Mushroom polysaccharide chitosan nanoparticle and preparation method thereof
A technology of chitosan nanoparticles and lentinan, which is applied in the direction of pharmaceutical formulations, medical preparations with non-active ingredients, medical preparations containing active ingredients, etc., can solve the problems of difficult preparation and unfavorable encapsulation, and achieve encapsulation High efficiency and high stability
Inactive Publication Date: 2010-06-16
ZHEJIANG UNIV
View PDF1 Cites 20 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
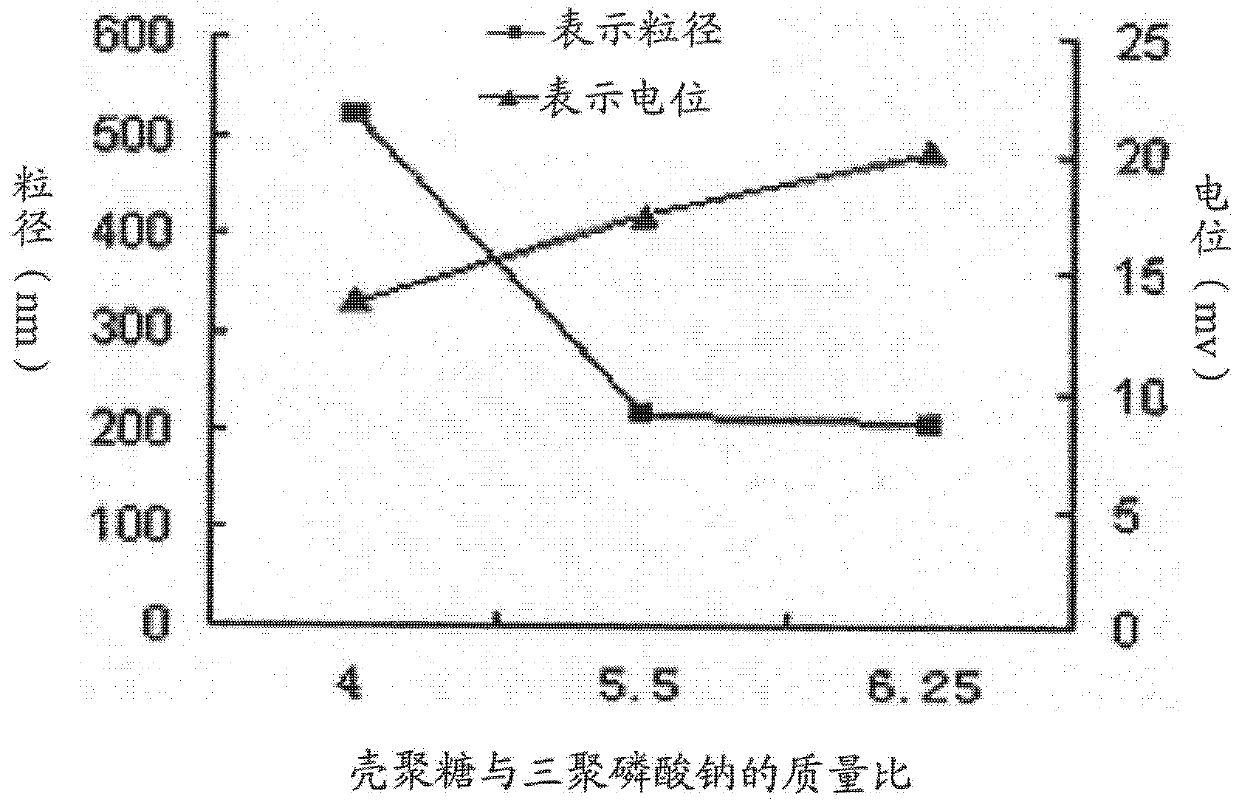
The invention provides a nano-particle of lentinan and chitosan, which is used to make up the blank of the existing nano-preparation of lentinan, which adopts chitosan and sodium tripolyphosphate as carrier materials, and carries the lentinan by ion gelation Successfully prepared nanoparticles with high stability and good encapsulation efficiency, which overcomes the difficulty of preparing nano-preparations due to its water solubility, macromolecules and other properties that are not conducive to encapsulation
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
Embodiment 2
Embodiment 3
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
| Property | Measurement | Unit |
|---|---|---|
| Particle size | aaaaa | aaaaa |
| Concentration | aaaaa | aaaaa |
| Concentration | aaaaa | aaaaa |
Login to View More
Abstract
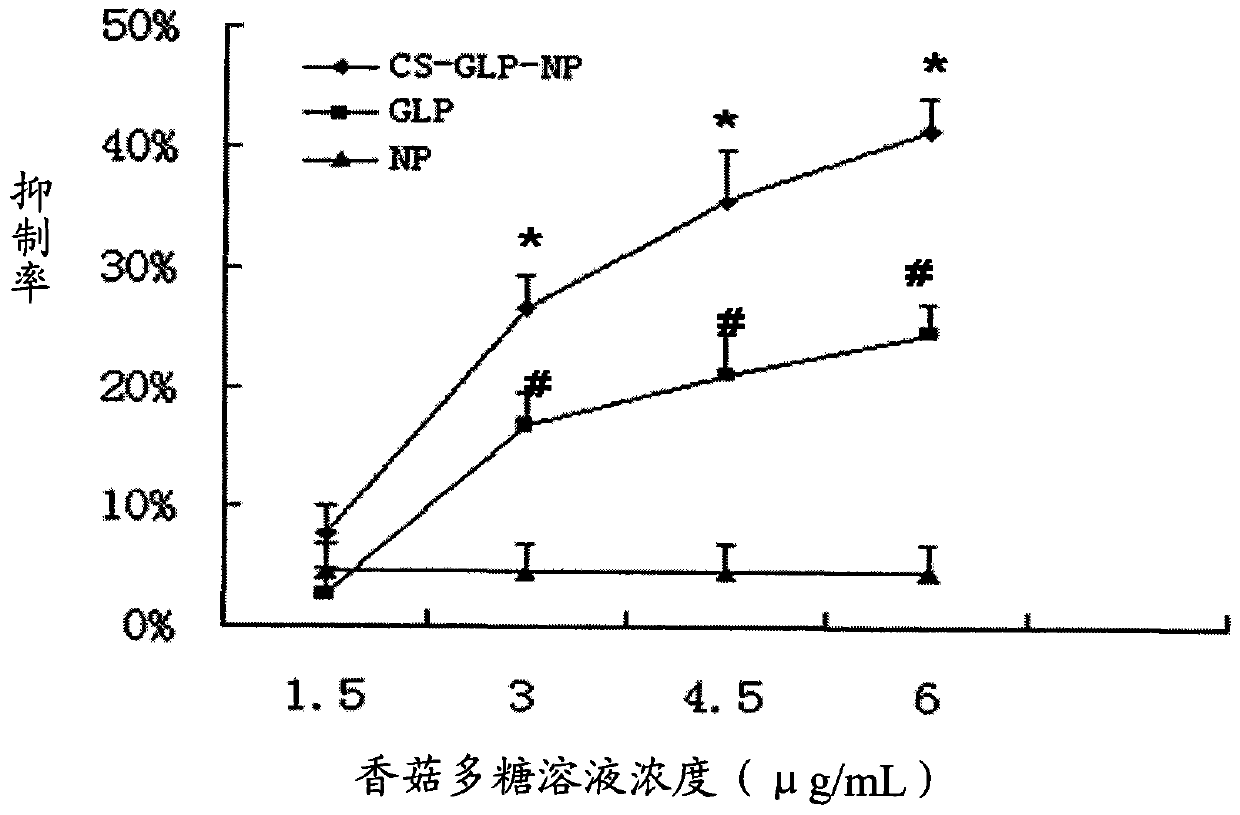
The invention discloses a mushroom polysaccharide chitosan nanoparticle and a preparation method thereof, and the mushroom polysaccharide chitosan nanoparticle is prepared by the following parts by weight of pharmaceutical raw materials and carrier material: 150-250 parts of chitosan, 30-70 parts of sodium tripolyphosphate and 0.6-1.5 parts of mushroom polysaccharide. The chitosan is adopted as a carrier, a drug-loaded nanoparticle is self-formed through the electrostatic action of anions and cations of the chitosan and