Preparation of libraries of protein variants expressed in eukaryotic cells and use for selecting binding molecules
a technology of protein variants and libraries, applied in the field of protein variant libraries expressed in eukaryotic cells and selection of binding molecules, can solve the problems of limited ability to screen the output, inability to resolve the extent of expression and the binding affinity of resultant clones, and limitations of the prokaryotic display system including phage display
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Construction of Vectors for Expression of IgG Formatted Antibodies
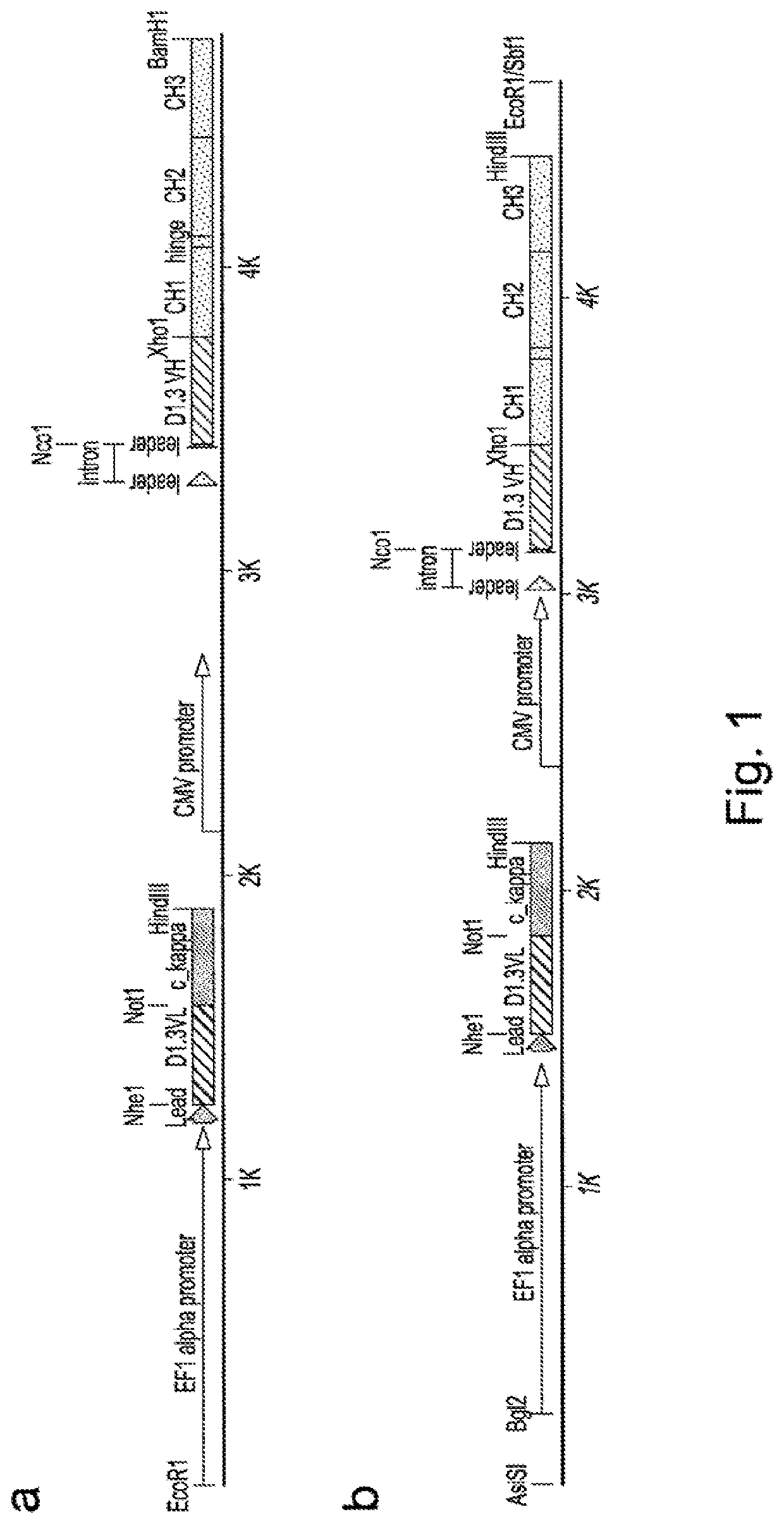
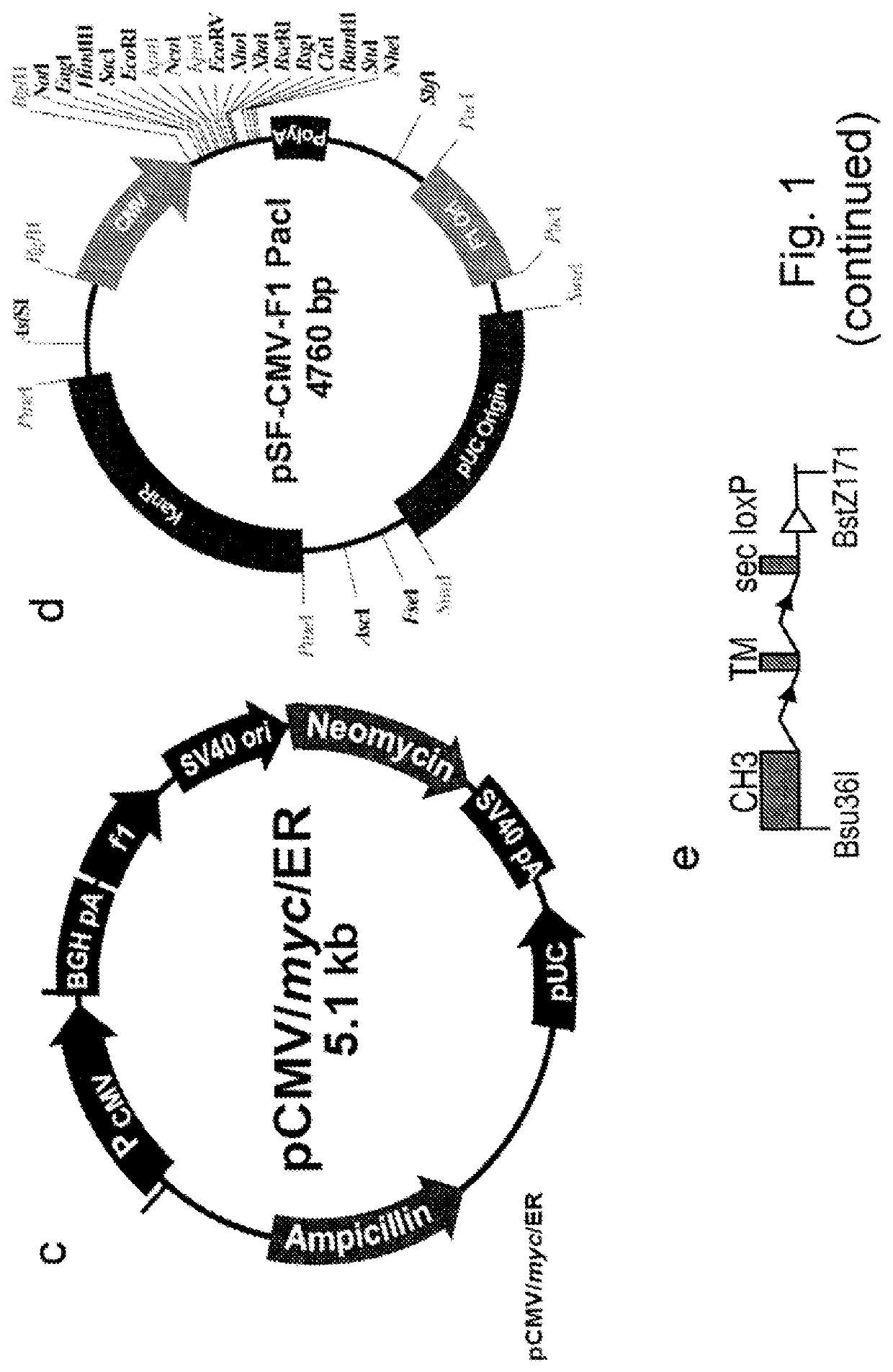
[0286]To effect genetic selections of binders (e.g. antibody, protein or peptide) it is necessary to introduce a gene encoding this binder and to drive expression of this gene from an exogenous promoter, or by directing integration of the transgene downstream of a promoter pre-existing in the cellular DNA, e.g., an endogenous promoter. Antibodies represent the most commonly used class of binders and they can be formatted for expression in different forms. In examples below, we describe expression of a single gene format where a scFv is fused to a Fc domain (scFv-Fc). We also exemplify expression of antibodies formatted as human IgG2 molecules. To express IgG or FAb formatted antibodies in producer cells such as higher eukaryotes, it is necessary to express the separate heavy and light chains. This can be done by introducing separate plasmids encoding each chain or by introducing them on a single plasmid. Within a sing...
example 2
Construction of Vector (pD2) for Targeting an Antibody Cassette to the AAVS Locus
