Selective preventing and therapeutic agents for progressive lesion after organic damage
a technology of organic damage and selective prevention, applied in the direction of drugs, instruments, immunological disorders, etc., can solve the problems of severe side effects, prolonged steroid treatment, and further worsening of hereditary lesion, so as to achieve selective and effective therapy, prevent the induction of effector macrophages, and reduce the effect of side effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
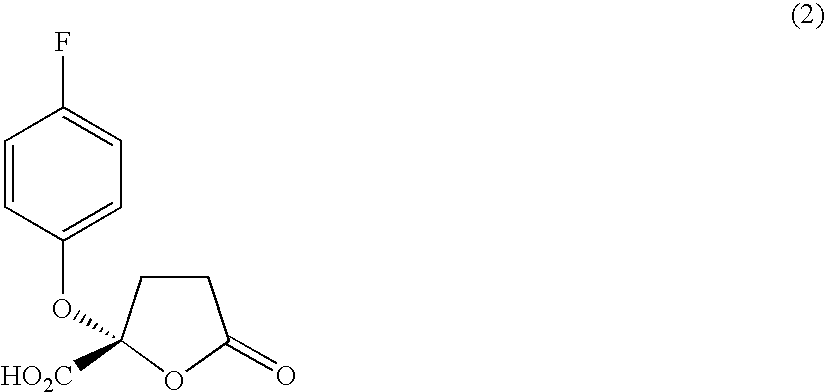
Manufacture of (RS)-(−)-α-methyl-2-naphthalene-methyl 2-(4-fluorophenoxy)-5-oxotetrahydrofuran-2-carboxylate
[0257]
[0258] To a solution prepared by dissolving 164 mg (0.6 mmol, 1 equivalent) of 2-(4-fluorophenoxy)-5-oxotetrahydrofuran-2-carboxylic acid manufactured by a method mentioned in paragraph [0018], page 5 of Japanese Patent Laid-Open No. 04 / 338,331 in 2.2 ml of ether at 0° C. were added 240 μl (2.7 mmol, 4 equivalents) of (COCl)2 gradually and then two drops of dimethylformamide (DMF) were added thereto. At that time, discharge of gas was observed.
[0259] The reaction solution was allowed to stand for 1 hour with stirring at 0° C., ether was removed in vacuo and the product was dried in vacuo.
[0260] The product was dissolved in 2.5 ml of ether, then 130 mg (0.75 mmol, 1.1 equivalents) of (S)-(−)-α-methyl-2-naphthalenemethanol and 8 mg (0.07 mmol, 0.1 equivalent) of dimethylaminopyrrolidone (hereinafter, abbreviated as DMAP) were gradually added and, at the same time, 140 μ...
example 2
Manufacture of (±)-2-(4-fluorophenoxy)-5-oxotetrahydrofuran-2-carboxylic Acid
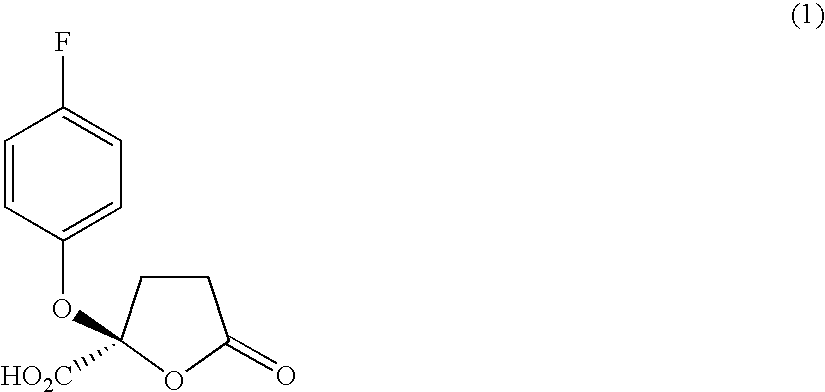
(a) Manufacture of (−)-2-(4-fluorophenoxy)-5-oxotetrahydrofuran-2-carboxylic Acid
[0277]
[0278] C11H9FO5
[0279] Molecular weight 240.1
[0280] White solid
[0281] Tf=128° C.
[0282] To a solution of 98 mg (0.24 mmol, 1 equivalent) of (S)-(−)-α-methyl-2-naphthalenemethyl 2-(4-fluorophenoxy)-5-oxotetrahydrofuran-2-carboxylate represented by the formula (3-1) manufactured in Example 1 dissolved in 4 ml of ethyl acetate and 8 drops of ethanol was added a Pd / C catalyst in an amount of 10 parts by weight to 100 parts by weight of the above solution. The resulting solution was washed with water several times, stirred at room temperature and subjected to a catalytic reduction using hydrogen for 4 hours. The product was dissolved in ethyl acetate, the catalyst was removed by filtering through Celite (manufactured by Johns Manville Sales Co.) and the solvent was removed in vacuo. The product was purified by means of a r...
example 3
Manufacture of Benzyl 2-(4-fluorobenzyl)-5-oxotetrahydrofuran-2-carboxylate
[0294]
[0295] C19H17FO4
[0296] Molecular weight=328.3
[0297] White crystals
[0298] Tf=70° C.
[0299] In the presence of Amberyst resin 15 (manufactured by Rohm & Haas Co.), a solution where 192 mg (0.4 mmol, 1 equivalent) of dibenzyl 2-(4-fluorobenzyl)-2-hydroxypentane-1,5-dicarboxylate were dissolved in 6 ml of anhydrous toluene was heated at 60° C. for 10 hours. This was returned to room temperature and filtered through cotton to remove the Amberyst resin 15. The solvent was removed in vacuo and the product was separated by chromatography (eluent: hexane / ethyl acetate, 8.5:2.5 (v / v)). After purification by recrystallization from ether-hexane twice, the present compound was obtained as thin flaky crystals in an amount of 99 mg (yield: 76%).
[0300] Thin-layer chromatography: Rf=0.24 (hexane / ethyl acetate, 7:3 (v / v))
[0301] RMN 1H (300 MHz; CDCl3) d ppm: 2.15-2.52 (4H, m); 3.1 (1H, d. J=14.4 Hz); 3.3 (1H, d, J=...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More