Traditional Chinese medicine monomer for treating early endometriosis and application thereof
A technology for endometriosis and ectopic diseases, applied to medical preparations containing active ingredients, drug combinations, pharmaceutical formulas, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0052] Embodiment 1 Chinese medicine granules
[0053] The preparation method is as follows:
[0054] Take traditional Chinese medicine monomer ginsenoside Rg3 and granule auxiliary agent, and prepare according to the conventional preparation method of granule.
Embodiment 2
[0055] Embodiment 2 Chinese medicine capsule
[0056] The preparation method is as follows:
[0057] Take traditional Chinese medicine monomer ginsenoside Rg3 and capsule auxiliary agent, and prepare according to the conventional preparation method of capsule.
Embodiment 3
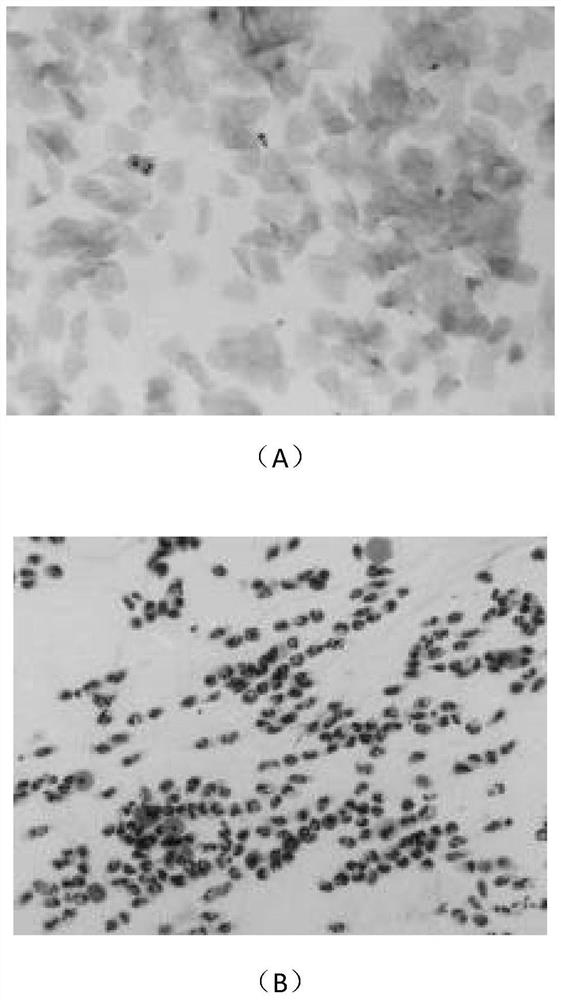
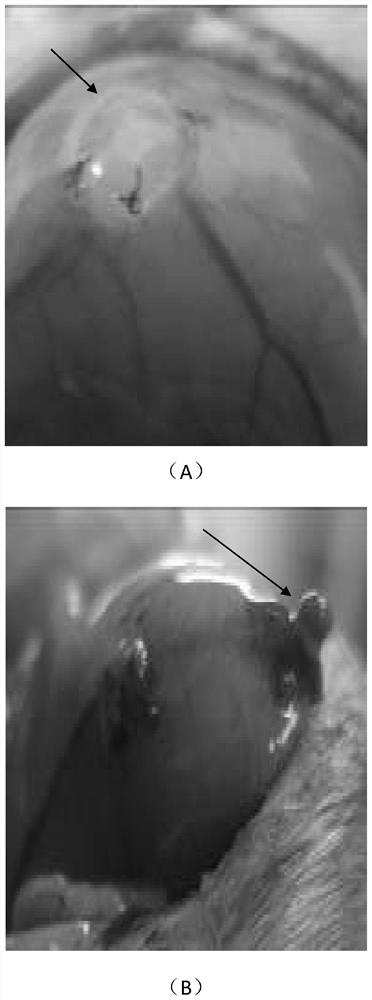
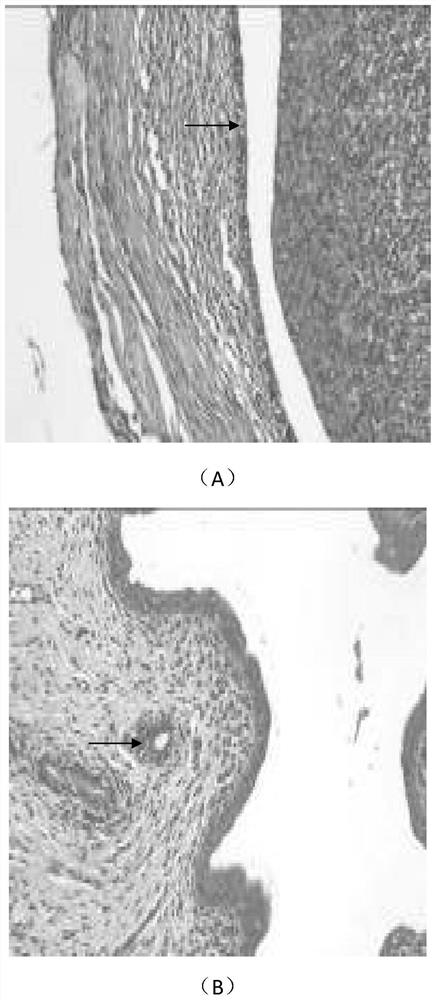
[0058] The therapeutic effect of embodiment 3 ginsenoside Rg3 on endometriosis rats
[0059] 1.1 Experimental materials
[0060] 1.1.1 Experimental animals
[0061] Healthy adult (6-7 weeks old) SPF grade female Sprague-Dawley (SD) rats (180-200g) were purchased from Shanghai Xipuer-Bikay Laboratory Animal Co., Ltd., license number: SCXK (Shanghai) 2013-0016 , certificate number: 0022017.
[0062] 1.1.2 Reagents and equipment
[0063] 1.1.2.1 Drugs:
[0064] (1) Ginsenoside Rg3 was purchased from Shanghai Shengzhong Pharmaceutical Chemical Co., Ltd.; batch number 140321; content ≥ 95%; white powder.
[0065] (2) Gestrinone was purchased from Beijing Zizhu Pharmaceutical Co., Ltd.; batch number 06001011002; content 99.9%; white powder.
[0066] 1.2 Experimental method
[0067] 1.2.1 Feeding and estrous cycle monitoring of female SD rats:
[0068] Adult female SD rats were kept in the SPF-level rodent experimental animal room of Shanghai Institute of Family Planning Scien...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More