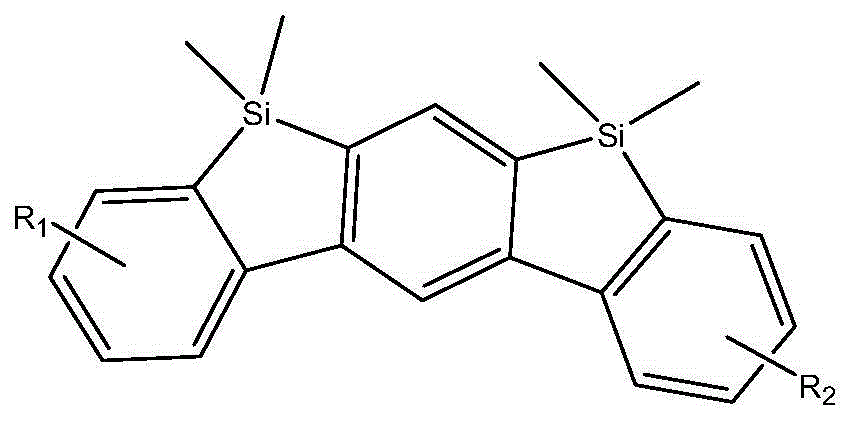
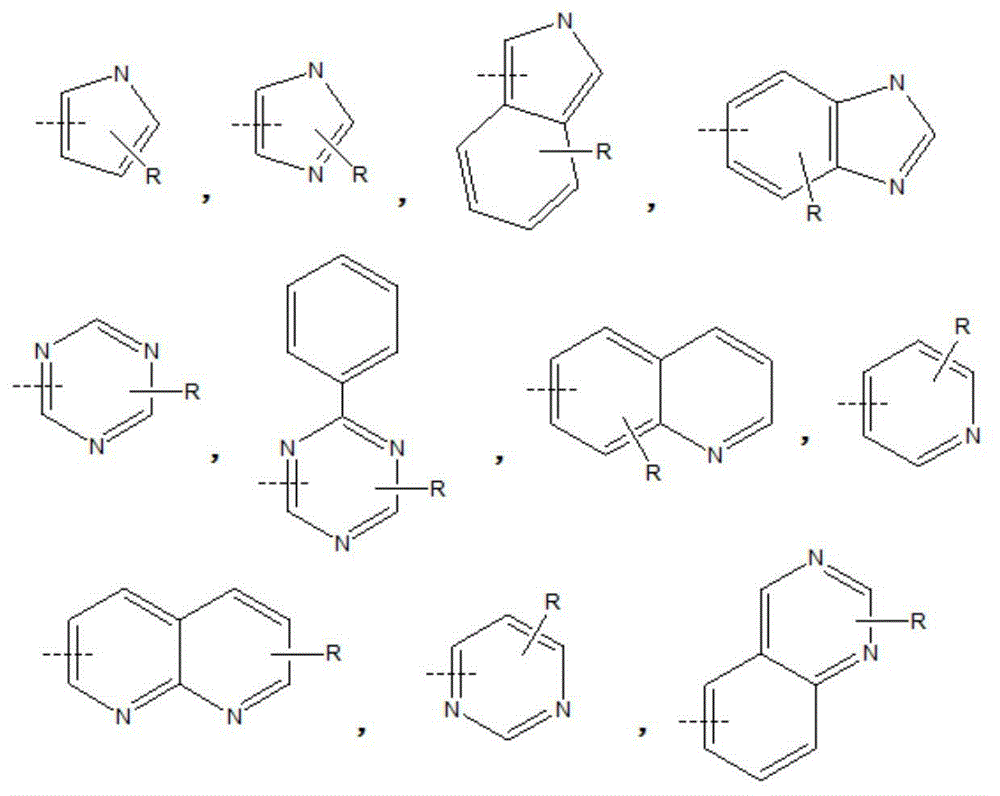
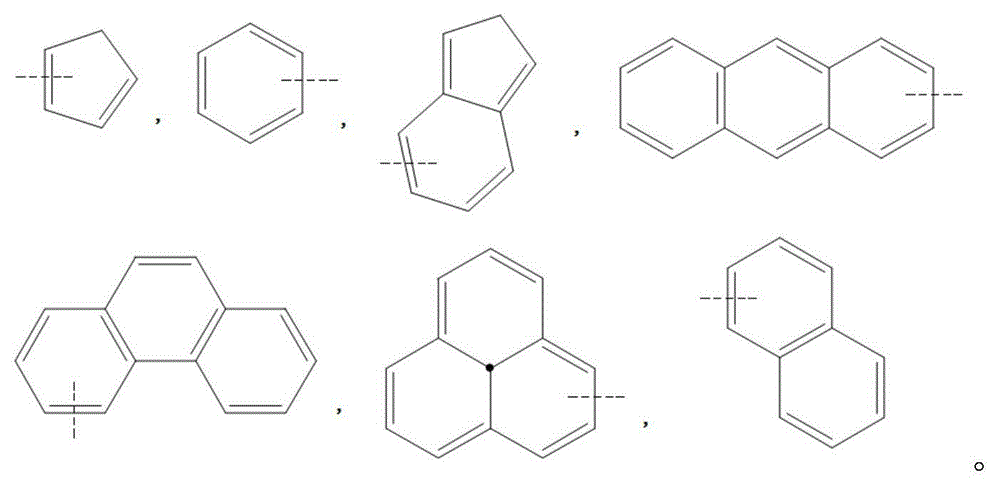
Compound, and material and organic light-emitting device including same
A compound and aromatic compound technology, applied in the field of compounds, can solve the problems of power consumption, narrow viewing angle, slow display screen response rate, etc., and achieve the effects of good color coordinates, low driving voltage and high efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0038]
[0039] Add 0.1 mol of reactant A, 0.1 mol of reactant B, and 1000 mL of solvent toluene into the reaction flask, heated to reflux for 24 hours under nitrogen protection, cooled, removed toluene, added with dichloromethane, washed with water, dried, the crude product was passed through the column, and then used Dichloromethane and ethanol were recrystallized and purified to obtain the product compound 1. The MS and H-NMR characterization data are as follows.
[0040] MS: m / e=701.24 (M+H + ).
[0041] H-NMR (400M, CDCl 3 ): 7.53(2H d), 7.62(4H t), 7.71(4H t), 8.58(1H s), 7.67(1H s), 7.76(1H s), 7.34(2H d), 8.32(2H d), 7.34 (4Ht), 7.29 (2Hs), 1.61 (12Ht).
Embodiment 2
[0043]
