Sophora flavescens flavonoid active fraction with tyrosinase inhibitory activity and antibacterial activity as well as preparation method and application of sophora flavescens flavonoid active fraction
A technology with inhibitory activity and antibacterial activity, applied in the field of medicine, can solve the problems of green, sustainable development of the Sophora flavescens resource industry, large amount of solid waste and by-products, waste of resources, etc. The effect of high flavonoid content
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Example Embodiment
[0025] Example 1 Preparation of effective parts
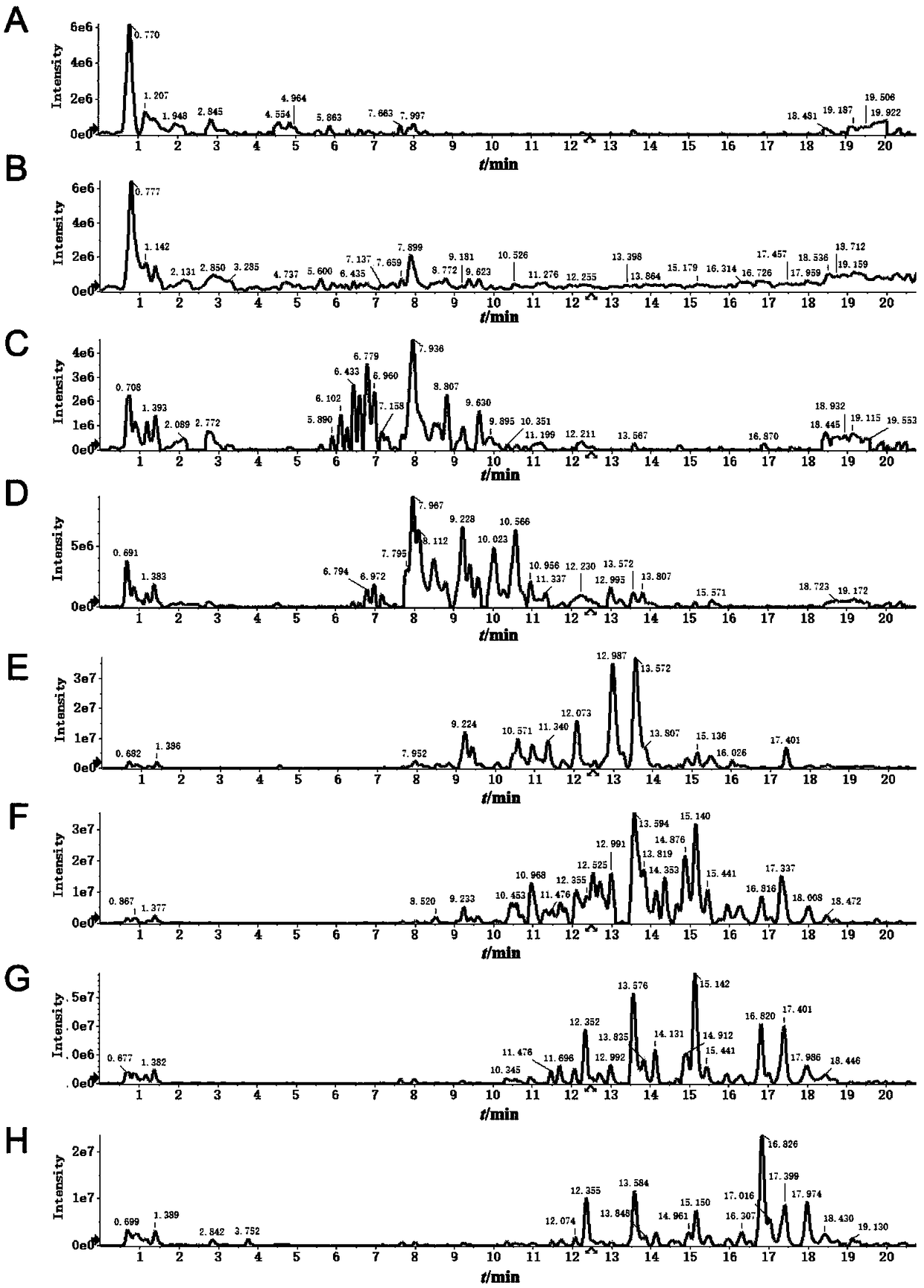
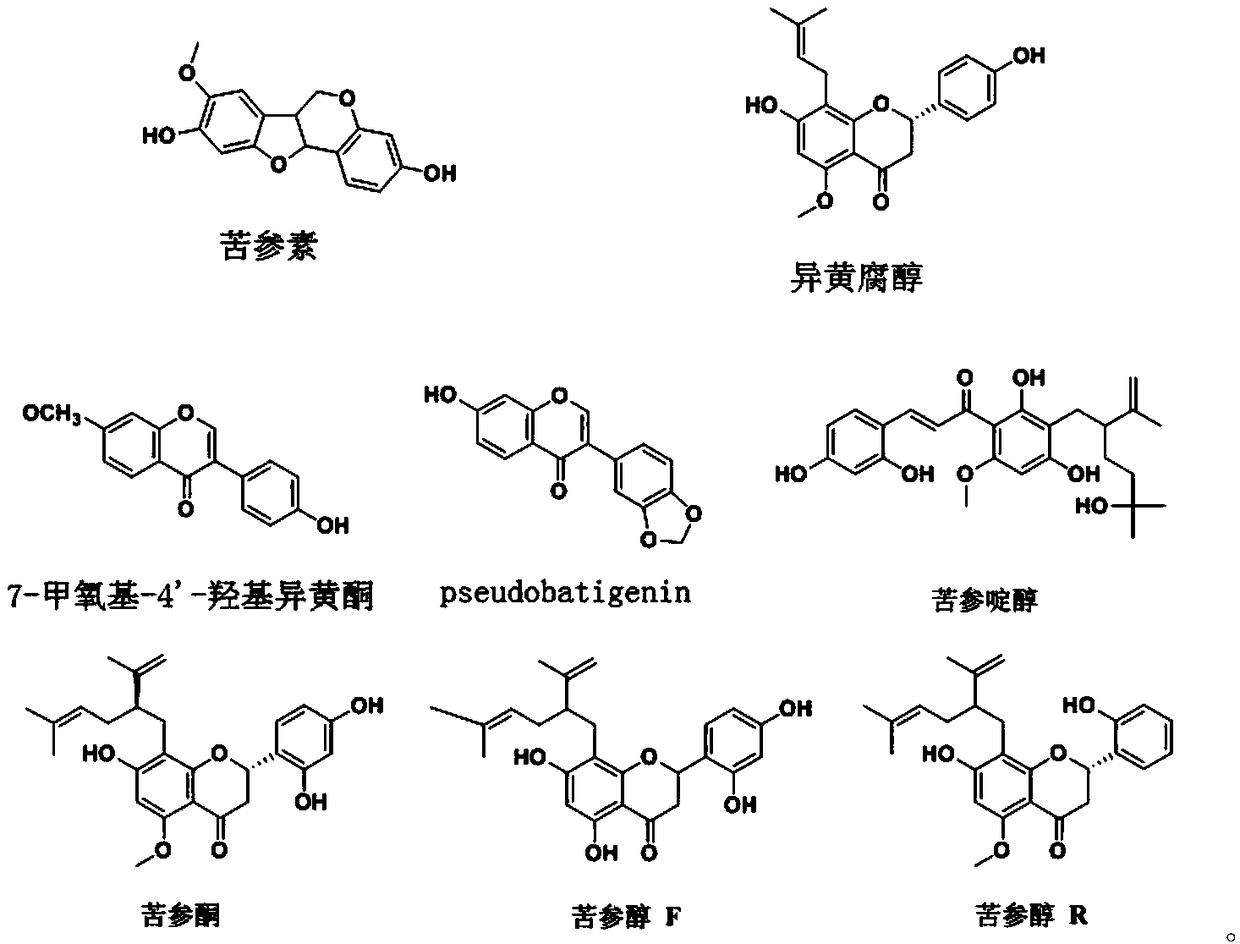
[0026] 10 kg of Sophora flavescens dregs (15 times the volume concentration of 1% acetic acid aqueous solution dialysis method to extract alkaloids Sophora flavescens dregs) was ultrasonically extracted 3 times with 8 times the amount of 80% ethanol, each time for 1 hour, and concentrated to Dry, take the dried product and dissolve it with 95% ethanol by volume, filter, concentrate the filtrate under reduced pressure, dry, then add methanol to ultrasonically dissolve, and then apply the macroporous resin AB-8 chromatography column, using 8 column volumes with a volume concentration of 10 %, 20%, 30%, 40%, 50%, 60%, 70% and 80% ethanol were eluted in sequence, and each elution site was collected.
