Purifying method of carperitide
A technology of capecritide and purification method, which is applied in the field of medicinal chemistry, and can solve problems such as the difficulty in achieving the purity of finished medicine and the yield of industrial production, affecting the purity and yield, and poor stability of capecritide hydrochloride
Inactive Publication Date: 2013-01-16
HYBIO PHARMA
View PDF5 Cites 6 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
This patent discloses a capecritide purification process using phosphate buffer purification and acetate conversion to salt, but in the process of purification with phosphate buffer salt, it is difficult to effectively control impurities, which in turn affects the purity and yield; During the salt process, it was found that the stability of capecritide hydrochloride was poor
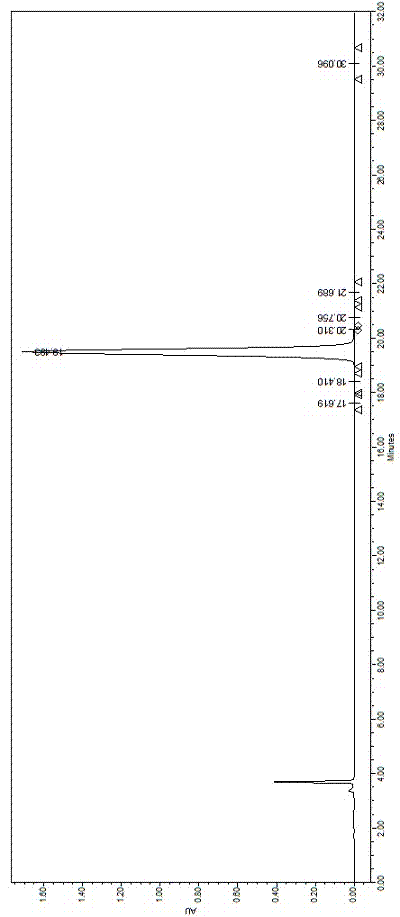
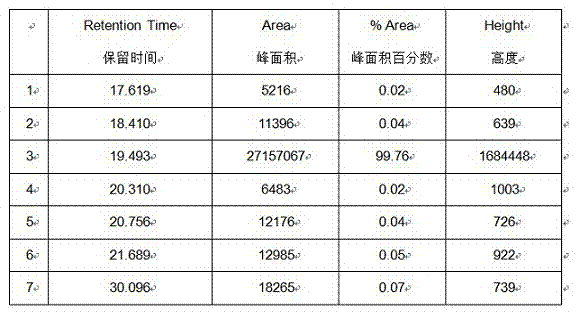
[0005] The method of prior art CN102382188A is used to purify capecritide, the purity can only reach 98.5%, and the total yield is about 32.1%, which is difficult to achieve the purity of finished medicine and the yield of industrial production
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0028] Embodiment 1 sample processing
[0029] Dissolve the crude peptide with 3% DMSO aqueous solution at a concentration of 100 g / L, stir to dissolve the sample completely, filter it with a filter membrane, and collect the filtrate.
Embodiment 2
[0030] Embodiment 2 sample processing
[0031] Dissolve the crude peptide with 10% acetonitrile aqueous solution at a volume ratio of 100 g / L, stir to dissolve the sample completely, filter it with a filter membrane, and collect the filtrate.
Embodiment 3
[0032] Embodiment 3 sample processing
[0033] Dissolve the crude peptide at a concentration of 100 g / L with 18% methanol aqueous solution by volume, stir to dissolve the sample completely, filter it with a filter membrane, and collect the filtrate.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention provides a purifying method of carperitide. The purifying method comprises the following steps of: 1) dissolving a sample through 3 to 18% of an aqueous solution of an organic solvent based on volume by volume concentration, wherein the organic solvent has high polarity; preparing to obtain 100g / L of solution based on concentration; 2), getting octadecylsilane bonded silica gel as a stationary phase, getting 10 to 50mmol / L of sulfuric acid buffer salt in which 0.1 to 0.8% of perchloric acid is added based on a volume ratio as phase A, and getting acetonitrile as phase B, wherein volume ratio concentration of the phase B is 17 to 37%; feeding a solution prepared in step 1) to carry out gradient elution; and 3) transferring the carperitide sulfate into hydrochloride; carrying out a reverse phase HPLC (High Performance Liquid Chromatography) method; getting the octadecylsilane bonded silica gel as the stationary phase; carrying out the gradient elution; and drying to obtain the carperitide in a freezing way. The purifying method of the carperitide provided by the invention is stable and controllable in technology, high in yield, high in purity, wide in practical value and wide in application prospect.
Description
technical field [0001] The invention relates to a purification method of polypeptide medicine, in particular to a purification method of capecritide, and belongs to the field of medicinal chemistry. Background technique [0002] Acute heart failure (including acute exacerbation of chronic heart failure) Heart failure refers to a kind of overload cardiomyopathy caused by abnormal gene expression caused by insufficient energy, and finally caused by insufficient energy, myocardial ischemia, apoptosis of myocardial cells, myocardial The number of systolic units decreases, and this overload enters a vicious circle. [0003] Capecritide, trade name Caperitine, is a cycle regulating hormone composed of 28 amino acids, its molecular formula is: H-Ser-Leu-Arg-Arg-Ser-Ser-Cys-Phe-Gly-Gly -Arg-Met-Asp-Arg-IIe-Gly-Ala-Gln-Ser-Gly-Leu-Gly-Cys-Asn-Ser-Phe-Arg-Tyr-OH. As a polypeptide drug for the treatment of acute heart failure (including the exacerbation of chronic heart failure), cap...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): C07K14/58C07K1/16
Inventor 黄志云刘建马亚平袁建成
Owner HYBIO PHARMA