Radix angelica pubescens traditional Chinese medicine formula granules and preparation method thereof
A technology of traditional Chinese medicine formula granules and independent living, which is applied in the direction of medical formulas, medical preparations with non-active ingredients, medical preparations containing active ingredients, etc., to achieve high yield, high content, and solve complex process effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0037] The preparation method of Duhuo traditional Chinese medicine formula granule of the present invention comprises the following steps:
[0038] Take lovage herb powder and extract it with supercritical carbon dioxide to prepare lovage extract and medicinal residue;
[0039] Using excipients to absorb the lovage extract to prepare lovage extract powder;
[0040] Adding water to the medicinal dregs, decocting, concentrating the obtained extract and adding auxiliary materials to prepare the dry extract powder of chrysalis;
[0041] Mixing the Lok Huo extract powder and the Lok Huo dry extract powder to prepare Lok Huo traditional Chinese medicine formula granules;
[0042] The excipient is selected from one or more of silicon dioxide, magnesium aluminum silicate and kaolin, and the particle size of the excipient is 1 μm to 200 μm.
[0043] Understandably, the lovage medicinal material is purified and crushed into a raw material powder with a particle size of 10-60 mesh to ...
Embodiment 1
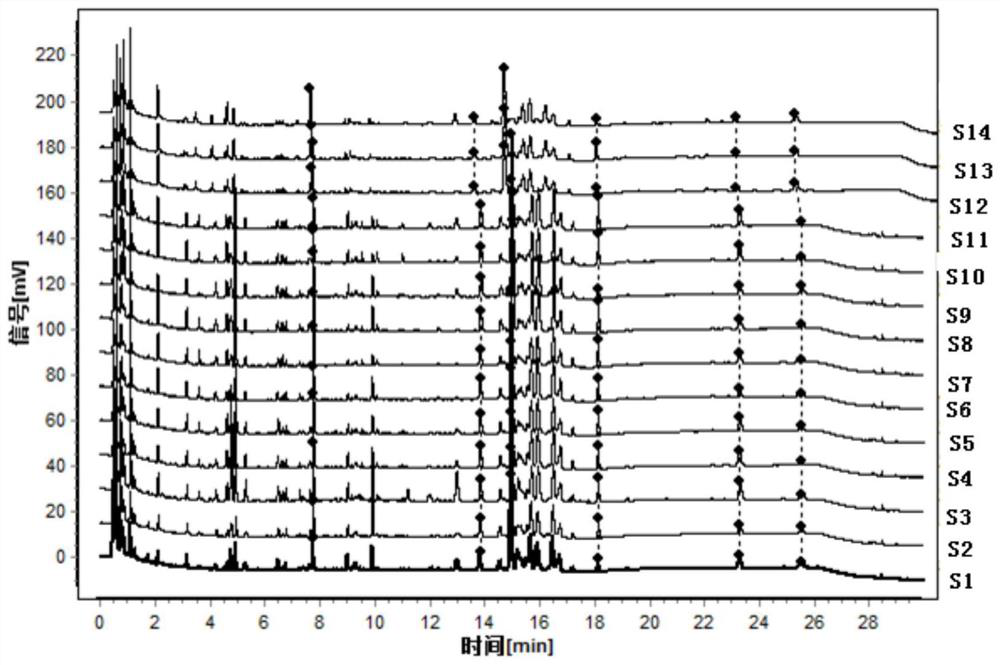
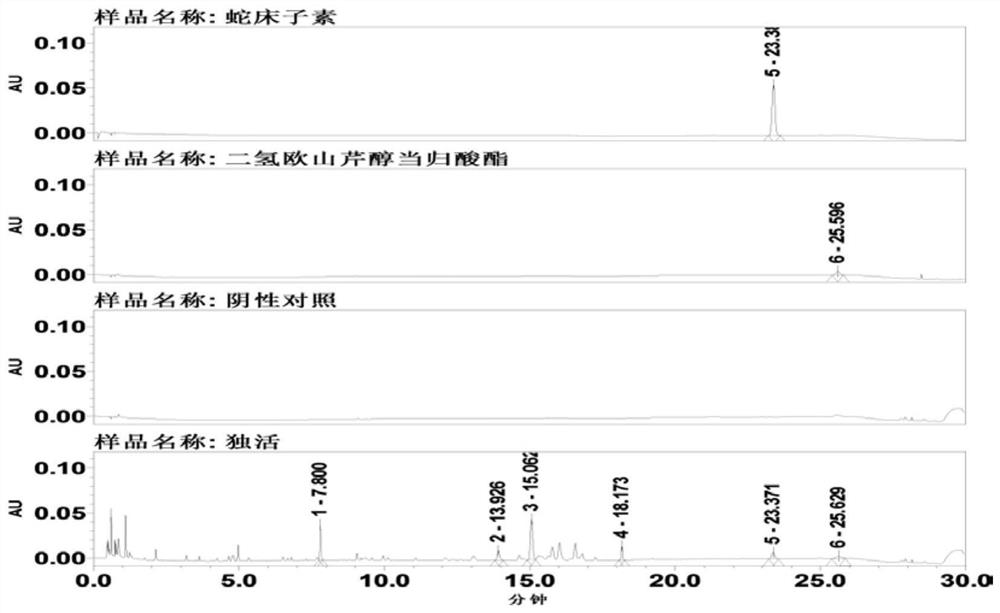
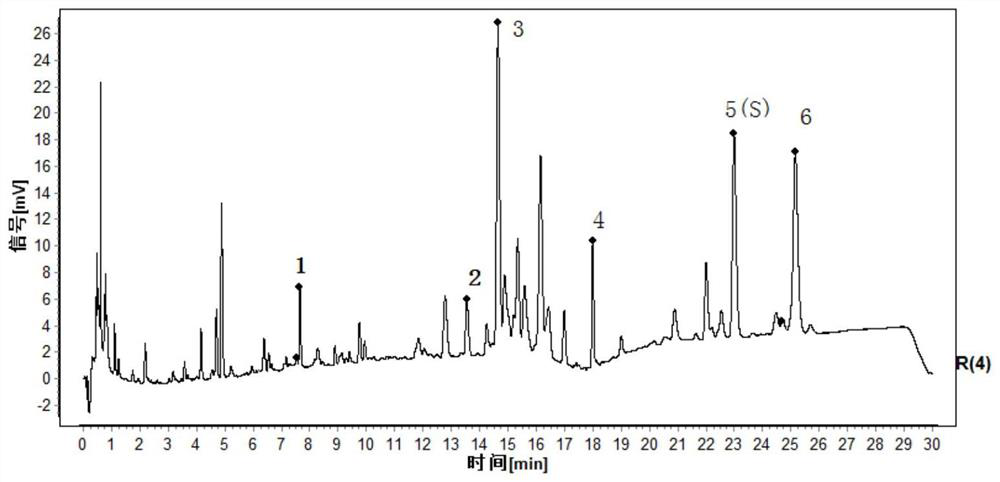
[0136] This embodiment provides a kind of Duhuo traditional Chinese medicine formula granule and its preparation method, such as Figure 6 As shown, the specific steps are as follows:
