A method for producing intestinal membrane protein powder by using enzymatic hydrolysis method for heparin to absorb residual liquid
A technology of intestinal membrane protein powder and enzymatic hydrolysis, which is applied in the direction of protein food components, protein components of waste, applications, etc., can solve problems such as poor efficacy, low protein recovery rate, and inability to exert the efficacy of intestinal membrane protein powder, and achieve good results. Social and economic benefits, increased additional income, and good feeding effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
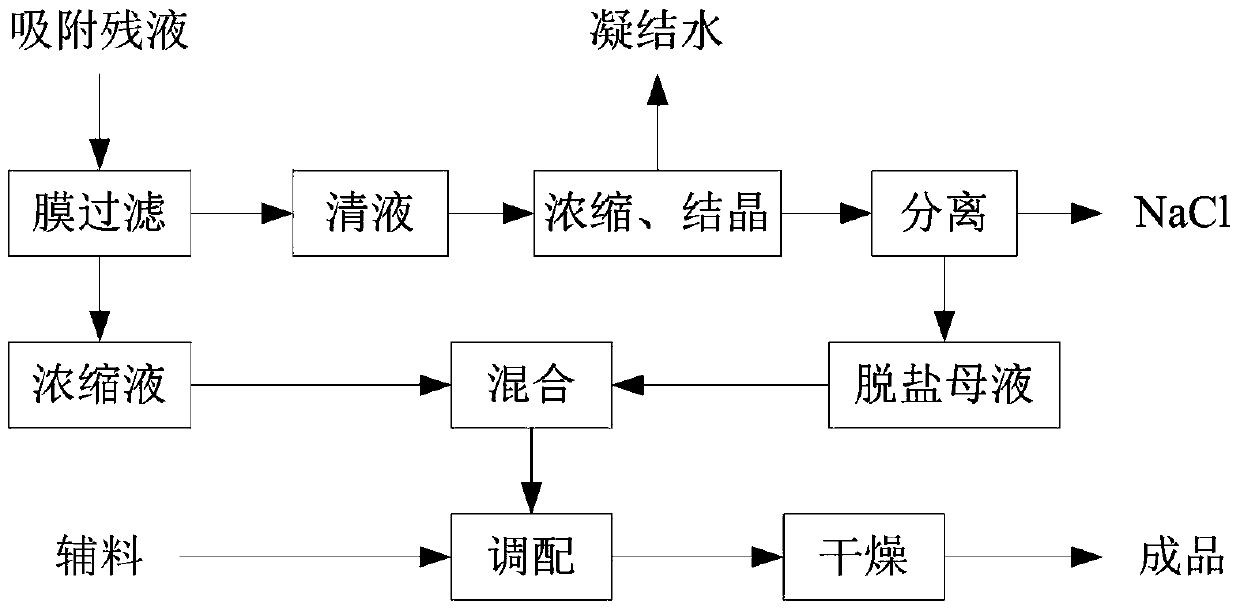
[0032] The technological process of the method for producing intestinal membrane protein powder by using enzymatic hydrolysis method heparin to absorb raffinate of the present invention is as follows figure 1 As shown, after the adsorption residual liquid is filtered through the membrane, the clear liquid and concentrated liquid are obtained. The clear liquid is evaporated, concentrated, and crystallized to separate and remove NaCl, and the remaining desalted mother liquid is mixed with the concentrated liquid of membrane filtration, and then dried to form intestinal membrane after preparation of auxiliary materials. Finished protein powder.
[0033] A sausage casing factory enzymatically hydrolyzed heparin sodium to absorb the residual liquid, with a total solid content of 4.95%, organic matter 1.97%, NaCl 2.50%, and a gray-white suspension.
[0034] Use 0.45μm microfiltration membrane to filter the heparin adsorption residual liquid in a cross-flow cycle. When the concentrat...
