Transgenic non-human animals for producing heterologous and chimeric antibodies
a technology of chimeric antibodies and transgenic animals, which is applied in the field of transgenic nonhuman animals capable of producing heterologous antibodies, can solve the problems of serious obstacles to obtaining b-lymphocytes which produce human immunoglobulins, and affecting the expression of endogenous immunoglobulin chains. , to achieve the effect of suppressing the expression and suppressing the expression of one or more endogenous immunoglobulin
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
experimental examples
Methods and Materials
[0392] Transgenic mice ate derived according to Hogan, et al., “Manipulating the Mouse Embryo: A Laboratory Manual”, Cold Spring Harbor Laboratory, which is incorporated herein by reference.
[0393] Embryonic stem cells are manipulated according to published procedures (Teratocarcinomas and embryonic stem cells: a practical approach, E. J. Robertson, ed., IRL Press, Washington, D.C., 1987; Zjilstra et al., Nature 342:435-438 (1989); and Schwartzberg et al., Science 246:799-803 (1989), each of which is incorporated herein by reference).
[0394] DNA cloning procedures are carried out according to J. Sambrook, et al. in Molecular Cloning: A Laboratory. Manual, 2d ed., 1989, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y., which is incorporated herein by reference.
[0395] Oligonucleotides are synthesized on an Applied Bio Systems oligonucleotide synthesizer according to specifications provided by the manufacturer.
[0396] Hybridoma cells and antibodies ...
example 1
Genomic Heavy Chain Human Ig Transgene
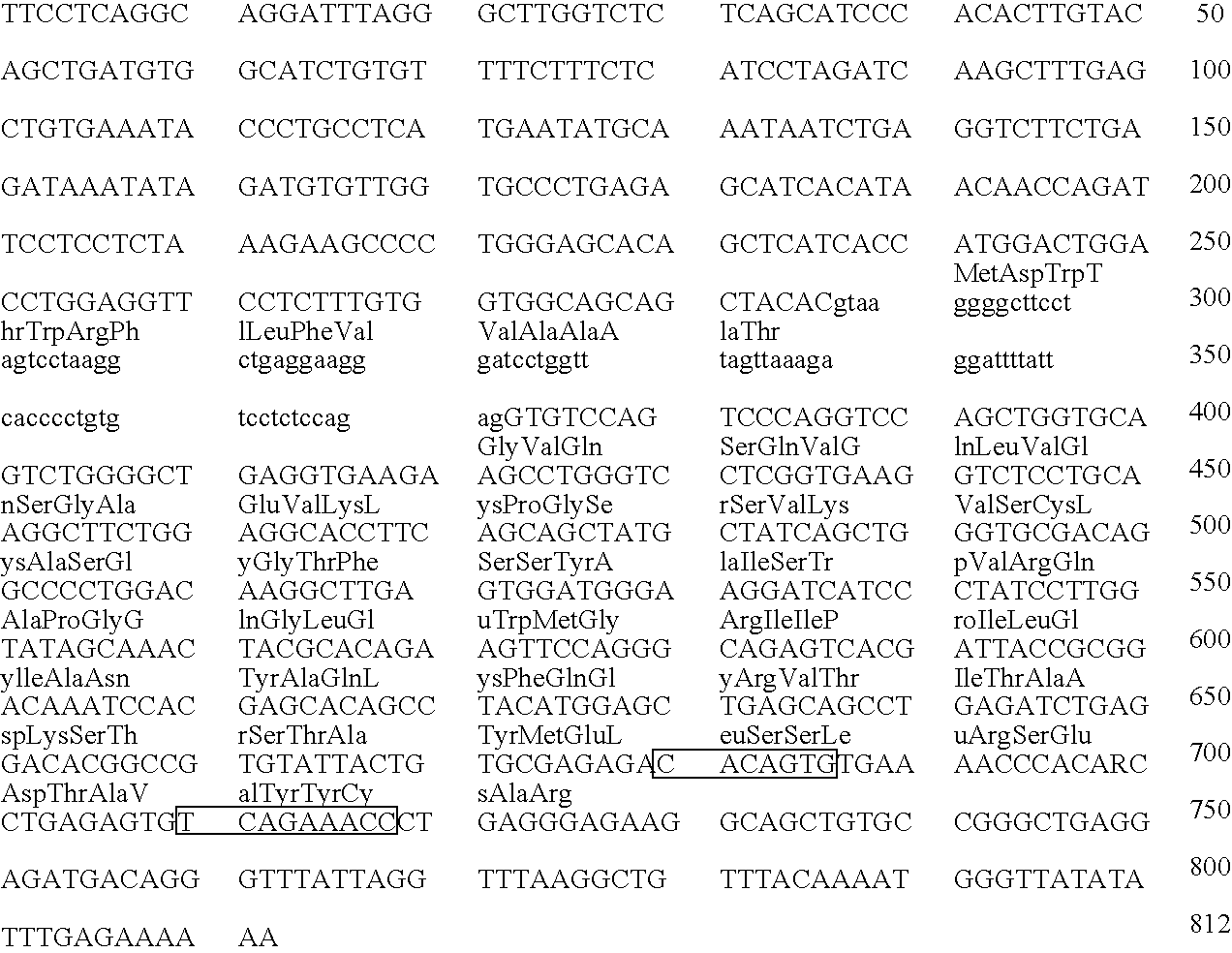
[0397] This Example describes the cloning and microinjection of a human genomic heavy chain immunoglobulin transgene which is microinjected into a murine zygote.
