An anti-H7N9 completely humanized monoclonal antibody 2L11, and a preparing method and applications thereof
A monoclonal antibody, 2L11 technology, applied in the field of immunology, can solve the problems of rimantadine drug resistance and no effective treatment methods, and achieve the effect of reducing cumbersome operations and costs, low production costs, and high affinity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0037] (1) Construction of NTH-3T3 cell line stably expressing CD40L
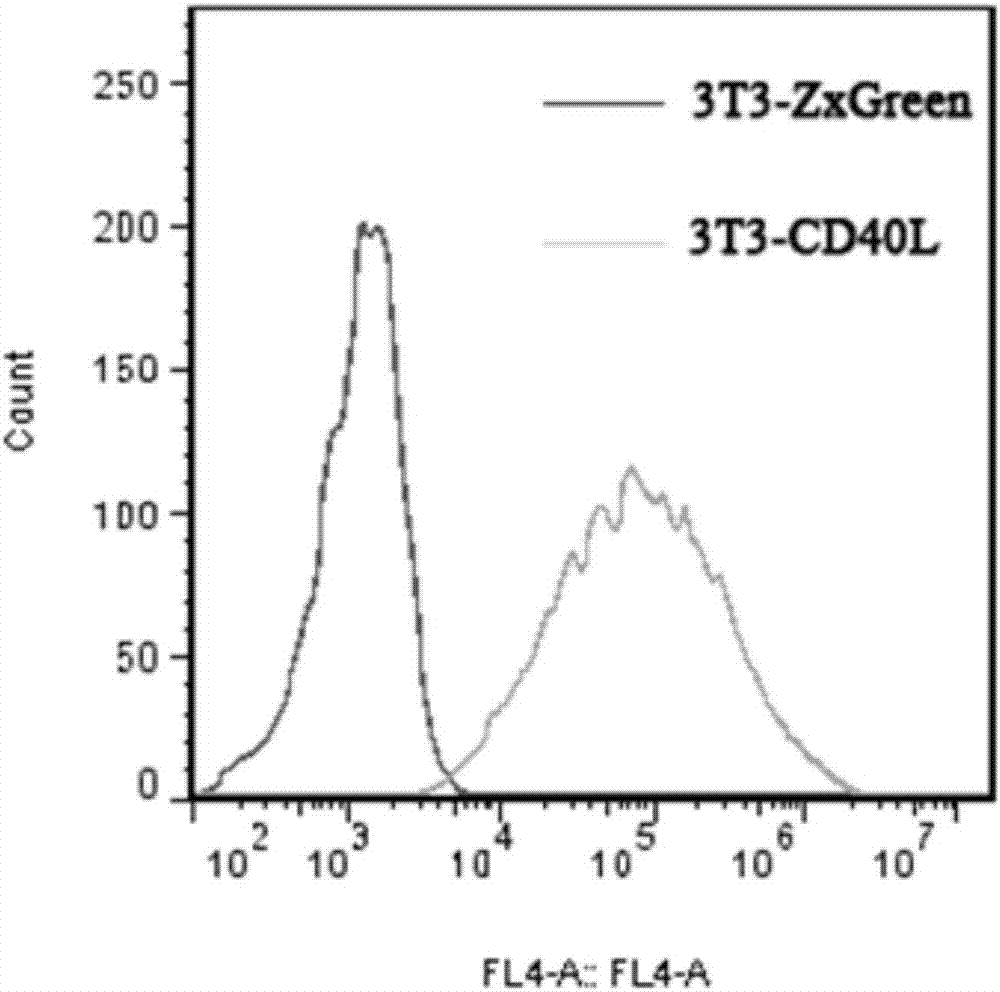
[0038]3T3-CD40L feeder cells were established using lentivirus. The lentiviral expression vector pLVX-CD40L was constructed, transfected into 293T cells, and the virus supernatant was collected on the fourth day of transfection. NIH-3T3 cells were activated, cultured for 3 generations, infected with lentivirus, continued to be cultured and passed 3 times. Use a flow cytometer to sort the cells whose FITC fluorescence intensity is near the MFI, and add them back to the culture flask at 37°C, 5% CO 2 Cultivate and detect in the incubator, and the test results are as follows: figure 1 As shown, 3T3 cells expressing CD40L and 3T3 cells transfected with empty vector pLVX (with ZxGreen) were stained with anti-CD40L with APC, and then analyzed by flow cytometry. It was found that all 3T3-CD40L feeder cells expressed CD40L. When the cells grow to 80%-90%, digest and collect the cells at a concentration of 1×10 ...
Embodiment 2
[0057] Example 2 Cloning, recombination and expression of humanized monoclonal antibody 2L11 gene
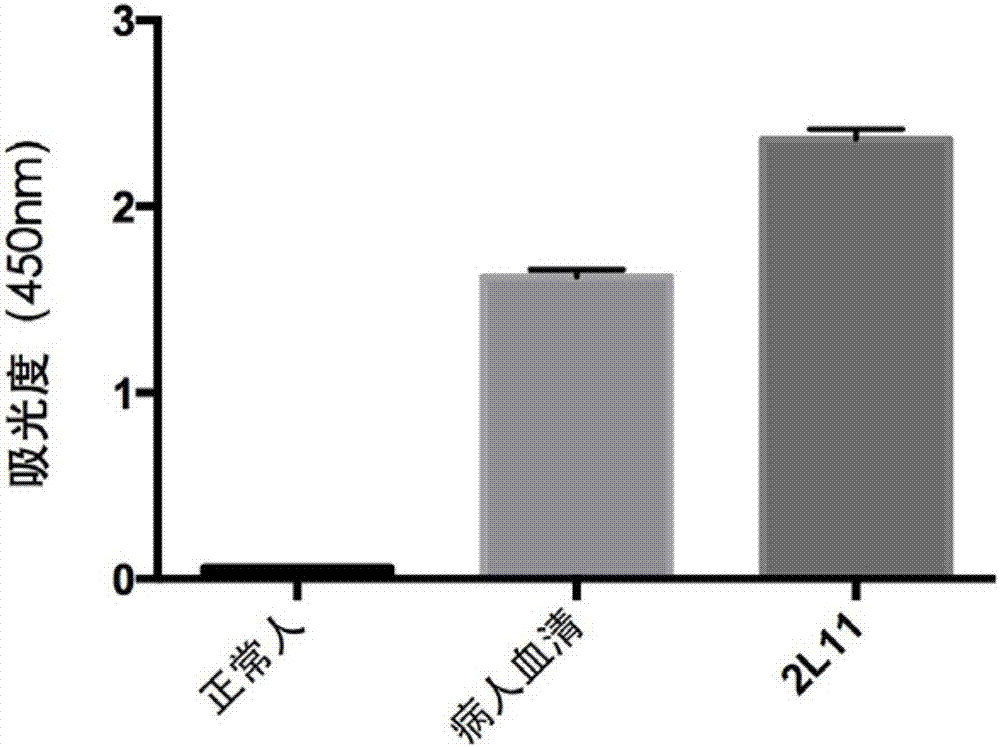
[0058] The B cells obtained in Example 1 capable of secreting antibodies binding to the H7N9 virus were lysed, and the lysate was taken for reverse transcription of RNA to obtain the PCR template cDNA of the human antibody gene. Design and synthesize primers for cloning antibody genes, clone heavy and light chain genes of antibodies using cDNA as a template, and express and purify in eukaryotic cells 293F or HEK293 recombinantly. specifically:
[0059] (1) Transfer the B cell liquid to a 96-well plate (Eppendorf, 030133366).
[0060] (2) Reverse transcription system: 150ng random primer (invitrogen, 48190-011), 0.5ul 10mM dNTP (Invitrogen, 18427-088), 1μl 0.1M DTT (Invitrogen, 18080-044), 0.5% v / v Igepal CA -630 (Sigma, I3021-50ML), 4U RNAsin (Promega), 6U Prime RNAse Inhibitor (Eppendorf) and 50U III reverse transcriptase (Invitrogen, 18080-044), add DEPC water to 14ul / well...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More