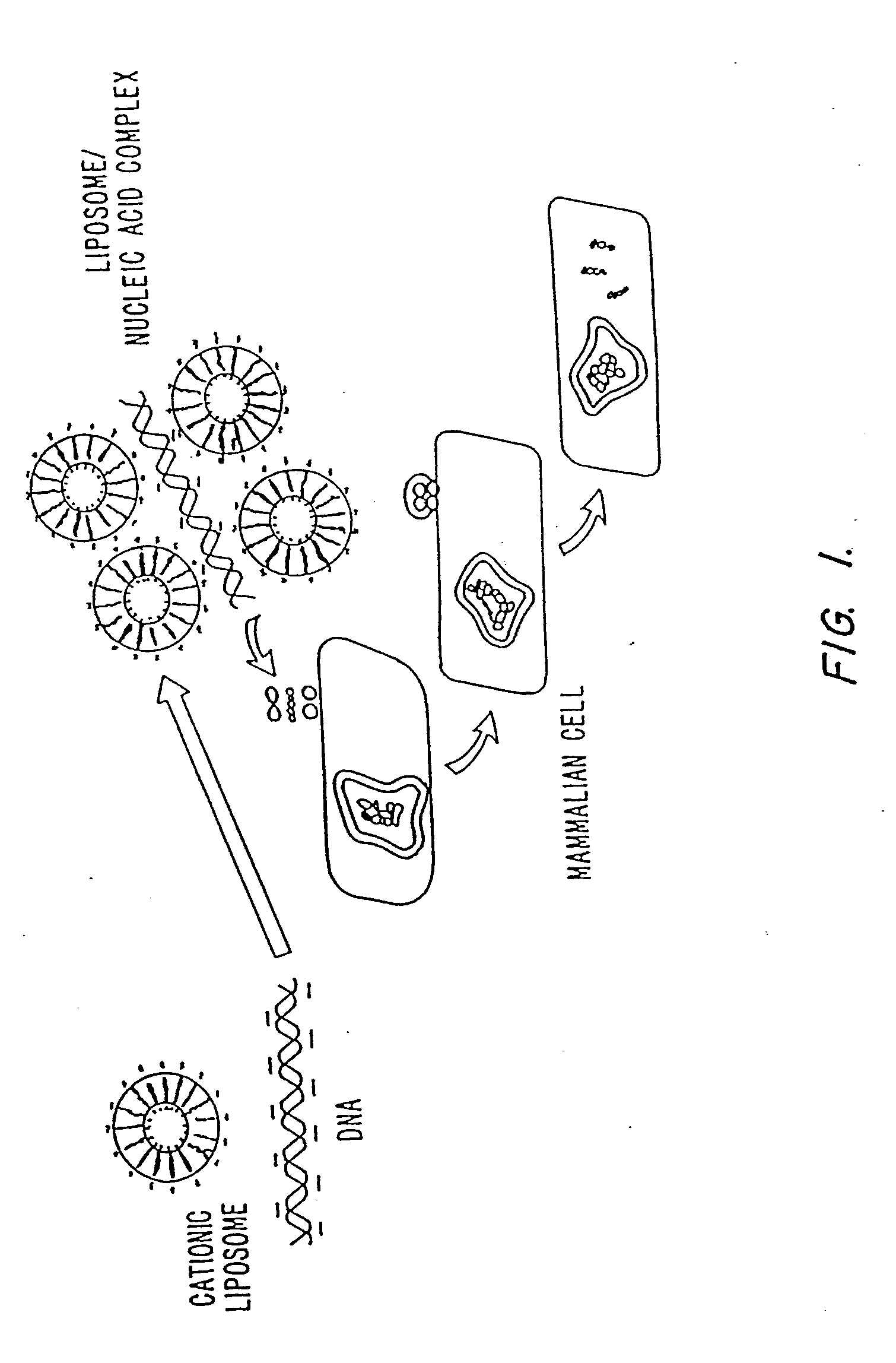
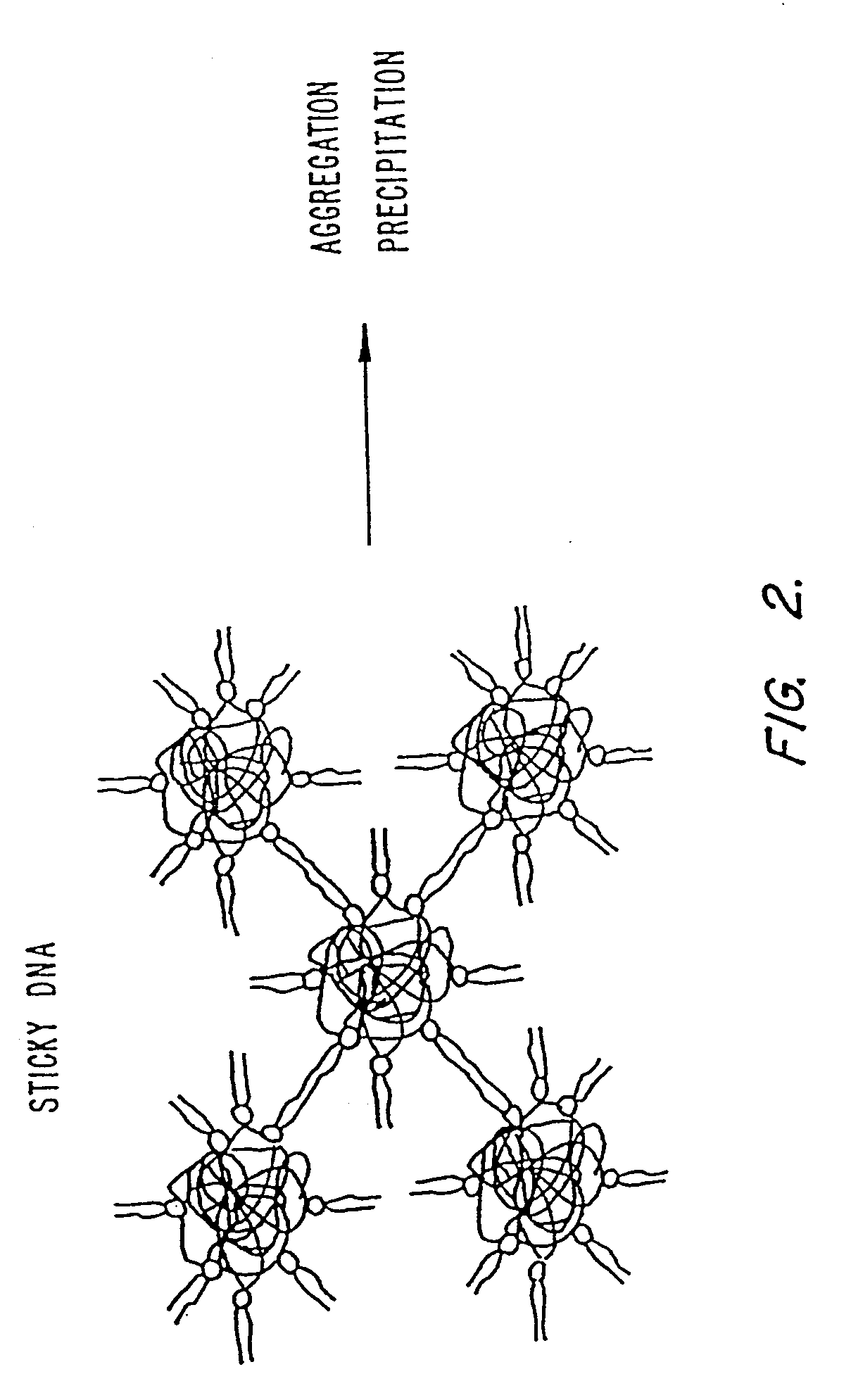
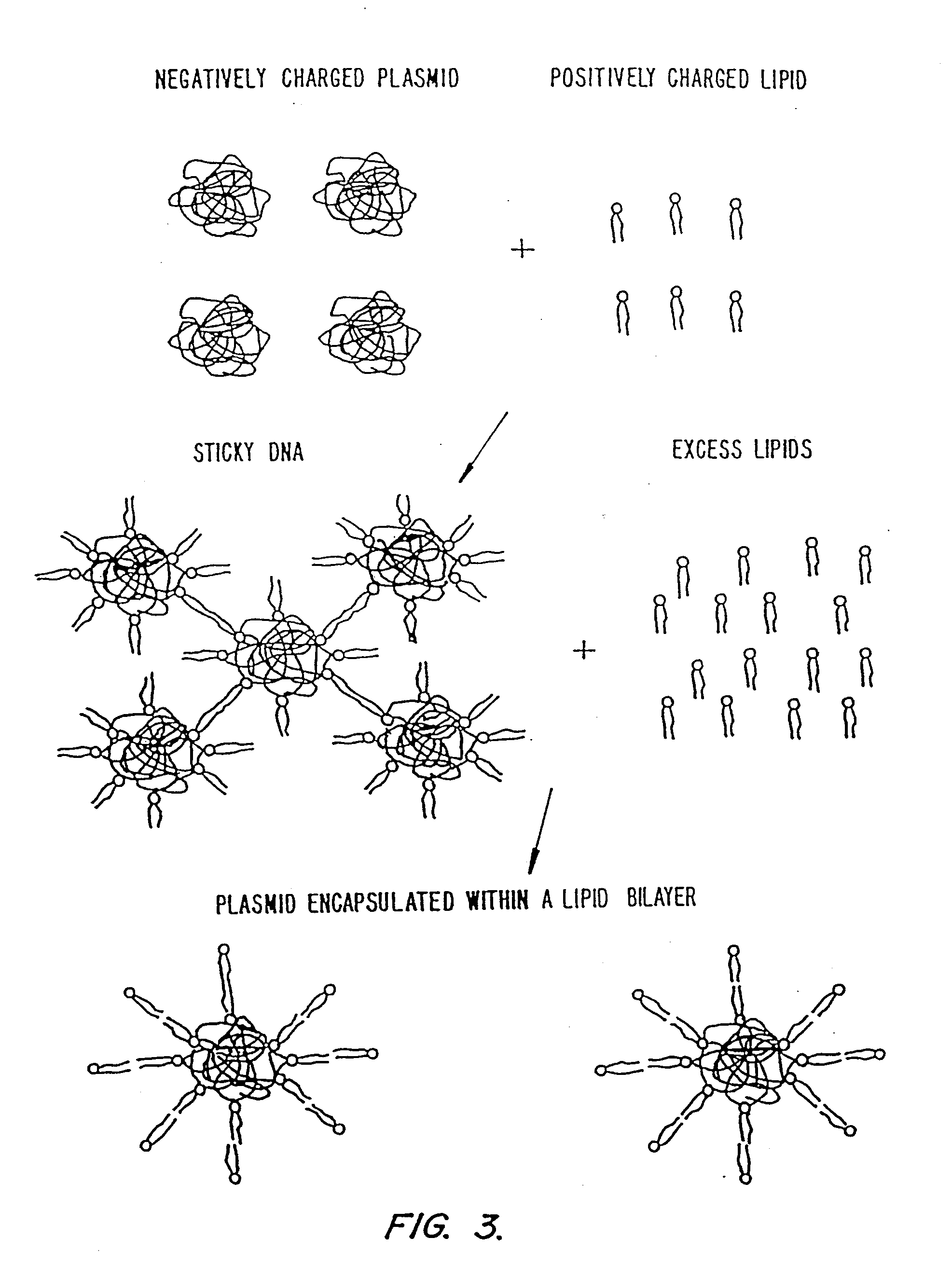
Methods for encapsulating plasmids in lipid bilayers
a technology of lipid bilayer and plasmid, which is applied in the field of lipid encapsulated plasmids or antisense constructs, can solve the problems of prohibitively expensive, less effective than dotma in most cell lines, and the most efficient dotma and dospa formulations
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0100]This example illustrates the encapsulation of a plasmid in a lipid bilayer system using either a reverse-phase method or a detergent dialysis method.
[0101]Reverse Phase Method
[0102]pCMV4-CAT plasmid (50 μg) was encapsulated in a lipid bilayer which was constructed using 20 mg POPC:PEG-Cer-C20 (95:5 mole % ratio) with between 0 and 0.3 mg DODAC. The encapsulation method utilized a modification of the classical reverse phase method for entrapment. Specifically, 1.050 mL of chloroform:methanol in a 1:2.1 mole % ratio was added to a lipid film containing 2 μL of 14C-cholesteryl hexadecyl ether (6.66 μL / μCi). This was followed by the addition of 220 μL H2O and 33 μL 3H-pCMV4-CAT plasmid (158,000 dpm / μL; 1.5 mg / mL). This combination provided a clear single phase. The CHCl3 and most of the methanol were removed under a stream of nitrogen while vortexing the mixture. The resulting 250 μL suspension of encapsulated plasmid was diluted with 1 mL of H2O and extruded 5 times through one 4...
example 2
[0105]This example illustrates the level of plasmid “protection” from external medium using anion exchange chromatography.
[0106]The extent of encapsulation or protection of the plasmid from the external medium was assessed by anion exchange chromatography as follows: a 50 μL aliquot of each sample was eluted on a DEAE Sepharose CL-6B column and the fractions were assessed for both 3H-plasmid and 14C-lipid by scintillation counting. Any exposed negative charges, such as those present on DNA molecules will bind to the anion exchange column and will not elute with the 14C-lipid. DNA which has its negative charge “protected” or non-exposed will not bind to the ion exchange resin and will elute with the 14C-lipid.
[0107]Reverse Phase Method (Particles with POPC:DODAC:PEG-Cer(C20)
[0108]FIG. 4 presents the results describing the relationship between DODAC:plasmid charge ratio (see Table 1 for amounts of POPC, DODAC and PEG-Cer(C20) using 20 mg total lipid) and percent recovery of plasmid af...
example 3
[0113]This example illustrates the serum stability achieved using plasmid:lipid particles prepared by the methods of Example 1.
[0114]To establish the serum stability of the plasmid:lipid particles, aliquots of the particle mixtures prepared according to both the reverse phase and dialysis methods of Example 1 were incubated in mouse serum (Cedar Lane) for 15 min and for 30 min at 37° C. Prior to incubation, the lipid associated plasmid was eluted on a DEAE Sepharose CL-6B column to remove unencapsulated plasmid. Following incubation, an aliquot of the incubation mixture was eluted in HBS on a Sepharose CL-4B column.
[0115]As a control, 1.5 mg of free 3H-pCMV4-CAT was eluted on a Sepharose CL-4B column in HBS, pH 7.4 (see FIG. 10). For comparison, 1.5 mg of free 3H-pCMV4-CAT was incubated in 500 μL of mouse serum at 37° C. for 30 min and eluted in the same manner (see FIG. 11). Note that in FIG. 10, the free plasmid eluted in the void volume of the column while, in FIG. 11, the plasmi...
PUM
| Property | Measurement | Unit |
|---|---|---|
| size | aaaaa | aaaaa |
| size | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More