Synthesis method of N, N-dimethyl trifluoromethanesulfonamide
A technology of dimethyltrifluoromethanesulfonamide and trifluoromethanesulfonyl fluoride, which is applied in the field of battery electrolyte additives, can solve the problems of harsh conditions, poor purity, and low reaction yield, and achieve a mild reaction process, Increased yield and simple reaction process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
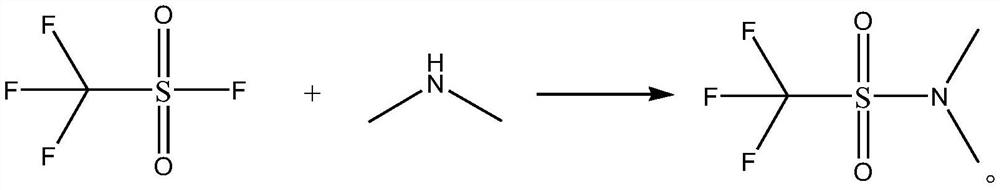
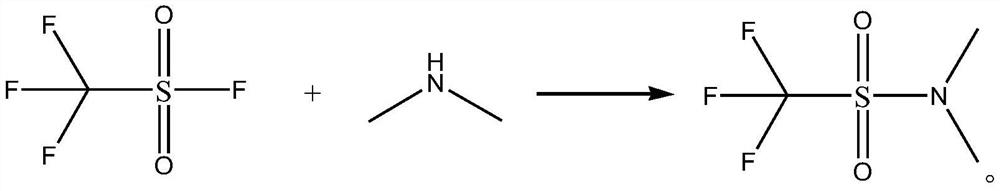
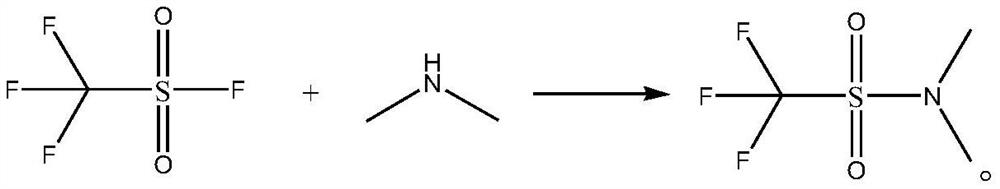
[0021] The synthetic method of N,N-dimethyltrifluoromethanesulfonamide, its synthetic steps are as follows:
[0022] Take 16.91g (0.15mol) of 40wt% dimethylamine aqueous solution and add it to the autoclave, cool down to 5°C, slowly add 15.2g (0.1mol) trifluoromethanesulfonyl fluoride dropwise, after the dropwise addition, add 8.1g (0.08mol) triethylamine, then increase the pressure to 0.5MPa, maintain 0.5MPa and raise the temperature to 20°C for amidation reaction for 20h, after the amidation reaction is completed, concentrate, and then use ether as the eluent to go through column chromatography Purify, collect, concentrate the eluent, and concentrate under reduced pressure to obtain 15.89 g of N,N-dimethyltrifluoromethanesulfonamide with a yield of 89.70% and a purity of 99.5%. The specific chemical reaction formula is as follows:
[0023]
Embodiment 2
[0025] The synthetic method of N,N-dimethyltrifluoromethanesulfonamide, its synthetic steps are as follows
[0026] Take 19.16g (0.17mol) of 40wt% dimethylamine aqueous solution and add it to the autoclave, cool down to 0°C, slowly add 15.2g (0.1mol) trifluoromethanesulfonyl fluoride dropwise, after the dropwise addition, add 7.08g (0.07mol) triethylamine, then increase the pressure to 0.1MPa, maintain 0.1MPa and raise the temperature to 15°C for amidation reaction for 22h, after the amidation reaction is completed, concentrate, and then use ether as the eluent to go through column chromatography After purification, the eluate was collected, concentrated, and concentrated under reduced pressure to obtain 15.64 g of N,N-dimethyltrifluoromethanesulfonamide, with a yield of 88.29% and a purity of 99.6%.
Embodiment 3
[0028] The synthetic method of N,N-dimethyltrifluoromethanesulfonamide, its synthetic steps are as follows:
[0029] Take 18.03g (0.16mol) of 40wt% dimethylamine aqueous solution and add it to the autoclave, cool down to 3°C, slowly add 15.2g (0.1mol) trifluoromethanesulfonyl fluoride dropwise, after the dropwise addition, add 6.07g (0.06mol) triethylamine, then increase the pressure to 0.3MPa, maintain 0.3MPa and raise the temperature to 10°C for amidation reaction for 20h, after the amidation reaction is completed, concentrate, and then use ether as the eluent to go through column chromatography After purification, the eluate was collected, concentrated, and concentrated under reduced pressure to obtain 15.73 g of N,N-dimethyltrifluoromethanesulfonamide, with a yield of 88.79% and a purity of 99.6%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More