[0137] Take 1000 g of the lovage medicinal material, remove impurities, and crush it into a 10-mesh lovage medicinal material powder with an average particle diameter. Send the lovage herb powder into a supercritical extraction kettle, use ethanol as an entrainer, input carbon dioxide fluid into the kettle, extract at an extraction pressure of 35MPa, and an extraction temperature of 65°C for an extraction time of 1.0 hour, and then extract the carbon dioxide in the extraction kettle The fluid is introduced into a supercritical separation kettle, and separated at a separation pressure of 10 MPa and a separation temperature of 60° C. for a separation time of 1.0 hour. The carbon dioxide is separated to obtain 27.2 g of a semi-solid oily cloaca extract and about 972 g of medicinal residues.
[0138...
Embodiment 2
[0149] This example provides a solitary traditional Chinese medicine formula granule and its preparation method, the preparation method of which is basically the same as that of Example 1, the only difference is that the excipient is silicon dioxide with a particle size of 1 μm to 100 μm, and the specific steps are as follows:
[0150] Take 1000 g of the lovage medicinal material, remove impurities, and crush it into a 10-mesh lovage medicinal material powder with an average particle diameter. Send the lovage herb powder into a supercritical extraction kettle, use ethanol as an entrainer, input carbon dioxide fluid into the kettle, extract at an extraction pressure of 35MPa, and an extraction temperature of 65°C for an extraction time of 1.0 hour, and then extract the carbon dioxide in the extraction kettle The fluid is introduced into a supercritical separation kettle, and separated at a separation pressure of 10 MPa and a separation temperature of 60° C. for a separation time...
PUM
| Property | Measurement | Unit |
|---|---|---|
| particle diameter | aaaaa | aaaaa |
| particle diameter | aaaaa | aaaaa |
| particle diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap