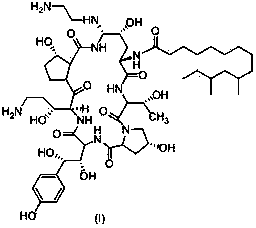
Azacyclohexapeptide preparation method
A nitrogen heterocycle and compound technology, applied in the field of salt preparation, can solve problems such as difficulties in industrialized production of caspofungin
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0031] The preparation of the compound shown in embodiment 1 formula III
[0032] .
[0033] The compound represented by formula II (100 g) was added to dry tetrahydrofuran (3 L), refluxed through 3A molecular sieves, dried until the water content was less than 10 mol%, added tetrahydrofuran to the original volume, and cooled in an ice bath.
[0034] Borane dimethyl sulfide complex (68.6 g) was slowly added dropwise, and the temperature was maintained at 0°C. After the dropwise addition, the reaction was completed under ice bath conditions (about 4 hours). The mixture was cooled in an ice bath to below 0°C, 2N hydrochloric acid solution (200ml) was slowly added dropwise, and stirred for 2 hours. Then separated and purified by reverse phase chromatography (C18) column (1:4 acetonitrile / water elution), collected appropriate fractions, and lyophilized to obtain the hydrochloride of the compound of formula III, 64.6g, yield 75%.
Embodiment 2
[0035] The preparation of the compound shown in embodiment 2 formula V-1 and caspofungin
[0036] .
[0037] (a) Under the protection of nitrogen, cool acetonitrile (600mL) to -5°C and maintain the solvent temperature, add the compound of formula III (10g) and p-methylthiophenol (3.2g, added in 3 times equivalent), and maintain the temperature of the reaction solution Not exceeding -10°C, add trifluoroacetic acid (48g, 32.7mL), react at -15°C to 0°C until the reaction is complete (about 2.5 hours), slowly add ice water solution (500ml), a large amount of precipitation occurs, filter , recrystallized from methanol, and dried to obtain 11.2 g of the product (trifluoroacetate salt of the compound of formula V), with a yield of 92.5%, an HPLC purity of 98%, and an ee value >98%. MS (ESI) 1156.4 (M+H + ).
[0038] (b) At room temperature, dissolve the trifluoroacetate salt of the compound of formula V-1 obtained in step (a) in 100 mL of methanol, slowly add 1,2-ethylenediamine...
Embodiment 3
[0039] The compound shown in embodiment 3 formula V-2 and the preparation of caspofungin
[0040] .
[0041] (a) Under the protection of nitrogen, cool acetonitrile (600mL) to -5°C, maintain the solvent temperature, add the compound of formula III (10g) and o-methylthiophenol (3.2g, added in 3 times equivalent), and maintain the temperature of the reaction solution Not exceeding -10°C, add trifluoroacetic acid (48g, 32.7mL), react at -15°C to 0°C until the reaction is complete (about 2.5 hours), slowly add ice water solution (500ml), a large amount of precipitation occurs, filter , recrystallized from methanol, and dried to obtain 11 g of the product (trifluoroacetate salt of the compound of formula V), with a yield of 91.2%, an HPLC purity of 97.6%, and an ee value >98%. MS (ESI) 1156.4 (M+H+ ).
[0042] (b) At room temperature, dissolve the trifluoroacetate salt of the compound of formula V-2 obtained in step (a) in 100 mL of methanol, slowly add 1,2-ethylenediamine (40 ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More