[0293]Cleavage within the genome using site-specific nucleases facilitates the insertion of heterologous DNA through homologous recombination or non-homologous end joining (NHEJ). Human HEK293 cells were cleaved with nucleases targeting the first intron of the protein phosphatase 1, regulatory subunit 12C (PPP1R12C) gene. This locus was identified as a common integration site of adeno-associated virus and is referred to as the AAVS site (FIG. 3a). The AAVS site is considered a “safe harbour” locus for insertion and expression of heterologous genes in human cells [124].
[0294]Following site-specific cleavage within the genome it is possible to promote integration of a protein expressing cassette using homologous recombination. To do this it is necessary to flank the expression cassette with regions homologous to the sequences found on either side of the genomic cleavage site. To direct integration into th...
example 3
AAVS TALEN-Directed Integration of IgG Construct for Cell Surface Antibody Expression and Antigen Binding
[0297]HEK293F cells (Life Technologies), grown in Freestyle medium were transfected with pD2-D1.3 DNA in the presence or absence of an AAVS directed TALEN vector pair. An AAVS TALEN pair (“AAVS original”) was previously described [125] and recognises the sequence:
LEFT TALEN:(SEQ ID NO: 70)5′ (T)CCCCTCCACCCCACAGT(SEQ ID NO: 71)Spacer 5′ GGGGCCACTAGGGACRight TALEN: (SEQ ID NO: 72)complement of 5′ AGGATTGGTGACAGAAAA(SEQ ID NO: 73)(i.e. 5′ TTTTCTGTCACCAATCCT
[0298]An alternative, more efficient AAVS targeted TALEN pair was identified and used in later experiments (pZT-AAVS1 L1 TALE-N and pZT-AAVS1 R1 TALE, Cat No GE601A-1 System Biosciences). This pair, which recognises the same site (but not the first “T” residue shown in brackets above), are referred to as the “AAVS-SBI” TALEN pair.
[0299]Cells were seeded at 0.5×106 cells / ml and transfected next day at 106 cells / ml using DNA:polyeth...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More