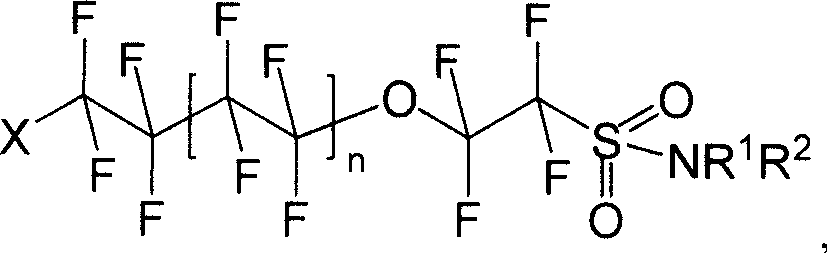
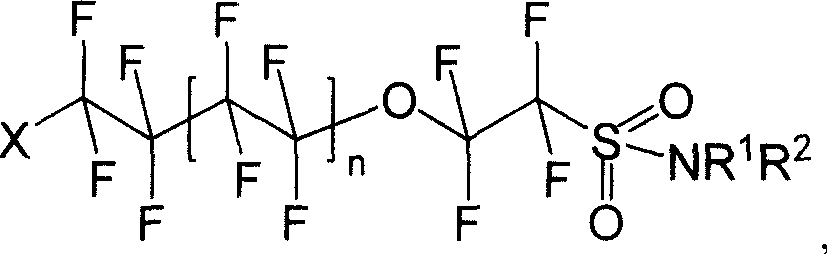
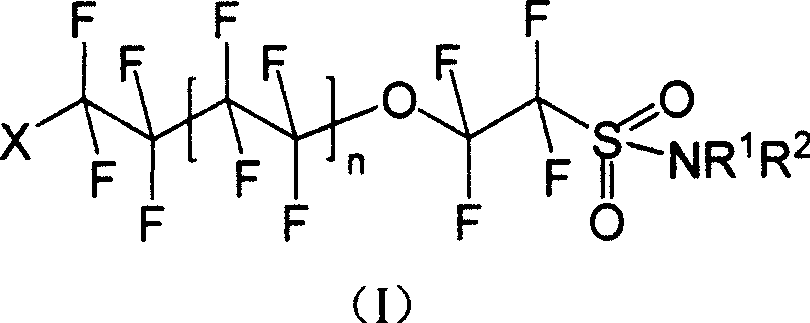
Oxo fluoroalkyl sulfamide compound and its preparing method and use
A technology of heterofluoroalkyl sulfonamides and compounds, which is applied in the field of oxafluoroalkyl sulfonamide compounds, preparation and application, can solve problems such as large hazards, and achieve reasonable toxicity, fast drug effect, and high-efficiency killing The effect of cockroach activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0041] Synthesis of I-1: Dissolve 8.520g (20mmol) of (II) in ether, add 5.228g (60mmol) of morpholine, stir and react for 8h under dark conditions, wash with water and saturated NaCl aqueous solution successively, separate liquids, and diethyl ether The layer was dried with anhydrous sodium sulfate and spin-dried to obtain 4.592 g of white solid I-1 with a yield of 46.56%.
[0042] Mp (melting point): 64-66°C
[0043] 1 H NMR (nuclear magnetic spectrum) (CD 3 Cl) (ppm): 3.77 (4H, s), 3.56 (4H, t).
[0044] 19 F NMR (CD 3 Cl) (ppm) (fluorine nuclear magnetic spectrum): -65.11 (2F, s), -82.27 (2F, t), -85.68 (2F, d), -115.86 (2F, s).
Embodiment 2
[0046] Synthesis of I-2: Dissolve 8.520 g (20 mmol) of (II) in diethyl ether, add 5.109 g (60 mmol) of hexahydropyridine, stir and react for 8 h under dark conditions, wash with water and saturated NaCl aqueous solution successively, and separate the liquids. The ether layer was dried over anhydrous sodium sulfate, spin-dried and distilled under reduced pressure. 6.815 g (1-2 mmHg, 106-108° C.) of pale yellow liquid of I-2 was obtained with a yield of 69.38%.
[0047] 1 HNMR (CD 3 Cl) (ppm): 3.70 (2H, m), 3.33 (2H, m), 1.68 (6H, s).
[0048] 19 F NMR (CD 3 Cl) (ppm): -65.07 (2F, s), -82.37 (2F, t), -85.79 (2F, d), -116.27 (2F, s).
Embodiment 3
[0050] Synthesis of I-3: Dissolve 8.520g (20mmol) of (II) in diethyl ether, add 4.38g (60mmol) of n-butylamine, stir and react for 8h under dark conditions, wash with water and saturated NaCl aqueous solution successively, and separate the liquids. The ether layer was dried over anhydrous sodium sulfate, spin-dried and distilled under reduced pressure. A pale yellow liquid of I-3 was obtained with a yield of 72.1%.
[0051] 1 H NMR (DMSO) (ppm): 3.15 (2H, t), 1.47 (2H, m), 1.31 (2H, m), 0.86 (3H, t)
[0052] 19 F NMR (DMSO) (ppm): -65.08 (2F, s), -82.27 (2F, t), -85.59 (2F, d), -116.30 (2F, s).
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More