[0027] The present invention uses the following method to determine the flavonoid content of the eluate with different volume concentrations of ethanol (10% to 80%).
[0028] UPLC-TOF-MS / MS detection conditions:
[0029] Instrument: AB Sciex...
Example Embodiment
[0043] Example 2 Evaluation of tyrosinase inhibitory activity of effective parts of Sophora flavescens dregs extract
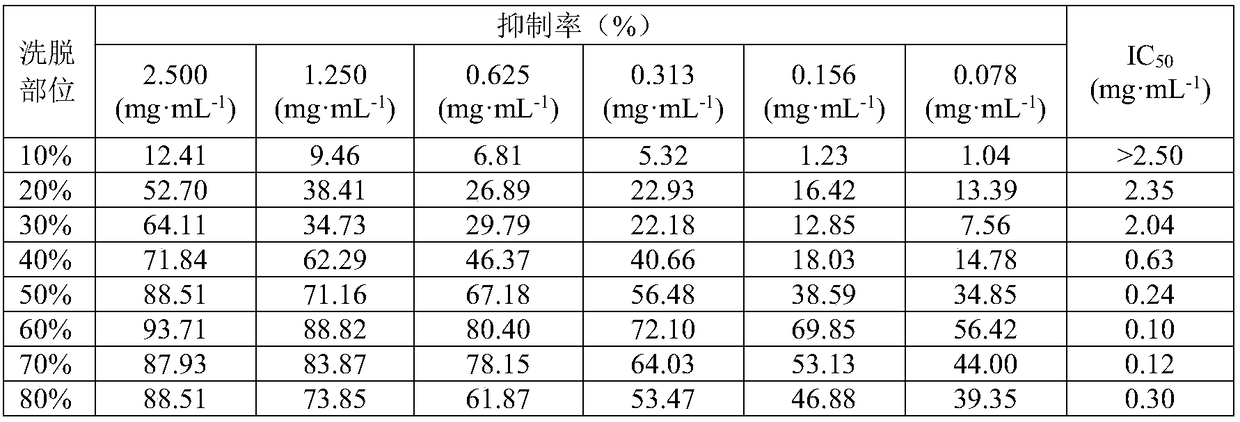
[0044] Take the 10-80% ethanol eluate prepared in Example 1, respectively, dissolve them in methanol, first prepare a 5mg / mL solution, and then dilute to 2.500, 1.250, 0.625, 0.312, 0.156, 0.078mg·mL -1 Six concentrations. Prepare 5mg·mL -1 L-tyrosine solution, 0.1mol / L phosphate buffer, 15mmol / L L-tyrosine solution, 0.25mg·mL -1 Tyrosinase and other solutions. Put each group of reaction solution in a test tube, after constant temperature in a 37℃ water bath for 10 minutes, add 0.50 mL of L-tyrosine solution, react for 15 minutes, quickly move it into a cuvette, measure the absorbance of each group at 475nm, and calculate Inhibition rate of tyrosinase by extracts of different polar parts.
[0045] Enzyme activity inhibition rate=[(A2-A1)-(B2-B1)] / (A2-A1)×100%
[0046] A1 is the absorption value without inhibitor at 0 min; A2 is the absorption value without inhibito...
Example Embodiment
[0051] Example 3 Evaluation of antibacterial activity of effective parts of Sophora flavescens medicine residue extract
[0052] Take the 10-80% ethanol eluate prepared in Example 1, respectively, dissolve them in methanol, first prepare a 5mg / mL solution, and then dilute to 2.500, 1.250, 0.625, 0.312, 0.156, 0.078mg·mL -1 Six concentrations. The qualitative filter paper is made into a 6mm diameter disc with a hole punch, and placed in a dry petri dish. After autoclaving at 121°C for 20 minutes, the filter paper is taken and placed in the sterilized petri dish. Add 5μL of liquid medicine dropwise to each piece of paper, and place the solvent in a drying box to evaporate the solvent for later use. Use a sterile cotton swab to evenly smear the diluted bacterial solution on the LB agar, take the sterilized paper slices in turn with sterilized tweezers, and dip them on the agar in turn. After placing them for 30 minutes, incubate them in a 37°C incubator for 24 hours Then observe th...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap