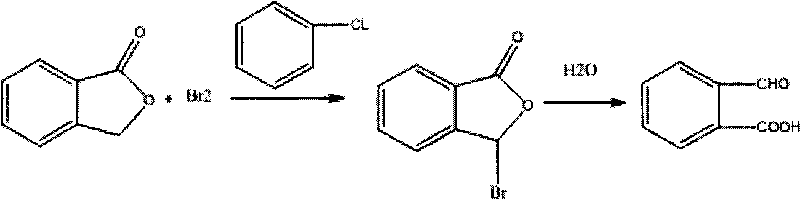
Preparation method of 2-carboxybenzaldehyde
A technology of o-carboxybenzaldehyde and toluene, applied in the preparation of carboxylate/lactone, organic chemistry, etc., can solve the problems of high production cost, low purity, and low yield, and achieve low cost, high purity, and high yield. high rate effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0013] Embodiment 1: the preparation method of a kind of o-carboxybenzaldehyde of the present invention is that phthalide takes chlorobenzene as solvent, first reacts with bromine to prepare 3-bromophthalide, then adds water to initially synthesize o-carboxybenzaldehyde wet product, and finally Add water and refine to obtain high-quality o-carboxybenzaldehyde, specifically comprising the following steps: (1) preparing 3-bromophthalide: adding chlorobenzene 200kg and phthalide 225kg in a 500L reactor equipped with a reflux distillation condenser, heating and dissolving; Put 270kg of bromine into the high-level tank, raise the temperature to about 100-110°C, start to add bromine slowly, and control the speed just so that the escaping hydrogen bromide gas does not have red color, and the hydrogen bromide gas is absorbed by the absorption device as Hydrogen bromide; after bromine is added dropwise, heat up to 110-120°C and keep reflux reaction for 2 hours, then use nitrogen gas to ...
Embodiment 2
[0014] Embodiment 2: the preparation method of a kind of o-carboxybenzaldehyde of the present invention is that phthalide takes chlorobenzene as solvent, first reacts with bromine to prepare 3-bromophthalide, then adds water to initially synthesize o-carboxybenzaldehyde wet product, and finally Add water and refine to obtain fine-quality o-carboxybenzaldehyde; the mol ratio of phthalide, bromine, chlorobenzene, and water is 1:1:0.5:1, and the specific steps are as follows: (1) prepare 3-bromophthalide: ① in the reactor Add chlorobenzene, then add phthalide, heat to 100°C, then slowly add bromine dropwise; ②After the dropwise addition of bromine is completed, raise the temperature to 105°C, and keep the reflux reaction for 1.5 hours; ③Introduce nitrogen into the reaction kettle , and properly remove hydrogen bromide to make the reaction complete; ④ Cooling, the cooling temperature is 15 ° C, crystallization is precipitated, and suction filtration and centrifugation obtain 3-brom...
Embodiment 3
[0015] Embodiment 3: the preparation method of a kind of o-carboxybenzaldehyde of the present invention is that phthalide takes toluene as solvent, first reacts with bromine to prepare 3-bromophthalide, then adds water to initially synthesize o-carboxybenzaldehyde wet product, and finally Add water and refine to obtain fine-quality o-carboxybenzaldehyde, and the molar ratio of phthalide, bromine, toluene, and water is 1:3:2:20. The specific steps are as follows: (1) Preparation of 3-bromophthalide: 1. Add Add toluene, then add phthalide, heat to 110°C, and then slowly add bromine dropwise; ②After the dropwise addition of bromine is completed, raise the temperature to 120°C, and keep the reflux reaction for 2.5 hours; ③Blow nitrogen into the reaction kettle, and properly Remove hydrogen bromide to make the reaction complete; ④ cool to 20°C, precipitate crystals, filter and centrifuge to obtain 3-bromophthalide; (2) Preliminary synthesis of o-carboxybenzaldehyde wet product: add ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More