Use of opioid antagonists to attenuate endothelial cell proliferation and migration
a technology of endothelial cells and opioid antagonists, which is applied in the field of attenuating the migration and/or proliferation of endothelial cells, can solve the problems of patients refusing opioid-based pain management, morphine and other opioid medications can have severe side effects, and complete necrosis of the neoplasm, so as to improve the antiproliferative effect of cancerous cells, increase the therapeutic effect, and improve the antiproliferative
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
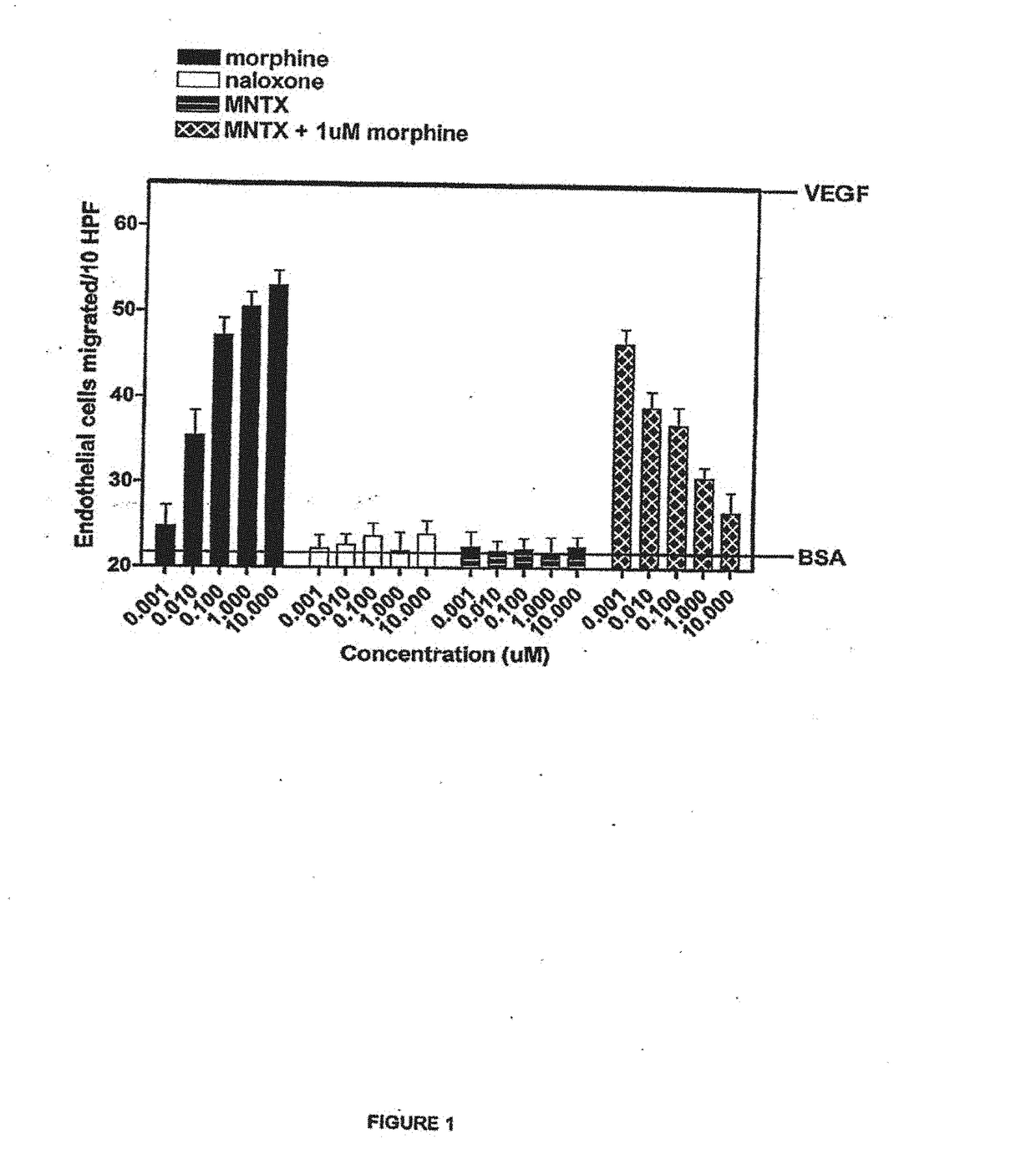
[0169]The antiangiogenic activity of the peripheral opioid antagonists in accordance with the present invention was evaluated in experiments testing the ability of the antagonist to inhibit or modulation capillary endothelial cell migration using a modified Boyden chamber.
[0170]The endothelial cell migration assay was performed as described by Lingen, M. W., Methods in Molecular Medicine, 78: 337-347 (2003), the disclosure of which is incorporated by reference. Briefly, Human Microvascular Endothelial Cells (HMVEC) (Cell Systems, Kirkland, W A.) were starved overnight in Endothelial Growth Medium (EGM) containing 0.1% bovine serum albumin (BSA). Cells were then trypsinized and resuspended in Dulbecco's Modified Eagle Medium (DME) with 0.1% BSA at a concentration of 1×106 cells / mL. Cells were added to the bottom of a 48-well modified Boyden chamber (NeuroPore Corporation, Pleasanton, Calif.). The chamber was assembled and inverted, and cells were allowed to att...
example 2
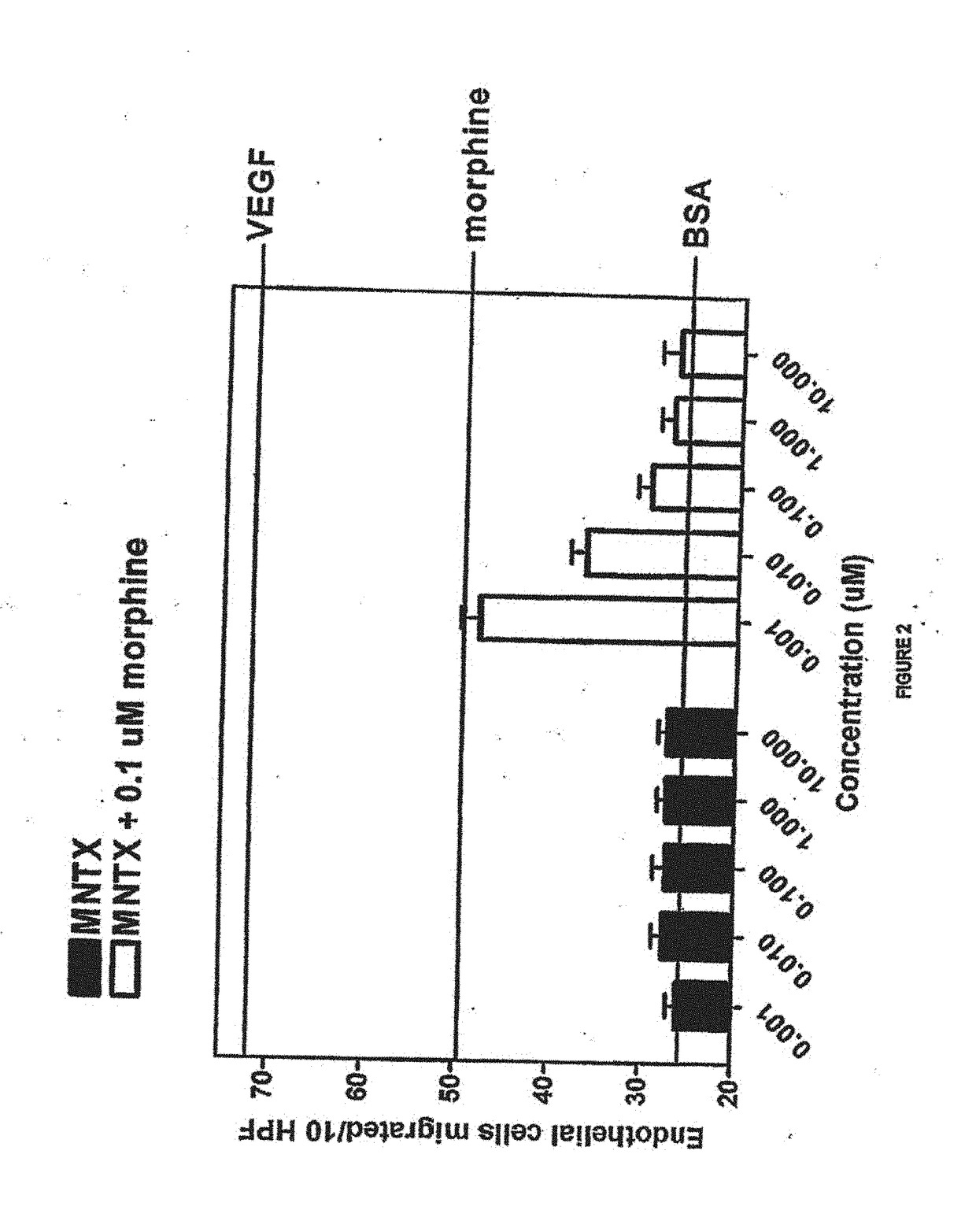
[0172]Another set of experiments was conducted in accordance with the procedure described in Example 1. In this set of experiments, methylnaltrexone and the combination of methylnaltrexone and morphine was again tested for ability to inhibit endothelial cell migration. The methylnaltrexone concentrations when tested alone varied from 0.001 to 10.0 μM. In combination, the concentrations of methylnaltrexone varied from 0.001 to 10.0 μM, while the morphine concentration remained constant at 0.1 μM as described in Example 1. The results are shown in FIG. 2.
