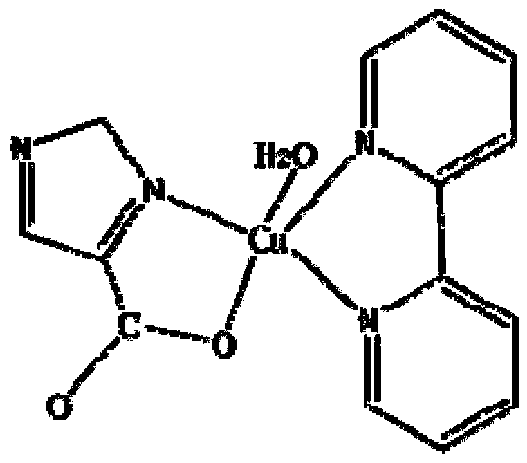
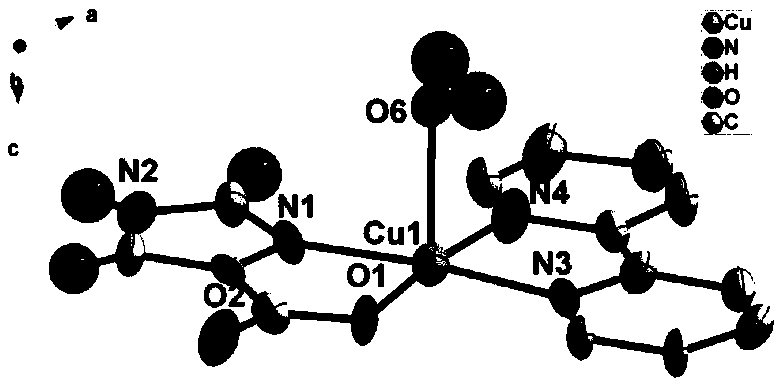
Imidazole-4-carboxylic acid-copper-2,2'-pyridine complex and its preparation method and application
A technology of complexes and copper compounds, applied in copper organic compounds, chemical instruments and methods, luminescent materials, etc., to achieve the effects of high yield, easy availability of raw materials, and high purity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0028] Embodiment 1 Preparation of imidazole-4-carboxylic acid-copper-2,2'-pyridine complex
[0029] in H 2 O (12ml) solvent, add Cu (NO 3 ) 2 ·3H 2 O (0.204g, 0.85mmol), 2,2'-pyridine (0.086g, 0.5mmol), imidazole 4,5-dioic acid (0.078g, 0.5mmol), NaOH (0.020g, 0.5mmol), placed in Stir on an electromagnetic stirrer for 30 minutes, place the reactant in a 25ml polytetrafluoroethylene-lined reactor, seal it and place it in a high-temperature oven, and after five days at a constant temperature of 160°C, drop it to room temperature at a rate of 5°C / h. Blue blocky crystals were precipitated with a yield of 60% (based on copper).
[0030] Elemental analysis value, calculated value: C43.95; H2.820; N12.06; experimental value: C44.07; H2.94; N12.13..
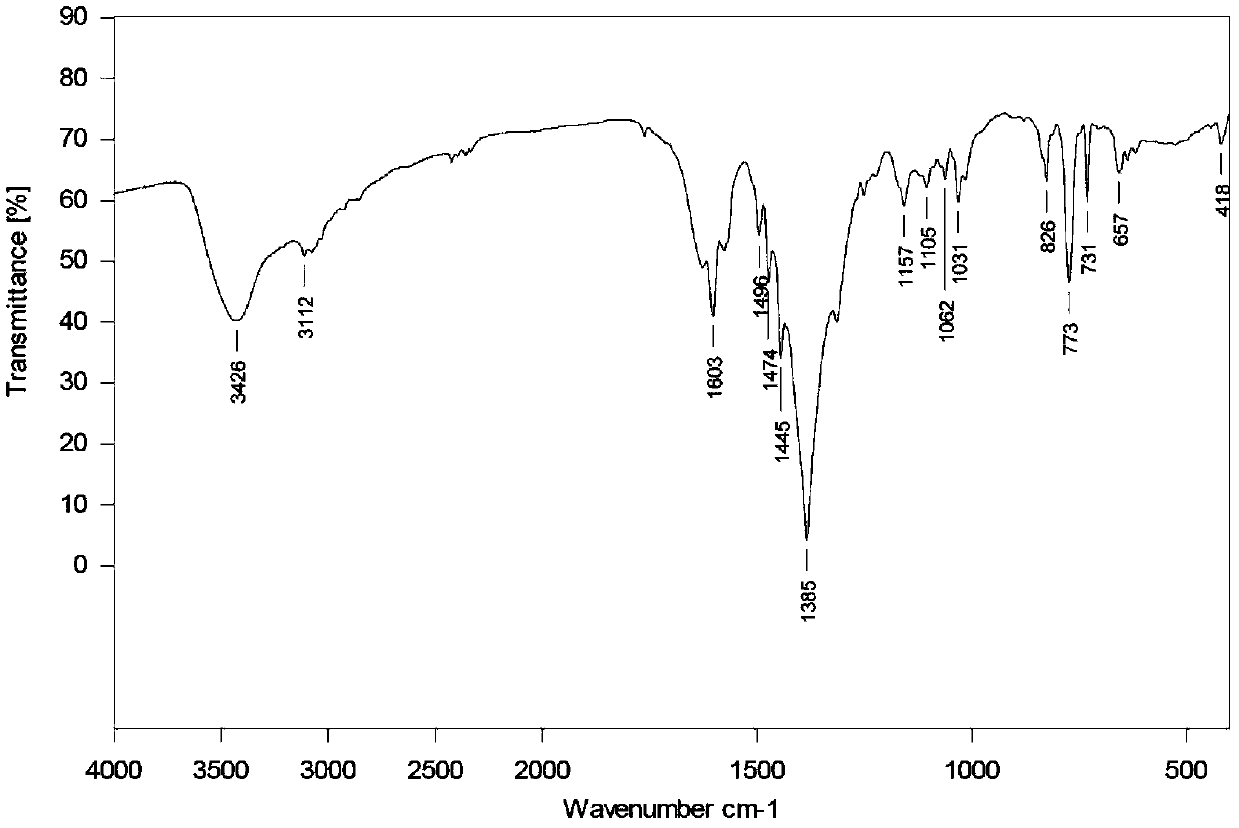
[0031] Infrared analysis, IR (KBr tablet) spectrum see figure 2 . 3426(s,b),1687(s),1663(s),1630(s),1518(m),1473(m),1426(m),1352(s),1335(s),1279(m ), 1087(m), 852(m), 790(m), 723(m), 657(m), 544(m).
[0032] Figure 1a Schemat...
Embodiment 2
[0036] Embodiment 2 Preparation of imidazole-4-carboxylic acid-copper-2,2'-pyridine complex
[0037] in H 2 In O (12ml) solvent, add Cu(SO 4 ) 2 ·5H 2 O (0.212g, 0.85mmol), 2,2'-pyridine (0.086g, 0.5mmol), imidazole 4,5-dioic acid (0.078g, 0.5mmol), NaOH (0.020g, 0.5mmol), placed in Stir on an electromagnetic stirrer for 30 minutes, place the reactant in a 25ml polytetrafluoroethylene-lined reactor, seal it and place it in a high-temperature oven, and after five days at a constant temperature of 160°C, drop it to room temperature at a rate of 5°C / h. Blue blocky crystals were precipitated with a yield of 60% (based on copper).
[0038] Elemental analysis value, calculated value: C43.95; H2.820; N12.06; experimental value: C44.07; H2.94; N12.13..
[0039] Infrared analysis, IR (KBr tablet) spectrum see figure 2. 3426(s,b),1687(s),1663(s),1630(s),1518(m),1473(m),1426(m),1352(s),1335(s),1279(m ), 1087(m), 852(m), 790(m), 723(m), 657(m), 544(m).
[0040] Figure 1a Schema...
Embodiment 3
[0042] Embodiment 3 Preparation of imidazole-4-carboxylic acid-copper-2,2'-pyridine complex
[0043] in H 2 O (12ml) solvent, add Cu (ClO 4 ) 2 ·6H 2 O (0.315g, 0.85mmol), 2,2'-pyridine (0.086g, 0.5mmol), imidazole 4,5-dioic acid (0.078g, 0.5mmol), NaOH (0.020g, 0.5mmol), placed in Stir on an electromagnetic stirrer for 30 minutes, place the reactant in a 25ml polytetrafluoroethylene-lined reactor, seal it and place it in a high-temperature oven, and after five days at a constant temperature of 160°C, drop it to room temperature at a rate of 5°C / h. Blue blocky crystals were precipitated with a yield of 60% (based on copper).
[0044] Elemental analysis value, calculated value: C43.95; H2.820; N12.06; experimental value: C44.07; H2.94; N12.13..
[0045] Infrared analysis, IR (KBr tablet) spectrum see figure 2 . 3426(s,b),1687(s),1663(s),1630(s),1518(m),1473(m),1426(m),1352(s),1335(s),1279(m ), 1087(m), 852(m), 790(m), 723(m), 657(m), 544(m).
[0046] Figure 1a Schema...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More