A kind of method for decomposing triazine ring residue and recovering methylhydrazine
A technology of triazine ring and methyl hydrazine, which is applied in the field of recycling and utilization of by-products of pharmaceutical intermediates, can solve the problems of difficulty in handling, large amount of by-products, low product yield and the like, and achieves the effects of low cost and easy handling.
Active Publication Date: 2022-06-28
SHANDONG HUIHAI PHARMA & CHEM
View PDF3 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0003] At present, the main problem of the triazine ring in the industrial production process is the low yield of the product, and the yield of the triazine ring can reach 71%. The yield is not high, and the amount of by-products produced during the synthesis process is large and difficult to handle. Under the tense environmental protection situation, it is imperative to effectively recycle and treat triazine ring residues
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0020] Add 30 g of triazine ring residue, 300 g of water, and 2 g of copper chloride to the reaction flask, turn on stirring, heat up to 130 ° C and keep the temperature for 2 h. of aqueous solution.
Embodiment 2
[0022] Add 30g of triazine ring residue, 400g of water, and 6g of copper chloride to the reaction flask, turn on stirring, heat up to 110°C and keep the temperature for 4h. aqueous solution.
Embodiment 3
[0024] Add 30 g of triazine ring residue, 900 g of water, and 1.5 g of copper sulfate to the reaction flask, turn on stirring, heat up to 90 ° C and keep the temperature for 5 h. aqueous solution.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
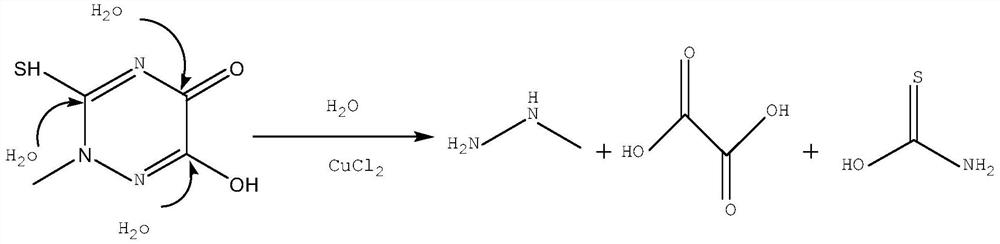
The invention discloses a method for decomposing and recovering methylhydrazine residues of triazine rings. Adding residues of triazine rings, water and copper salt into a reaction bottle, starting stirring, raising the temperature to 80-130°C for 2-5 hours, and following the reaction Carry out, extract distillate fraction from reaction bottle, distill distillate after distillate adds alkali, distill out composition and be the mixture of methylhydrazine and water. The present invention hydrolyzes the by-product triazine ring residue to recover methylhydrazine and is used for the synthesis of 2-methylthiosemicarbazide, further synthesizing the triazine ring, utilizing waste resources, solving the problem that the industrialization of the triazine ring residue is difficult to handle, and realizing The triazine ring residue is recycled to methylhydrazine and then to the triazine ring. No solvent is used, which eliminates the disadvantages of flammability, explosion and high cost of organic solvents; the raw material is only the triazine ring residue reacts with water, and the copper salt is used as a catalyst to recover and produce methylhydrazine, and hydrolyze the triazine ring residue into small Molecular salt, easy to handle, safe and environmentally friendly, low cost, suitable for industrial production.
Description
technical field [0001] The invention belongs to the field of recycling and utilization of by-products of pharmaceutical intermediates, and in particular relates to a method for decomposing and recovering methylhydrazine from triazine ring residues. Background technique [0002] Methylhydrazine is an important raw material for the synthesis of 2-methylthiosemicarbazide, and 2-methylthiosemicarbazide is further reacted with diethyl oxalate to synthesize triazine ring. The triazine ring is a key intermediate in the synthesis of ceftriaxone sodium, and after synthesizing 7-ACT (7-amino ceftriaxone) with 7-ACA (7-amino cephalosporanic acid), and then with DAMA to synthesize ceftriaxone sodium . Ceftriaxone sodium, also known as ceftriaxone, is a landmark third-generation cephalosporin antibiotic developed by Roche, Switzerland. , mainly used for infectious diseases caused by sensitive bacteria, such as respiratory system infections (especially pneumonia), ENT infections, urinar...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Patents(China)
IPC IPC(8): C07C241/02C07C243/14C07C337/06
CPCC07C241/02C07C337/06C07C243/14
Inventor 张国辉姜福元王乐强侯绪会王亮亮付德修张莉华
Owner SHANDONG HUIHAI PHARMA & CHEM