the sodium tripolyphosphate, and the mushroom polysaccharide chitosan nanoparticle with high stability, good encapsulation efficiency and high drug loading capacity is successfully prepared by combining the ion gelatinization method with the double emulsion-solvent evaporation method for loading water soluble and macromolecular mushroom polysaccharide. The mushroom polysaccharide chitosan nanoparticle has good anti-tumor effect and immune enhancement effect; compared with mushroom polysaccharide solution, the mushroom polysaccharide chitosan nanoparticle has stronger anti-tumor effect and higher stability. The preparation method has the advantages of simple operation, safety and effectiveness and is in line with the environmental protection requirement and applicable to industrial production.
Description
technical field The invention relates to the field of pharmaceutical nano preparations, in particular to a lentinan chitosan nanoparticle and a preparation method thereof. Background technique Polysaccharides are formed by the condensation and dehydration of multiple monosaccharide molecules. There are various types and complex structures, and they are widely distributed in animals and plants. At present, there are many studies on the physical and chemical properties and biological functions of polysaccharides. In recent years, some researchers have carried out further research and development on the potential functions of polysaccharides from different sources, which has attracted wide attention in the application of polysaccharides in the field of medicine. At present, an important application field of polysaccharides is anti-tumor. Through research, it is found that the anti-tumor effects of polysaccharides are as follows: on the one hand, they exert anti-tumor effects b...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): A61K9/14A61K31/715A61K47/36A61P1/16A61P31/20A61P35/00
Inventor 高建青李旎
Owner ZHEJIANG UNIV
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com