[0044] Add 0.1 mol of reactant C, 0.3 mol of reactant B, and 1000 mL of solvent toluene into the reaction flask, heated to reflux for 24 hours under nitrogen protection, cooled, removed toluene, added with dichloromethane, washed with water, dried, the crude product was passed through the column, and then used Dichloromethane and ethanol were recrystallized and purified to obtain the product compound 6. The MS and H-NMR characterization data are as follows.
[0045] MS: m / e=804.28 (M+H + ).
[0046] H-NMR (400M, CDCl 3 ): 7.53(4Ht), 7.62(8Ht), 7.71(8Ht), 8.34(2Hd), 7.61(2Hd), 7.64(4Ht), 1.61(12Ht).
Embodiment 3
[0048]
[0049] Add 0.1 mol of reactant D, 0.1 mol of reactant E, and 1000 mL of solvent toluene into the reaction flask, heated to reflux for 24 hours under nitrogen protection, cooled, removed toluene, added with dichloromethane, washed with water, dried, the crude product was passed through the column, and then Recrystallization and purification with dichloromethane and ethanol gave the product compound 5, and its MS and H-NMR characterization data are as follows.
[0050] MS: m / e=765.27 (M+H + ).
[0051] H-NMR (400M, CDCl 3 ): 7.53(2H d), 7.62(4H t), 7.71(4H t), 7.58(2H d), 7.77(2H d), 7.79(2H d), 7.54(2H d), 7.64(1H s), 8.32(2Hd), 8.34(2Hd), 8.61(2Hd), 8.64(2Hd).
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap