MRI contrast agent taking chitosan derivative as carrier and preparation method
A technology of chitosan derivatives and contrast agents, applied in the field of medical clinical magnetic resonance imaging, can solve the problems of slow excretion of macromolecules and limit the application, and achieve the effects of easy control, high relaxation rate and good biocompatibility
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
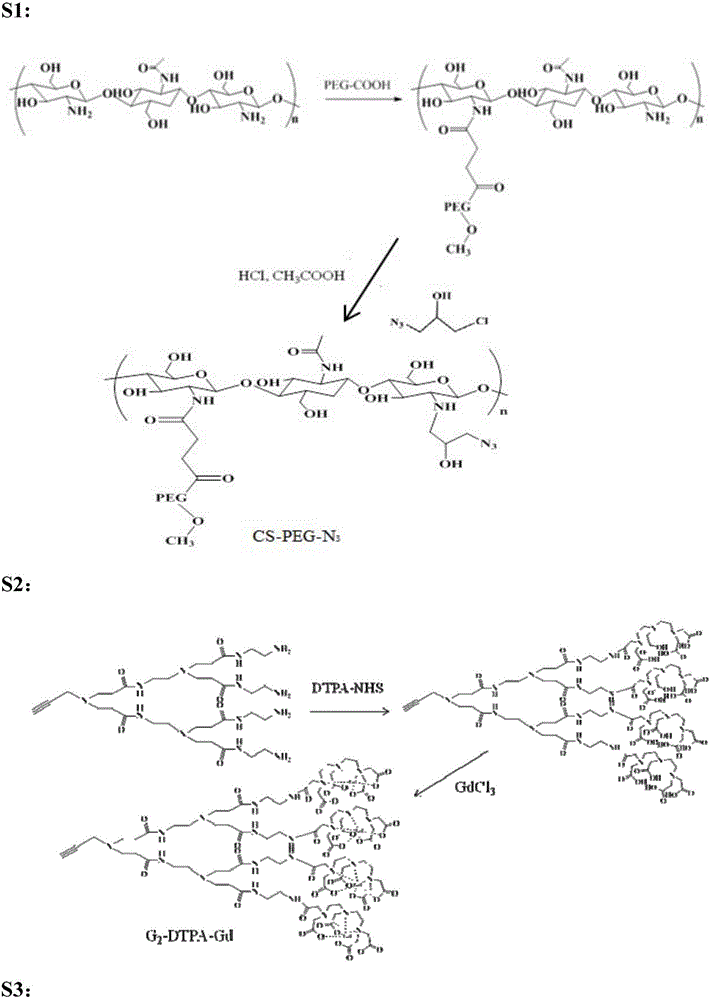
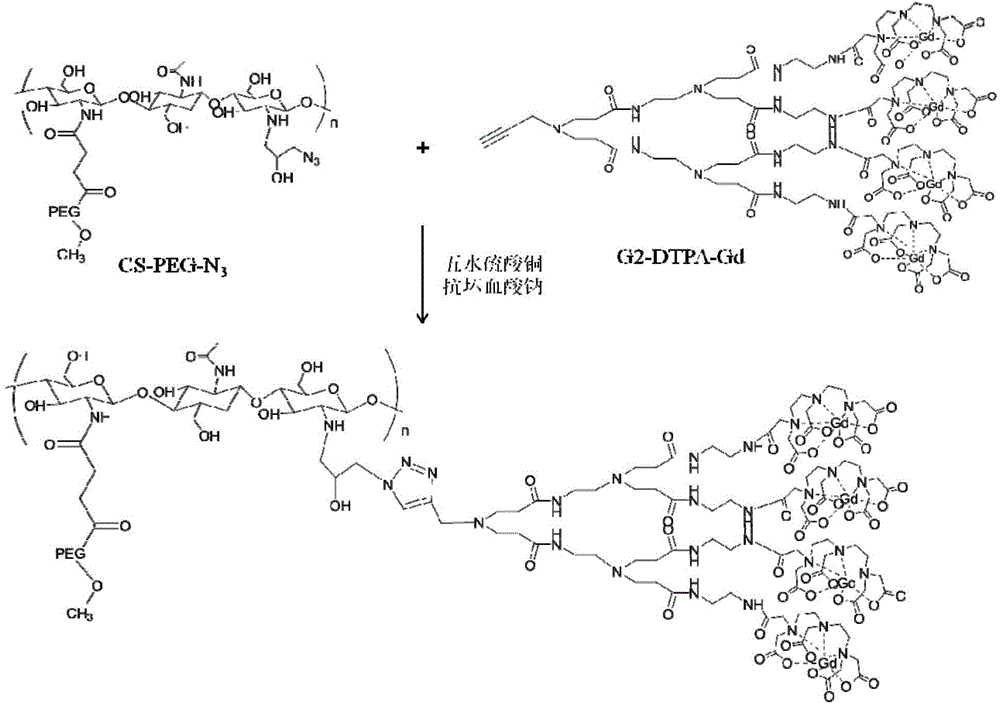
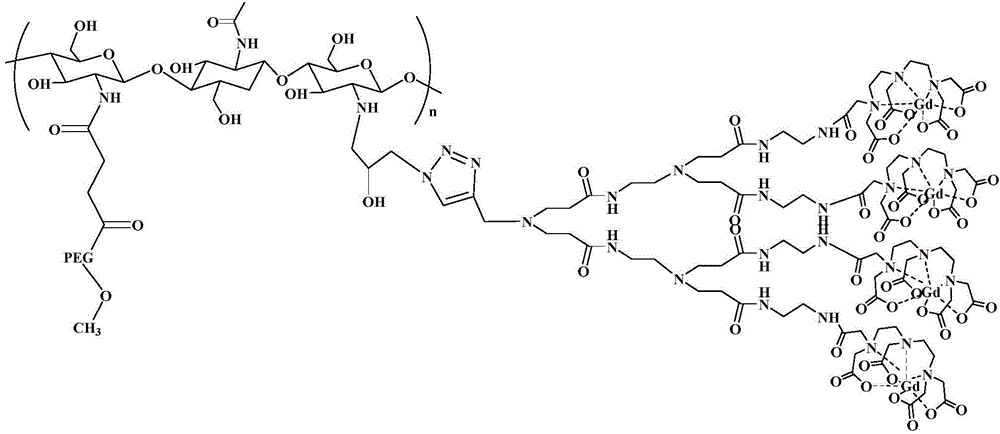
[0033] Embodiment 1 chitosan derivative is the preparation method of the MRI contrast agent of carrier, see Figure 1a , the specific steps are as follows: In an implementation case, the preparation method of the contrast agent may include the following steps:
[0034] (1) Dissolve PEG-COOH into a 5% (w / w) solution, then adjust the pH of the solution to 6, then add NHS, stir for 5 minutes and then add EDC to make the molar ratio of EDC:NHS:COO 1: 1:0.8. Stir the above solution for 30 minutes to activate the carboxyl group and slowly add it to the solution dissolved in 1% CH 3 In chitosan in COOH, the reaction solution was stirred at room temperature for 10 h. The reaction solution was dialyzed and then freeze-dried to obtain polyethylene glycol-modified chitosan (CS-PEG).
[0035] (2) Dissolving CS-PEG in an equal volume mixture of 5 wt% glacial acetic acid and hydrochloric acid, and adding azide epichlorohydrin. The reaction solution was protected from light with aluminum ...
Embodiment 2
[0039] Example 2 measures the infrared spectrograms of CS, CS-PEG, CS-PEG-N3 and CS-PEG-G2-DTPA-Gd, including: adopting the tablet method to prepare samples, and detecting chitosan raw materials, CS-PEG, CS respectively -Infrared absorption of PEG-N3 and CS-PEG-G2-DTPA-Gd, as figure 2 shown.
Embodiment 3
[0040] Example 3 Test the polymer contrast agent of the present invention and the relaxation time T of Gd-DTPA on a 0.5T MRI tester1 and T 1 Weighted imaging, including:
[0041] Prepare the above two samples with a concentration of 0.1 to 1.0 mM respectively. After the test, use the concentration as the abscissa and the reciprocal of the relaxation time as the ordinate to perform linear fitting to obtain the relaxation rates of the contrast agent of the present invention and Gd-DTPA, respectively. 9.53mM -1 ·s -1 and 4.25mM -1 ·s -1 ( image 3 ). The relaxation rate of the contrast agent of the present invention is obviously higher than that of Gd-DPTA. Through the T of both at different concentrations 1 weighted imaging ( Figure 4 ) It can be seen that both of them tend to become brighter with the increase of the concentration, but the contrast effect of the contrast agent of the present invention is obviously brighter than that of Gd-DTPA.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Relaxation rate | aaaaa | aaaaa |
| Relaxation rate | aaaaa | aaaaa |
| Molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More