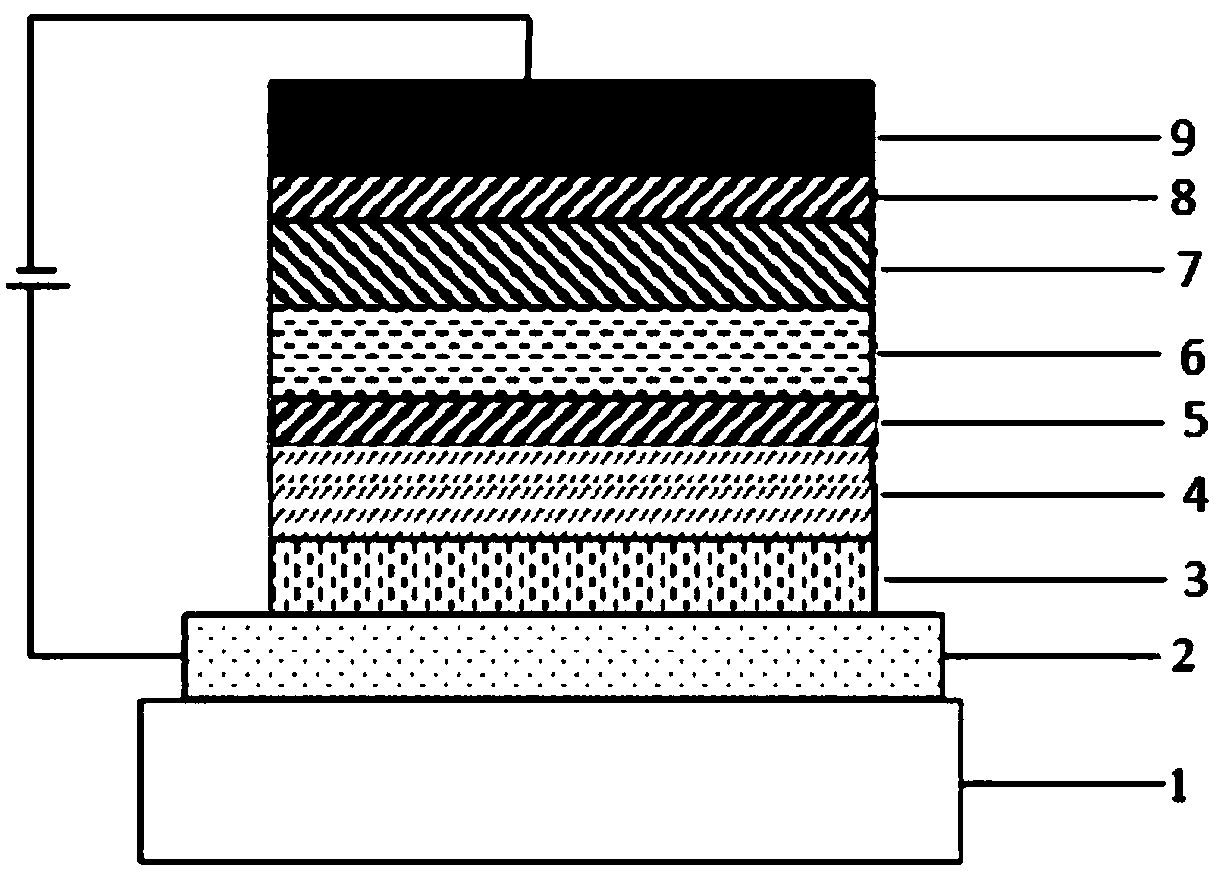
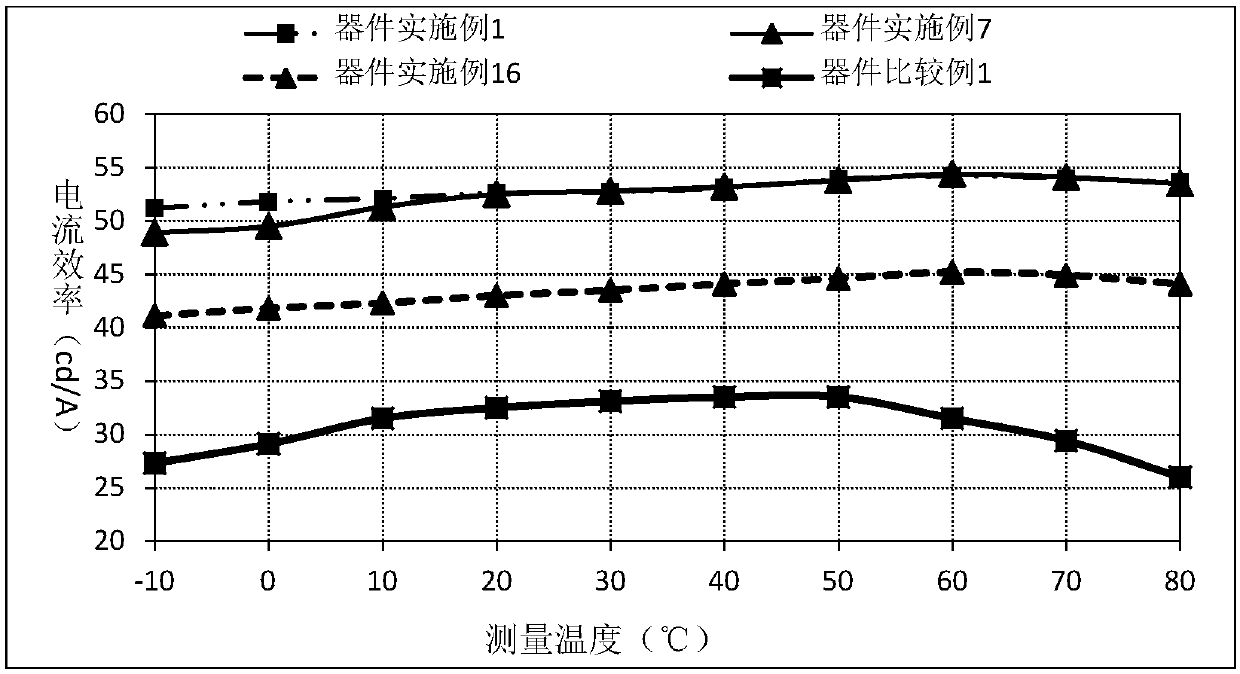
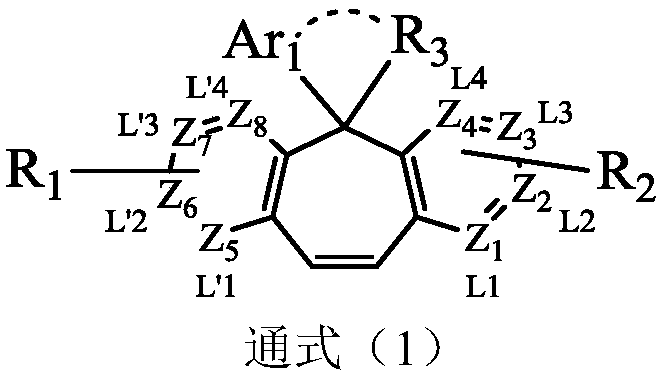
Compound taking cycloheptene as core and application of compound on organic light-emitting devices
A compound, the technology of cycloheptene, applied in the application field of organic electroluminescent devices, can solve different problems
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0223] Embodiment 1: the synthesis of compound 5:
[0224]
[0225] In a 250ml four-necked flask, add 0.05mol of intermediate A1, 0.06mol of Mg powder, and 60ml of tetrahydrofuran under nitrogen atmosphere, and heat to reflux for 4 hours. The reaction is complete and the Grignard reagent is generated;
[0226] Dissolve 0.05mol of raw material 1 in 50ml of tetrahydrofuran, add the above-mentioned Grignard reagent dropwise, react at 60°C for 24 hours, a large amount of white precipitate is formed, and finally add saturated NHCl 4 Convert the Grignard salt into alcohol; after the reaction, extract with ether, dry and rotary evaporate, and purify on a silica gel column with petroleum ether:dichloromethane mixed solvent (3:2) to obtain a slightly yellow solid tertiary alcohol (83% yield) ; using DEI-MS to identify the compound, formula C 34 h 26 O, ESI-MS (m / z) (M + ): The theoretical value is 450.58, and the measured value is 450.55;
[0227] Take 0.04mol of the above-prepa...
Embodiment 2
[0228] Embodiment 2: the synthesis of compound 25:
[0229]
[0230] The preparation method of compound 25 is the same as that of Example 1, except that raw material 1 is replaced by raw material 2, and intermediate A1 is replaced by intermediate A2.
[0231] Elemental analysis structure (molecular formula C 48 h 46 ): theoretical value C, 92.56; H, 7.44; test value: C, 92.55; H, 7.45. ESI-MS(m / z)(M + ): The theoretical value is 622.90, and the measured value is 622.92.
Embodiment 3
[0232] Embodiment 3: the synthesis of compound 41:
[0233]
[0234] The preparation method of compound 41 is the same as that of Example 1, except that intermediate A1 is replaced by intermediate A3.
[0235] Elemental analysis structure (molecular formula C 40 h 30 ): theoretical value C, 94.08; H, 5.92; test value: C, 94.10; H, 5.90. ESI-MS(m / z)(M + ): The theoretical value is 510.68, and the measured value is 510.70.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Thickness | aaaaa | aaaaa |
| Thickness | aaaaa | aaaaa |
| Thickness | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More