A cryopreservation method and recovery method without ice crystals in a supercooled state
A low-temperature preservation and state-of-the-art technology, applied in the field of low-temperature medicine, can solve problems affecting cell differentiation, uneven heat diffusion, and cell damage, and achieve the effects of large density and cell mass, inhibition of ice crystal formation, and high survival rate
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0049] The present embodiment provides a method for low-temperature storage without ice crystals in a supercooled state, which includes the following steps:
[0050] S1. Select UW solution as cryoprotectant solution.
[0051] S2. Acquiring human cells and tissues.
[0052] S3. Suspend human cells and tissues with cryoprotectant solution pre-cooled to 4°C, pour into a 15ml centrifuge tube, and make the cell density reach 1×10 6 / ml.
[0053] S4. Inject an appropriate amount of light chain mineral oil. It can be seen that the light chain mineral oil floats on the liquid surface of the cryoprotectant solution and gradually completely covers the liquid surface to form a sample. Air bubbles should be avoided during the operation.
[0054] S5. Place the sample at a temperature pre-cooled to 4°C for temperature balance for 15 minutes, and then quickly place it at a temperature pre-cooled to -16°C for 24 hours in a supercooled state.
Embodiment 2
[0056] The present embodiment provides a method for low-temperature storage without ice crystals in a supercooled state, which includes the following steps:
[0057] S1. Select DMEM medium containing 10% fetal bovine serum as cryoprotectant solution.
[0058] S2. Acquiring human cells and tissues.
[0059] S3. Suspend human cells and tissues with cryoprotectant solution, pour them into a 15ml centrifuge tube, and make the cell density reach 5×10 6 / ml.
[0060] S4. Inject an appropriate amount of olive oil. It can be seen that the olive oil floats on the liquid surface of the cryoprotectant liquid and gradually completely covers the liquid surface to form a sample. Air bubbles should be avoided during the operation.
[0061] S5. Place the sample at a temperature pre-cooled to 4°C for temperature balance for 20 minutes, and then quickly place it at a temperature pre-cooled to -16°C for cryopreservation in a supercooled state.
[0062] S6. Place the sample at a temperature of...
Embodiment 3
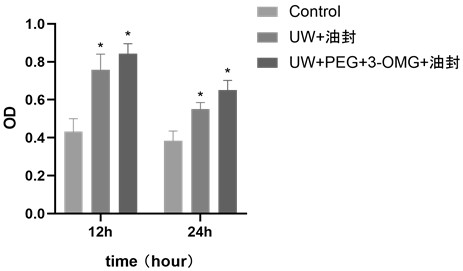
[0064] This example provides a cryopreservation method without ice crystals in a supercooled state, which is basically the same as Example 1, the difference is that the cryoprotectant solution in this application includes UW solution, 5% PEG and 0.2mol / L 3-OMG .
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More