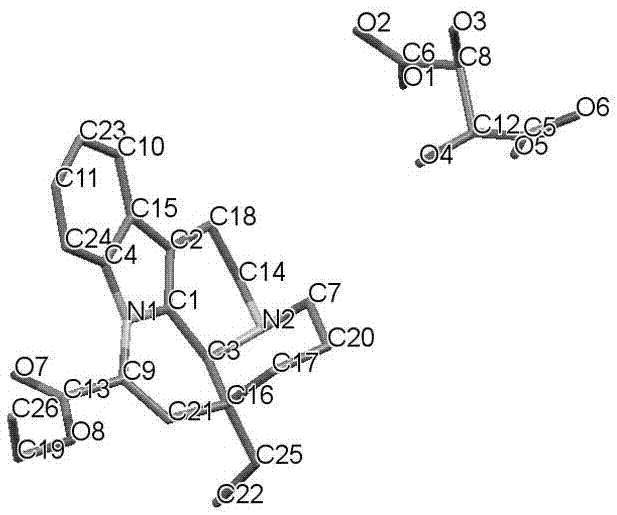
Crystal form and preparation method of a salt formed by vinpocetine and d-tartaric acid
A technology of vinpocetine and tartaric acid, applied in organic chemistry methods, organic chemistry, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0017] Add vinpocetine (350mg, 1mmol) and D-tartaric acid (150mg, 1mmol) into a 50ml Erlenmeyer flask at 0°C, add 20ml of methanol, and add in batches. When the solution is slightly turbid, stop adding and stir. After 1 hour, it was directly heated to 40 °C, a large amount of solids were precipitated, filtered and dried to obtain the product.
Embodiment 2
[0019] Add vinpocetine (350mg, 1mmol) and D-tartaric acid (150mg, 1mmol) into a 50ml Erlenmeyer flask at 10°C, add 20ml of methanol, and add in batches. When the solution is slightly turbid, stop adding and stir. After 1 hour, it was directly heated to 40 °C, a large amount of solids were precipitated, filtered and dried to obtain the product.
Embodiment 3
[0020] Embodiment 3: solid preparation (vinpocetine tablet) stability test data:
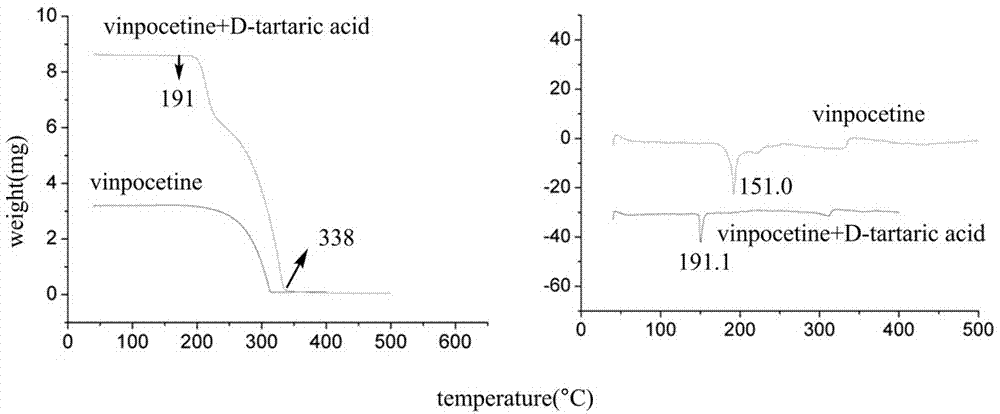
[0021] During the long-term six-month stability stakeout period, the new salt crystal has good stability and no new impurities appear. At the same time, the purity of the raw material can be further improved, and the solubility is also greatly improved.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More