphatidyl Choline
Saline (0.9% NaCl)
Method
Under Aseptic Conditions:
[0210]1. Dry 0.5 μmole of dioleoyl phosphatidyl choline ...
example 3
Localized Reprogramming of Endogenous Monocytes or Stem Cells into Functional Insulin-Producing Beta Cells in the Pancreas
[0214]a. Preparation of Liposomes Containing Expression Vectors that Encode Transcription Factors
Reagents
Dioleoyl Phosphatidyl Choline
Saline (0.9% NaCl)
Method
Under Aseptic Conditions:
[0215]1. Dry 0.5 μmole of dioleoyl phosphatidyl choline under nitrogen in a disposable glass tube.
2. Evacuate in dessicator under vacuum for 30 minutes.
3. Add saline to required volume and scrape the sides of the glass tube to dislodge the lipid.
4. Add expression vectors that encode Ngn3, Pdx1 and Mafa, 0.1 μg / μl of lipid used.
5. Vortex for 30 seconds. Sonicate twice in a bath sonicator at 7° C. for 30 sec. This makes multilamellar vesicles that become small unilamellar vesicles (SUV) with prolonged sonication time. To make large unilamellar vesicles, an extruder can be used.
Another Method to Produce Liposomes Containing Transcription Factors for Targeting Monocytes:
[0216]Expression ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Time | aaaaa | aaaaa |
| Electrical resistance | aaaaa | aaaaa |
| Biological properties | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More