Nucleotide sequence for preventing infection of toxoplasmas and application of nucleotide sequence
A nucleotide sequence and infection prevention technology, applied in anti-infective drugs, medical preparations containing active ingredients, using vectors to introduce foreign genetic material, etc., can solve problems such as economic losses in animal husbandry
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
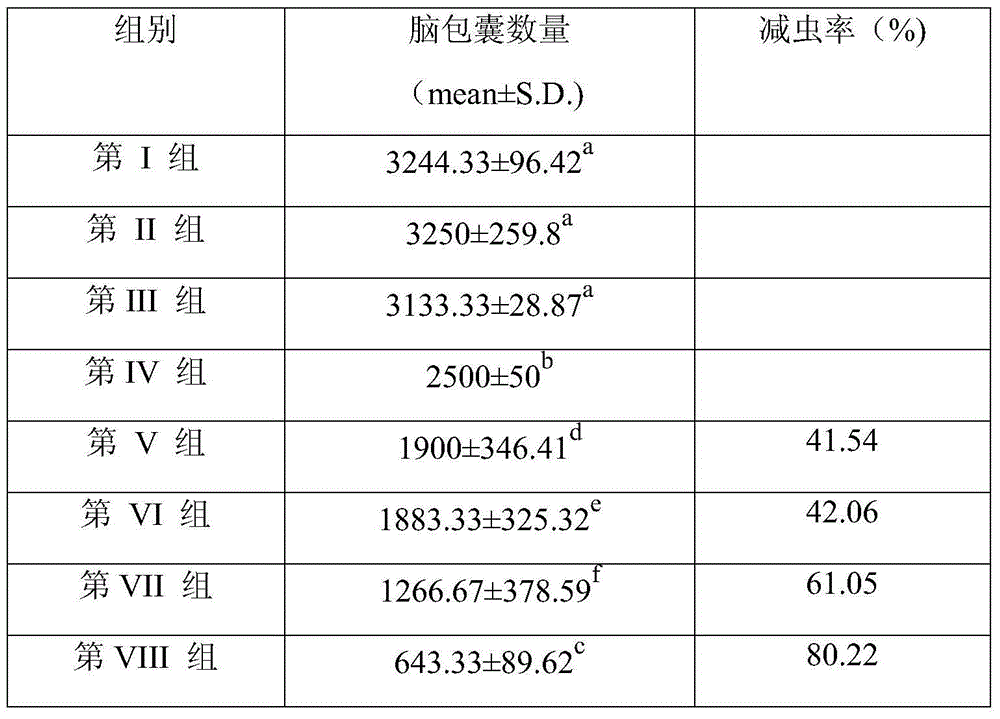
Embodiment 1
[0021] (1) Preparation of Toxoplasma gondii RH strain pVAX-profilin
[0022] 1. Take out the Toxoplasma gondii RH strain from liquid nitrogen, thaw in a water bath at 37°C, and inject Kunming mice into the abdominal cavity. Centrifuge at 6000rpm for 10min, discard the supernatant, and store the pellet in a -80°C refrigerator for RNA extraction. according to The operating instructions of Total RNA Kit I (purchased from Omega Company) were used to extract recombinant total RNA.
[0023] 2. Amplification of the Profilin gene:
[0024] 2.1 Primer design
[0025] Referring to the sequence of the profilin gene of Toxoplasma gondii, the primer sequences for amplifying the profilin gene of Toxoplasma gondii were designed, including:
[0026] Upstream: 5'-CGG GGTACC ATGTCCGACTGGGACCCTGTTGTCAAGG-3',
[0027] Downstream: 5'-CCG GAATTC TTAGTACCCAGACTGGTGAAGATACTCG-3',
[0028] Wherein, the underlined parts are Kpn I and EcoR I restriction sites respectively.
[0029] 2.2PCR amp...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More