Preparation method of HIV-1 nucleic acid detection quality control product based on influenza virus vector
A HIV-1 and influenza virus technology, applied in the direction of microorganism-based methods, viruses/phages, botanical equipment and methods, etc., can solve the problems of biological safety hazards, poor stability, unstable nucleic acid load, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0055] In order to make the technical problems, technical solutions and advantages to be solved by the present invention clearer, the following will describe in detail with reference to the drawings and specific embodiments.
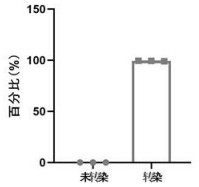
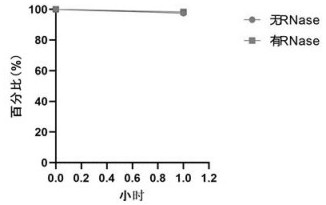
[0056] The technical problem solved by the present invention is to provide a quality control product for HIV-1 nucleic acid detection using CRISPR-Cas9 large fragment knock-in technology combined with influenza virus as a carrier and its preparation method. The quality control product of the present invention has a high similarity with the actual HIV-1 virus structure, can simulate HIV-1 virus particles very realistically, and realize all-round monitoring of HIV-1 nucleic acid detection; at the same time, refer to standardized influenza inactivated vaccine preparation Process preparation of quality control products has the advantages of low cost, high stability, easy storage and transfer, and is convenient for large-scale production at the industrial leve...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap