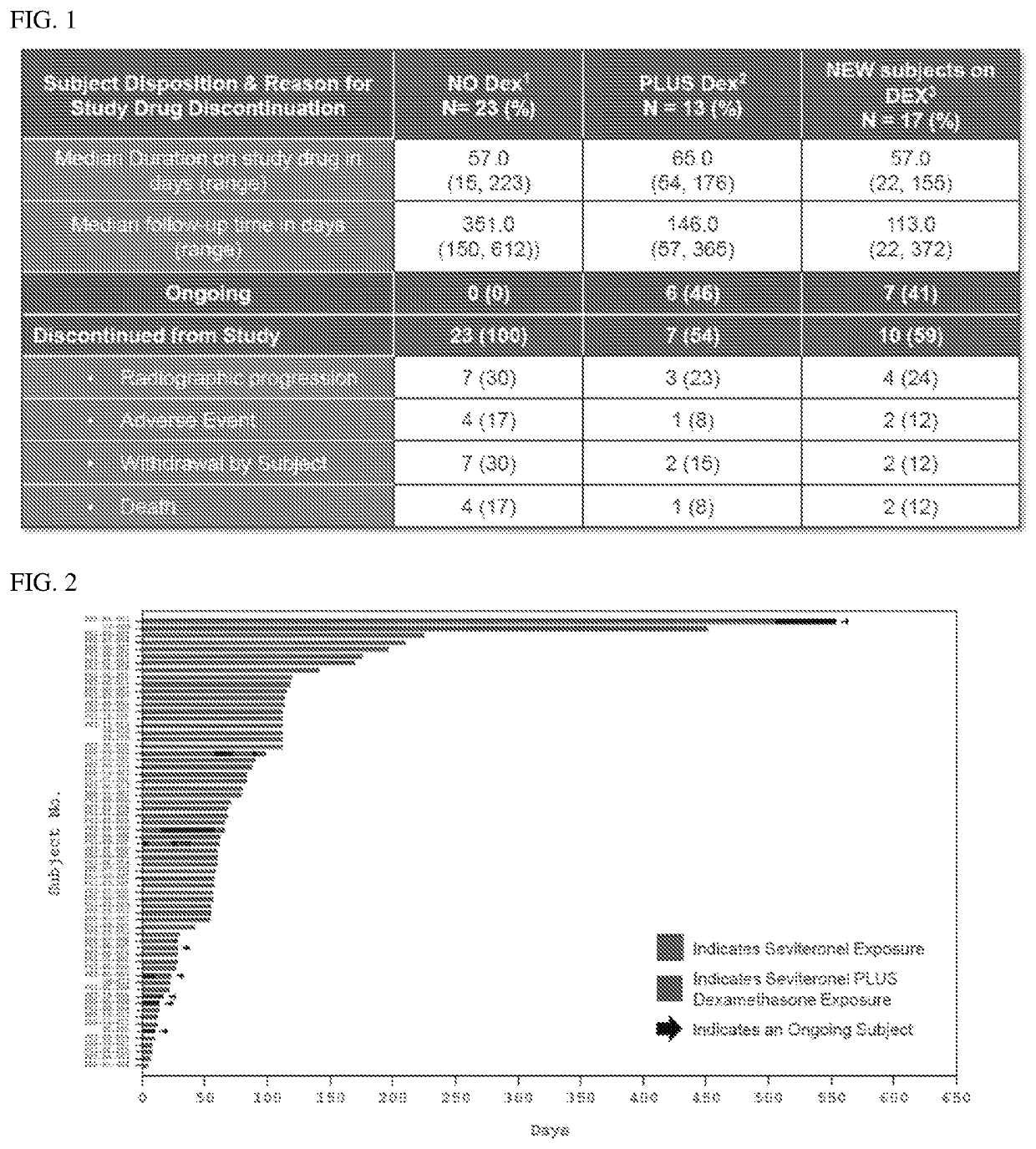
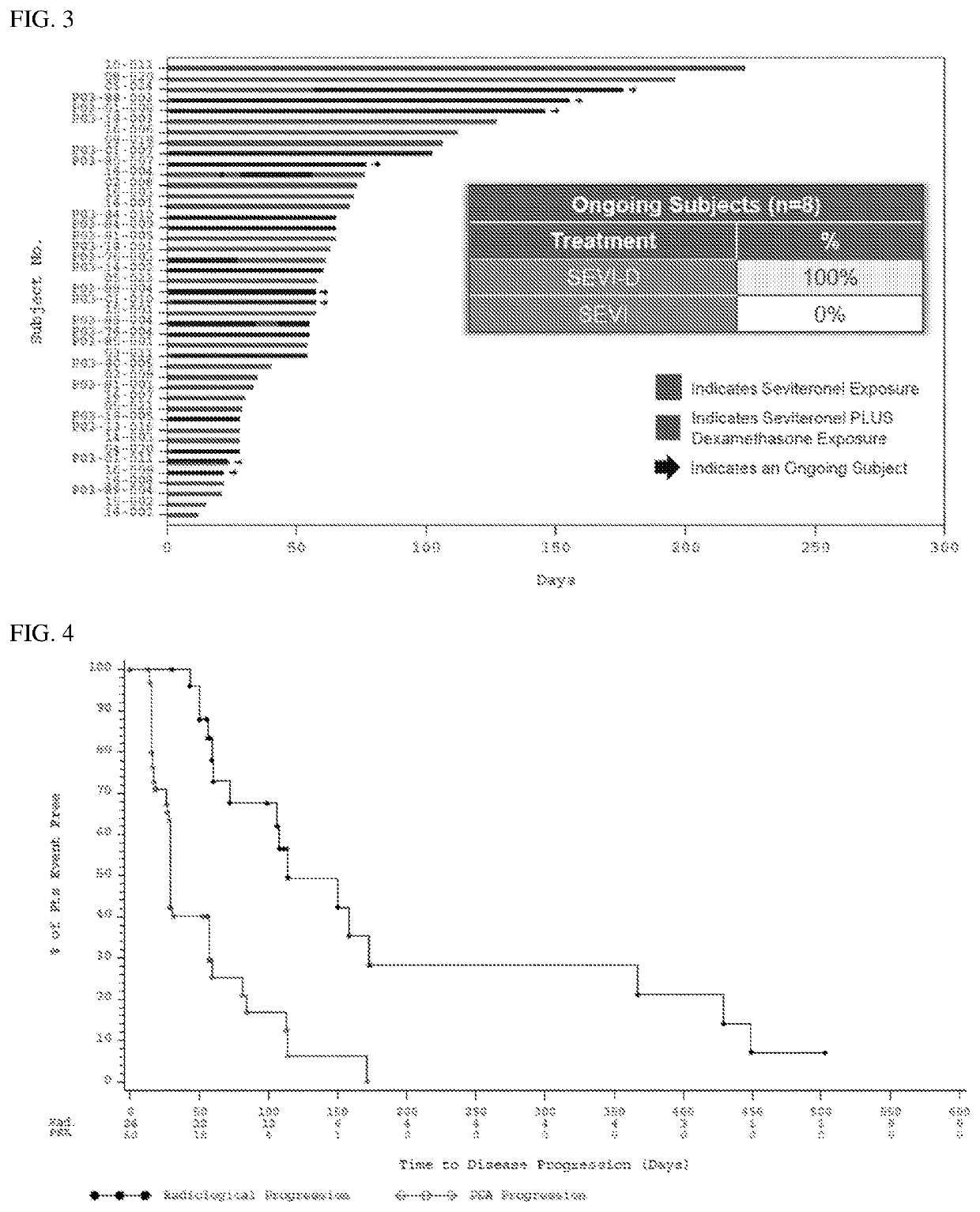
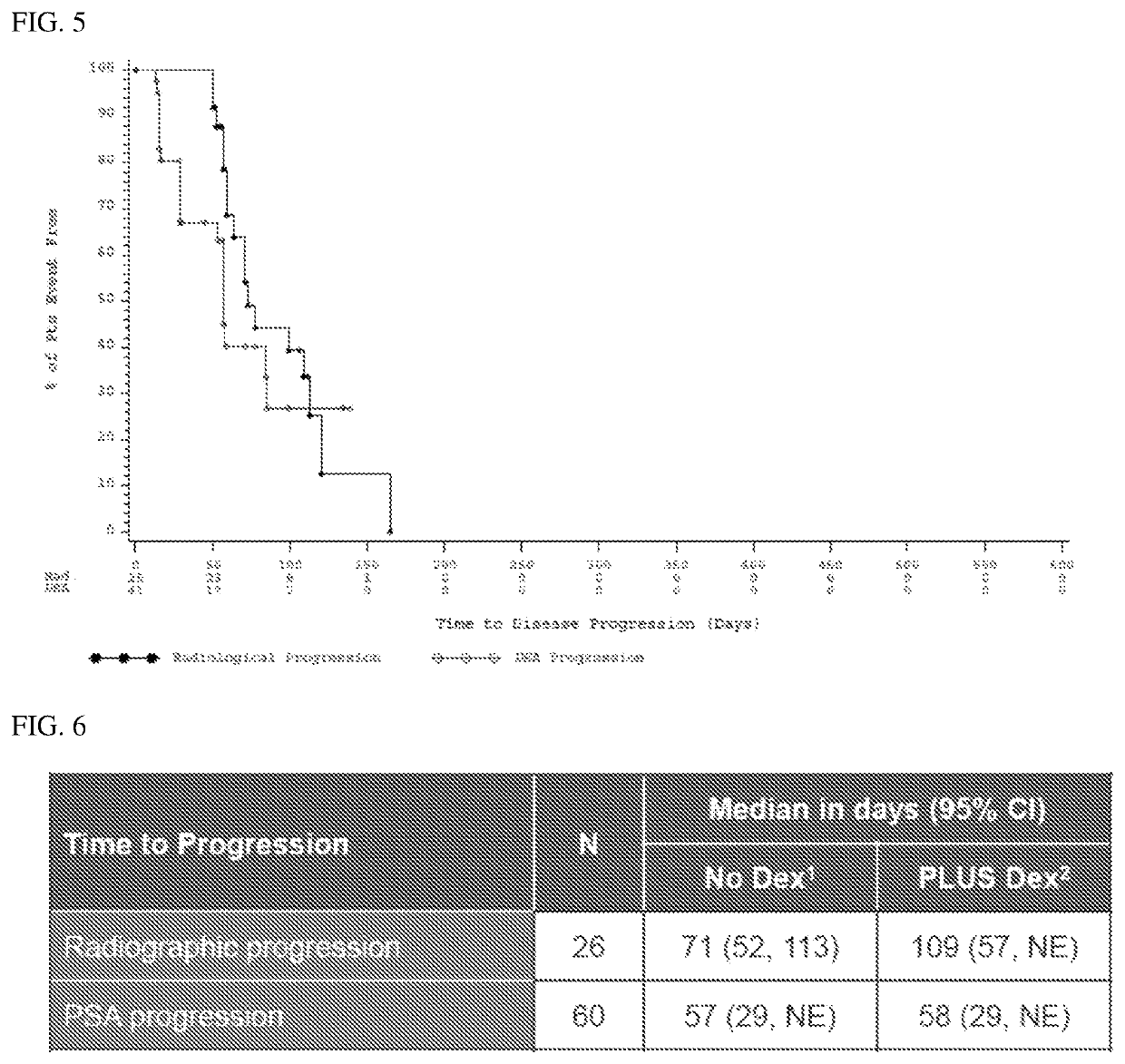
Treatment paradigm for an Anti-cd19 antibody and venetoclax combination treatment
an anti-cd19 antibody and treatment paradigm technology, applied in the field of treatment paradigm for anticd19 antibody and venetoclax combination treatment, can solve the problem of dramatic decrease in the activity of the enzyme, and achieve the effect of facilitating the processing of active compounds and preventing inactivation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