[0173]FIG. 2 shows the combination of methylnaltrexone and morphine decreased migration in a concentration-dependent manner, while methylnaltrexone alone did not affect migration.
example 3
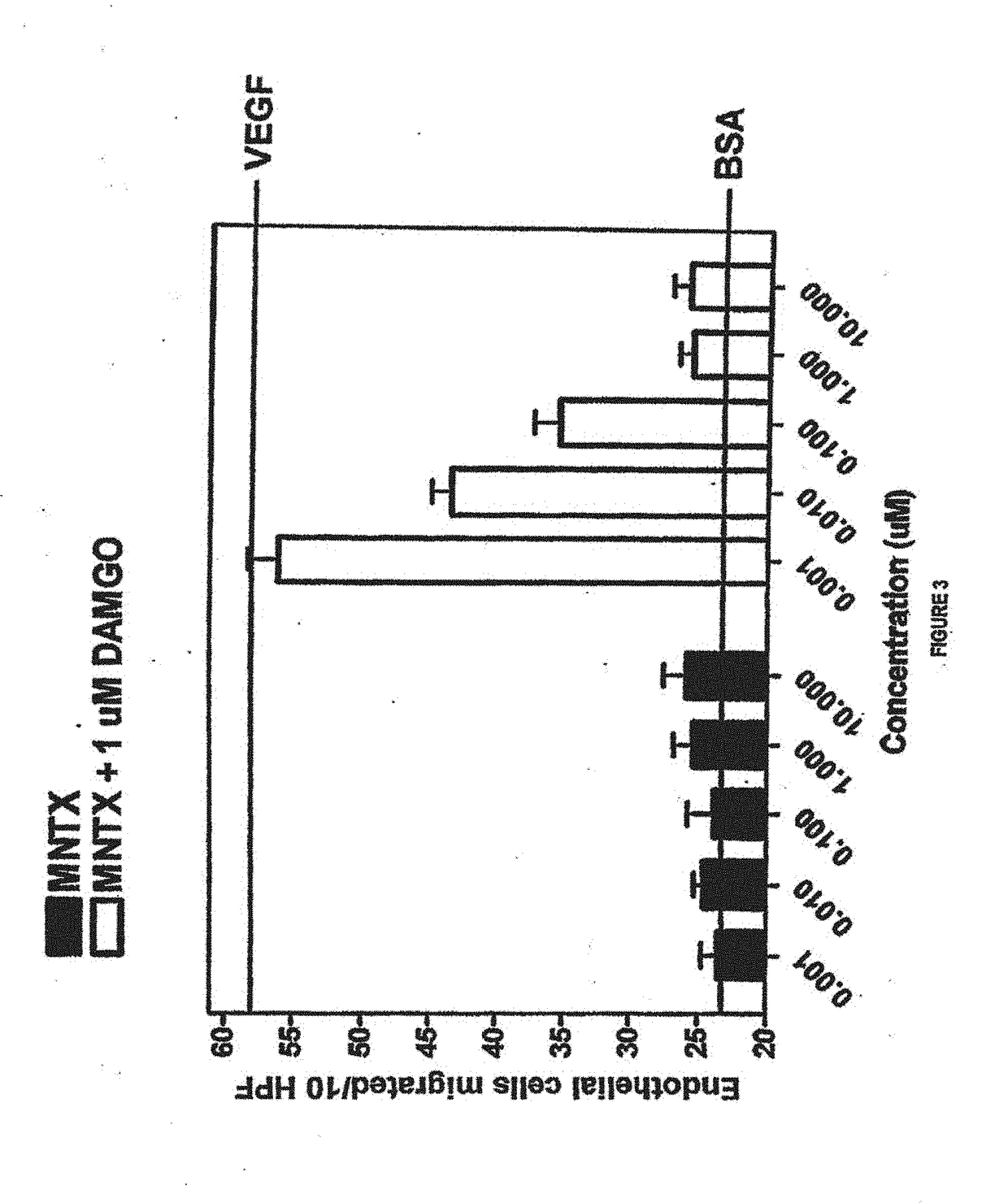
al Cell Migration Induced by DAMGO
[0174]The drugs used in this study were [D-Ala 2, N-McPhe4, Gly5-ol] enkephalin or DAMGO (Sigma, St. Louis, Mo.); naloxone (Sigma, St. Louis, Mo.); N-methylnaltrexone bromide or methylnaltrexone (Mallinckrodt Specialty Chemicals, Phillipsburg, N.J.). The endothelial cell migration assay was performed as previously described (9). Human dermal microvascular endothelial cells (Cell Systems, Kirkland, W A) were starved overnight in media containing 0.1% bovine serum albumin (BSA), harvested, resuspended into Dulbecco's Modified Eagle's media (DME) with 0.1% BSA, and plated on a semi-porous gelatinized membrane in a modified Boyden chamber (Nucleopore Corporation, Pleasanton, Calif.). Test substances were then added to the wells of the upper chamber and cells were allowed to migrate for four hours at 37° C.
[0175]Membranes were recovered, fixed, and stained and the number of cells that had migrated to the upper chamber per ten high power fields counted by...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com