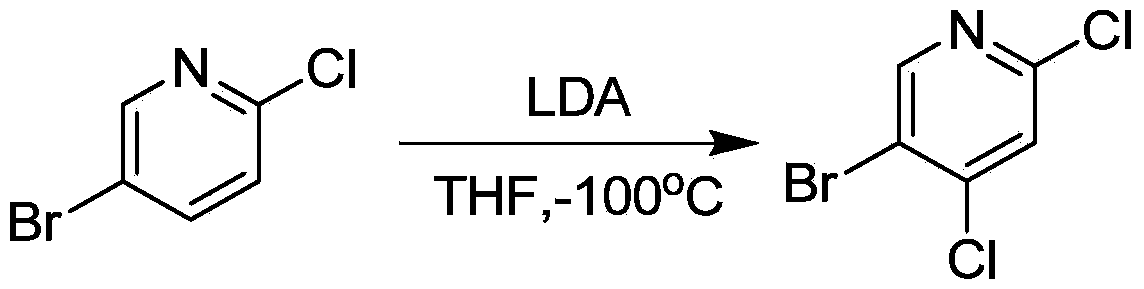
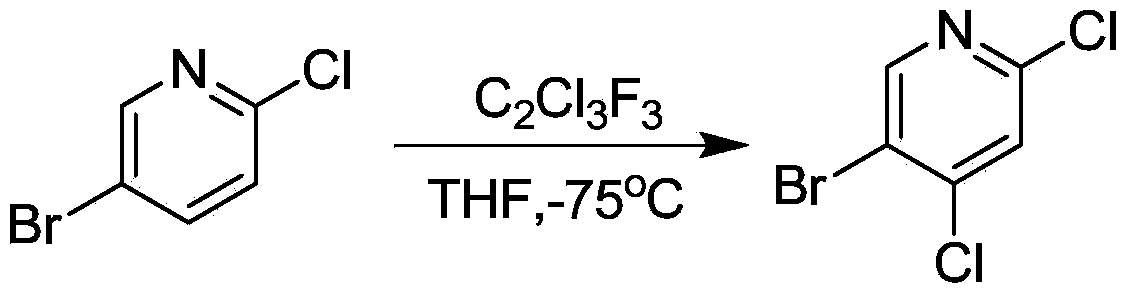
Method for synthesizing 5-Bromo-2, 4-dichloropyridine
A technology of bromopyridine and chlorpyridine, which is applied in the field of synthesis of pharmaceutical intermediates, can solve the problems of difficult industrialization, low reaction yield, serious pollution, etc., and achieve the effect of easy purification, high reaction yield and simple operation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0023] Embodiment 1: laboratory synthesis
[0024] 1. Bromination reaction
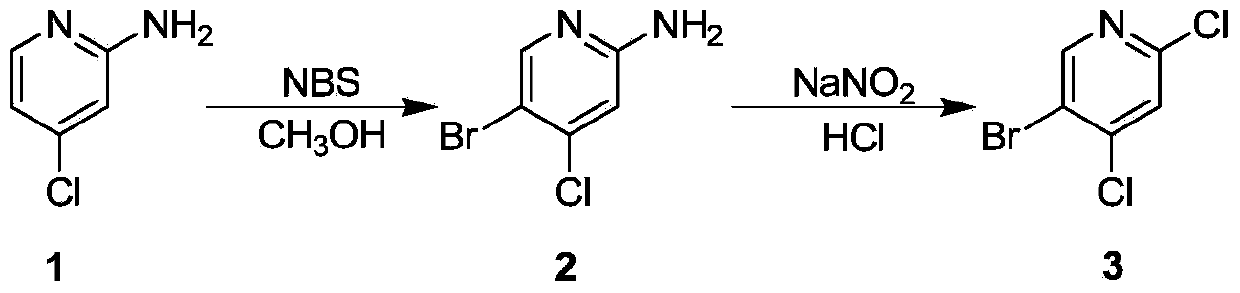
[0025] Dissolve 2-amino-4-chloropyridine (50g) in dichloromethane (500mL), cool down to 0°C, slowly add N-bromosuccinimide (76.5g) in batches, stir well after adding , reacted for 30 minutes, and TLC detected that the reaction was complete. Spin the solvent of the reaction solution to dryness first, dissolve the crude product in ethyl acetate, wash with 1mol / L hydrochloric acid, adjust the base and extract with ethyl acetate, combine the organic phases, wash with salt water, dry over sodium sulfate, and concentrate to obtain Product 2 (70g), yield 87%.
[0026] 2. Diazotization reaction
[0027] Dissolve compound 2 (10g) in concentrated hydrochloric acid (200mL) at -30°C, stir evenly, and slowly add sodium nitrite (8.3g) in batches at -30°C under temperature control. Raise the temperature, complete the addition, react at -30°C for 1 hour, add cuprous chloride, raise to room temperature, TLC detect...
Embodiment 2
[0028] Embodiment 2: small test
[0029] 1. Bromination reaction
[0030] Dissolve 2-amino-4-chloropyridine (500g) in dichloromethane (5L), cool down to 0°C, slowly add N-bromosuccinimide (765g) in batches, stir evenly after adding, and react After 30 minutes, TLC detected that the reaction was complete. Spin the solvent of the reaction solution to dryness first, dissolve the crude product in ethyl acetate, wash with 1mol / L hydrochloric acid, adjust the base and extract with ethyl acetate, combine the organic phases, wash with salt water, dry over sodium sulfate, and concentrate to obtain Product 2 (700g), yield 87%.
[0031] 2. Diazotization reaction
[0032] Dissolve compound 2 (100g) in concentrated hydrochloric acid (2L) at -30°C, stir evenly, control the temperature at -30°C and slowly add sodium nitrite (83g) in batches, there will be bubbles during the addition process, and the temperature will rise , after the addition is complete, react at -30°C for 1 hour, add cu...
Embodiment 3
[0033] Embodiment 3: pilot test
[0034] 1. Bromination reaction
[0035] Dissolve 2-amino-4-chloropyridine (5kg) in dichloromethane (50L), cool down to 0°C, slowly add N-bromosuccinimide (7.65kg) in batches, stir well after adding, The reaction was carried out for 30 minutes, and TLC detected that the reaction was complete. Spin the solvent of the reaction solution to dryness first, dissolve the crude product in ethyl acetate, wash with 1mol / L hydrochloric acid, adjust the base and extract with ethyl acetate, combine the organic phases, wash with salt water, dry over sodium sulfate, and concentrate to obtain Product 2 (7kg), the yield was 87%.
[0036] 2. Diazotization reaction
[0037] Dissolve compound 2 (1kg) in concentrated hydrochloric acid (20L) at -30°C, stir evenly, control the temperature at -30°C and slowly add sodium nitrite (830g) in batches, there will be bubbles during the addition process, and the temperature will rise , after the addition is complete, reac...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More