1-(6,7-Bis(difluoromethoxy)naphthalen-2-yl)-2-methyl-1-(1H-1,2,3-triazol-4-yl)propan-1-ol (3)
[0139]To a stirred solution of A (18 g, 69 mmol) in DCM (180 mL) was added BBr3 (87.2 g, 348 mmol) dropwise at −40° C. After completion of addition, stirring was continued for 1 h at −40° C. and 1 h at RT. The reaction mixture was poured into cold water and aqueous layer was then extracted with DCM (2×200 mL). The combined organic extracts were washed with water (100 mL), brine (100 mL) and dried over anhydrous Na2SO4. After filtration and evaporation of solvent under reduced pressure, the crude material was purified by column chromatography (SiO2, 100-200 mesh) to afford K (9.0 g, 39 mmol, 56%) as a brown solid. 1H NMR (200 MHz, CDCl3): δ□8.29 (s, 1H), 7.88 (dd, J=8.8, 1.6 Hz, 1H), 7.68 (d, J=8.6 Hz, 1H), 7.36 (s, 1H), 7.26 (s, 1H), 5.88 (br s, 2H), 3.79-3.63 (m, 1H), 1.27 (d, J=6.8 Hz, 6H).
To a stirred solution of K (5.0 g, 21.7 mmol) in DMF (50 mL) were added ethyl bromo difluoroacetate (...
PUM
| Property | Measurement | Unit |
|---|---|---|
| enantiomeric excess | aaaaa | aaaaa |
| enantiomeric excess | aaaaa | aaaaa |
| enantiomeric excess | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More