Pharmaceutical composition for preventing and treating rhinitis and preparation method thereof
A composition and drug technology, applied in the field of medicine, can solve the problems of patients with repeated illnesses, toxic side effects, nasal mucosa damage, etc., and achieve the effect of broad application prospects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0020] The preparation of embodiment 1 ethanol extract of the present invention
[0021] Prescription: Rubia 9g, Comfrey 9g, Fangfeng 6g, Cicada slough 3g, Dilong 9g, Xu Changqing 9g, Ume 9g, Codonopsis 12g, Poria cocos 12g, Atractylodes macrocephala 12g.
[0022] Extraction process: 8 times the volume of 60% v / v ethanol aqueous solution for the above 10 medicinal flavors, reflux extraction twice, each time for 3 hours, the reflux liquid was combined to recover ethanol, concentrated, and dried to a density of 1.20-1.25g / ml (60°C) , get extract.
[0023] The above obtained extract can be further prepared to obtain hard capsules, soft capsules, granules or tablets according to conventional methods.
[0024] Prove the beneficial effects of the present invention below by drug effect experiment.
experiment example 1
[0025] Experimental Example 1 Drug efficacy experiment of the pharmaceutical composition of the present invention
[0026] 1. Test drugs and reagents
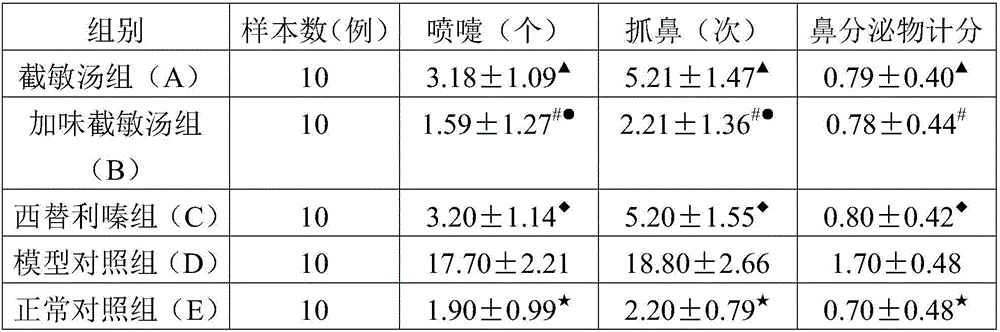
[0027] Jiemin Decoction: Formula granules are used. The composition of the prescription is: Rubia 10g, Comfrey 10g, Fangfeng 6g, Chanyi 3g, Dilong 10g, Xu Changqing 10g, Wumei 10g. The traditional Chinese medicine is dissolved in distilled water in proportion to contain 3g of crude drug per milliliter.
[0028] Modified Jiemin Decoction (that is, the pharmaceutical composition of the present invention): formula granules are adopted, and the prescription is composed of: Rubia 9g, Comfrey 9g, Fangfeng 6g, Cicada slough 3g, Dilong 9g, Xu Changqing 9g, Ume 9g, Codonopsis 12g, Poria cocos 12g, Atractylodes macrocephala 12g. The traditional Chinese medicine is dissolved in distilled water in proportion to contain 3g of crude drug per milliliter.
[0029] Cetirizine: produced by Yangzijiang Pharmaceutical Industry, 10mg / tablet. Ov...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More