[0398] Nuclei are isolated from fresh human placental tissue as described by Marzluff et al., “Transcription and Translation: A Practical Approach”, B. D. Hammes and S. J. Higgins, eds., pp. 89-129, IRL Press, Oxford (1985)). The isolated nuclei (or PBS washed human spermatocytes) are embedded in a low melting point agarose matrix and lysed with EDTA and proteinase K to expose high molecular weight DNA, which is then digested in the agarose with the restriction enzyme NotI as described by M. Finney in Current Protocols in Molecular Biology (F. Ausubel, et al., eds. John Wiley & Sons, Supp. 4, 1988, Section 2.5.1).
[0399] The NotI digested DNA is then fractionated by pulsed field gel electrophoresis as described by Anand et al., Nucl. Acids Res. 17:3425-3433 (1989). Fractions enriched f...
example 2
Genomic K Light Chain Human Ig Transgene Formed by In Vivo Homologous Recombination
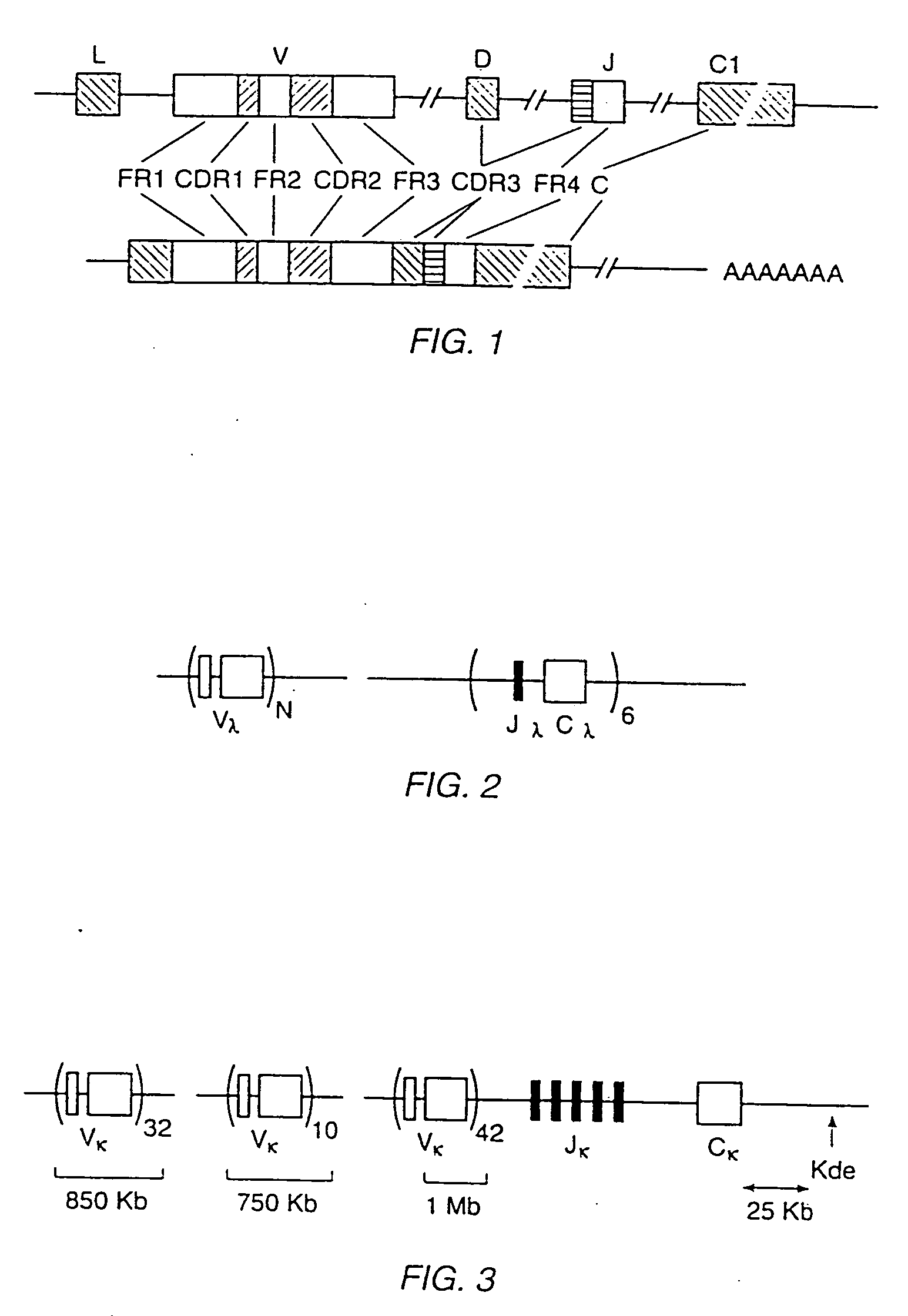
[0401] A map of the human K light chain has been described in Lorenz et al., Nucl. Acids Res. 15:9667-9677 (1987), which is incorporated herein by reference.
[0402] A 450 kb XhoI to NotI fragment that includes all of Cκ, the 3′ enhancer, all J segments, and at least five different V segments is isolated and microinjected into the nucleus of single cell embryos as described in Example 1.
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentrations | aaaaa | aaaaa |
| concentrations | aaaaa | aaaaa |
| concentrations | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More