Embodiment 2
[0036] A sausage casing factory enzymatically hydrolyzed heparin sodium to absorb the residual liquid, with a total solid content of 4.42%, organic matter 1.28%, NaCl 2.77%, and a gray-white suspension.
[0037] Use the ultrafiltration membrane with a molecular weight cut off of 100,000 to filter the heparin adsorption residual liquid in a cross-flow cycle. When the concentration of solids in the concentrated liquid reaches 16%, the filtration is stopped to obtain a gray concentrated liquid and a transparent light yellow clear liquid; the clear liquid is first passed through four effects Evaporate and concentrate 10 times. At this time, salt particles have been precipitated. Transfer to a single-effect evaporator and continue to slowly concentrate 2.2 times at a temperature of 70-75°C (total concentration ratio is 22 times), so that NaCl crystals continue to precipitate and crystals grow. ; The concentrated material is pumped into a conical blue centrifuge while it is hot to se...
Embodiment 3
[0039] A sausage casing factory enzymatically hydrolyzed heparin sodium to absorb the residual liquid, with a total solid content of 5.82%, organic matter 2.41%, NaCl 3.25%, and a gray-white suspension.
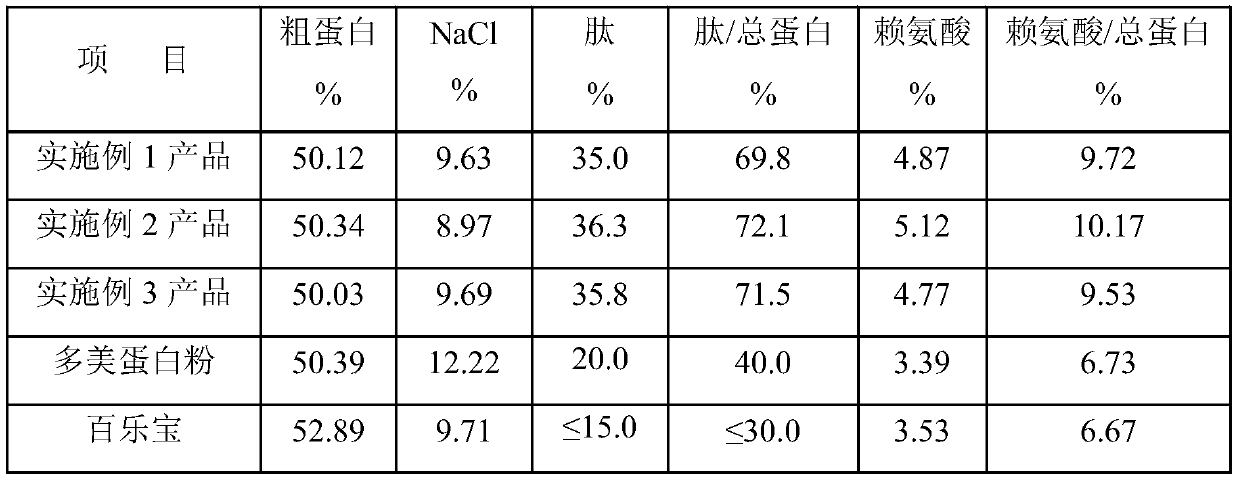
[0040] Use an ultrafiltration membrane with a molecular weight cut-off of 100,000 to filter the heparin adsorption residue in a cross-flow cycle. When the concentration of solids in the concentrated solution reaches 15.1%, the filter is stopped to obtain a gray concentrated solution and a transparent light yellow clear liquid; the clear liquid is evaporated by three effects , concentrated 14 times; the concentrated material was pumped into a decanter centrifuge while it was hot to separate NaCl crystals to obtain desalted mother liquor and NaCl crystals; washed with membrane filtrate of 1 times the quality of NaCl crystals to obtain NaCl crystals with a dry basis purity of 96%; desalted mother liquor Mix with the membrane filter concentrate, after testing, the protein content ...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap