Aspartate of vortioxetine or hydrate thereof as well as preparation method and application thereof
A technology of aspartate and vortioxetine, which is applied in the field of medicine and can solve the problems of human injury and harm
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
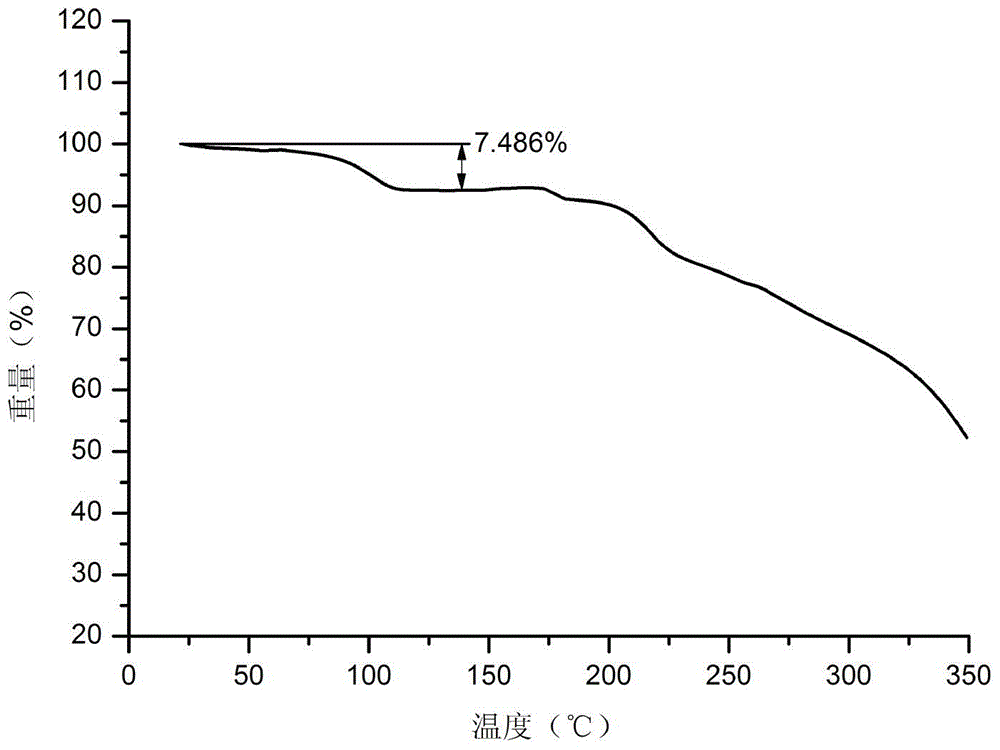
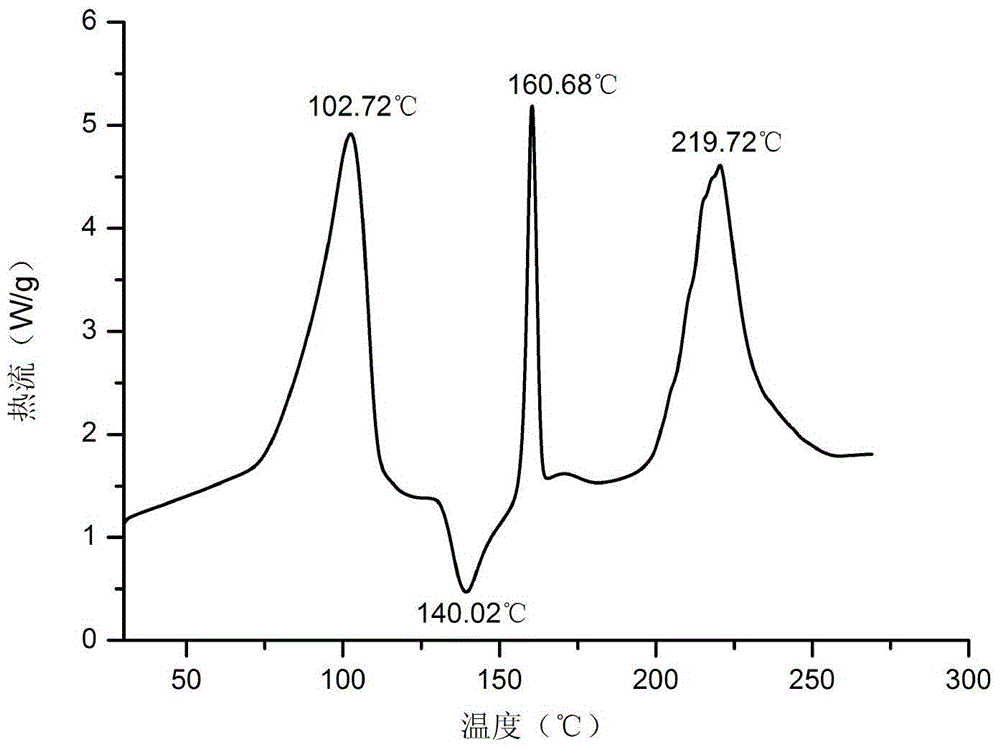
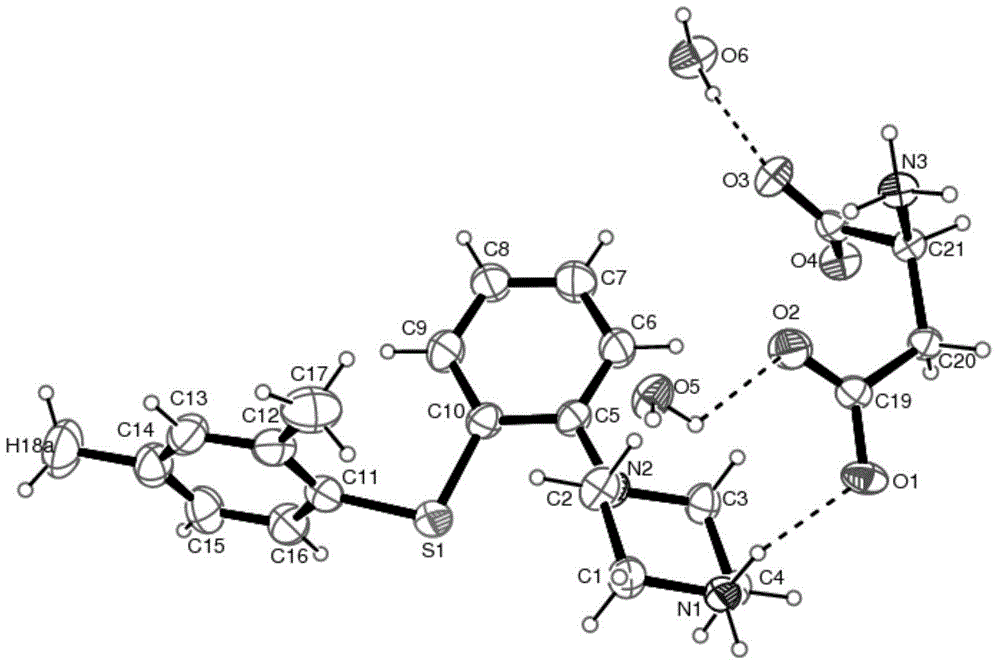
[0083] Embodiment 1: Preparation of vortioxetine aspartate dihydrate
[0084] Suspend 2.9g vortioxetine in 15ml water, heat to 80°C, add 1.3g aspartic acid, stir to dissolve, continue to stir for 2 hours, cool down naturally, and slowly precipitate white crystals after reaching room temperature, filter with suction , to obtain a white solid, put it in a vacuum drying oven (40-50° C.) and dry it to obtain vortioxetine aspartate dihydrate.
Embodiment 2
[0085] Embodiment 2: Preparation of vortioxetine aspartate dihydrate
[0086] Dissolve 15g of vortioxetine in 150ml of ethyl acetate, dissolve 6.5g of aspartic acid in 10ml of hot water and add dropwise into the vortioxetine solution, stir for 1 hour, cool down naturally, and slowly precipitate out after reaching room temperature White crystals. After 48 hours, the white solid was obtained by suction filtration, and dried in a vacuum oven (40-50° C.) to obtain vortioxetine aspartate dihydrate.
Embodiment 3
[0087] Embodiment 3: Preparation of vortioxetine aspartate dihydrate
[0088] Dissolve 2.9g of vortioxetine in 120ml of 5% ethanol aqueous solution, add 1.3g of aspartic acid, heat to 70°C, continue to stir, after dissolving, cool down naturally, and slowly precipitate white crystals after reaching room temperature, after 24 hours , filtered with suction to obtain a white solid, which was dried in a vacuum oven (40-50° C.) to obtain vortioxetine aspartate dihydrate.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More