Predicting immunogenicity of t cell epitopes
a technology of t cell epitopes and immunogenicity, which is applied in the field of methods for predicting t cell epitopes, can solve the problems of not being able to predict no algorithm has been put forth that is capable of predicting with a high degree of certainty, and the accuracy of sequence-based methods is still far behind
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
ing a Model for Predicting Immunogenicity of T Cell Epitopes
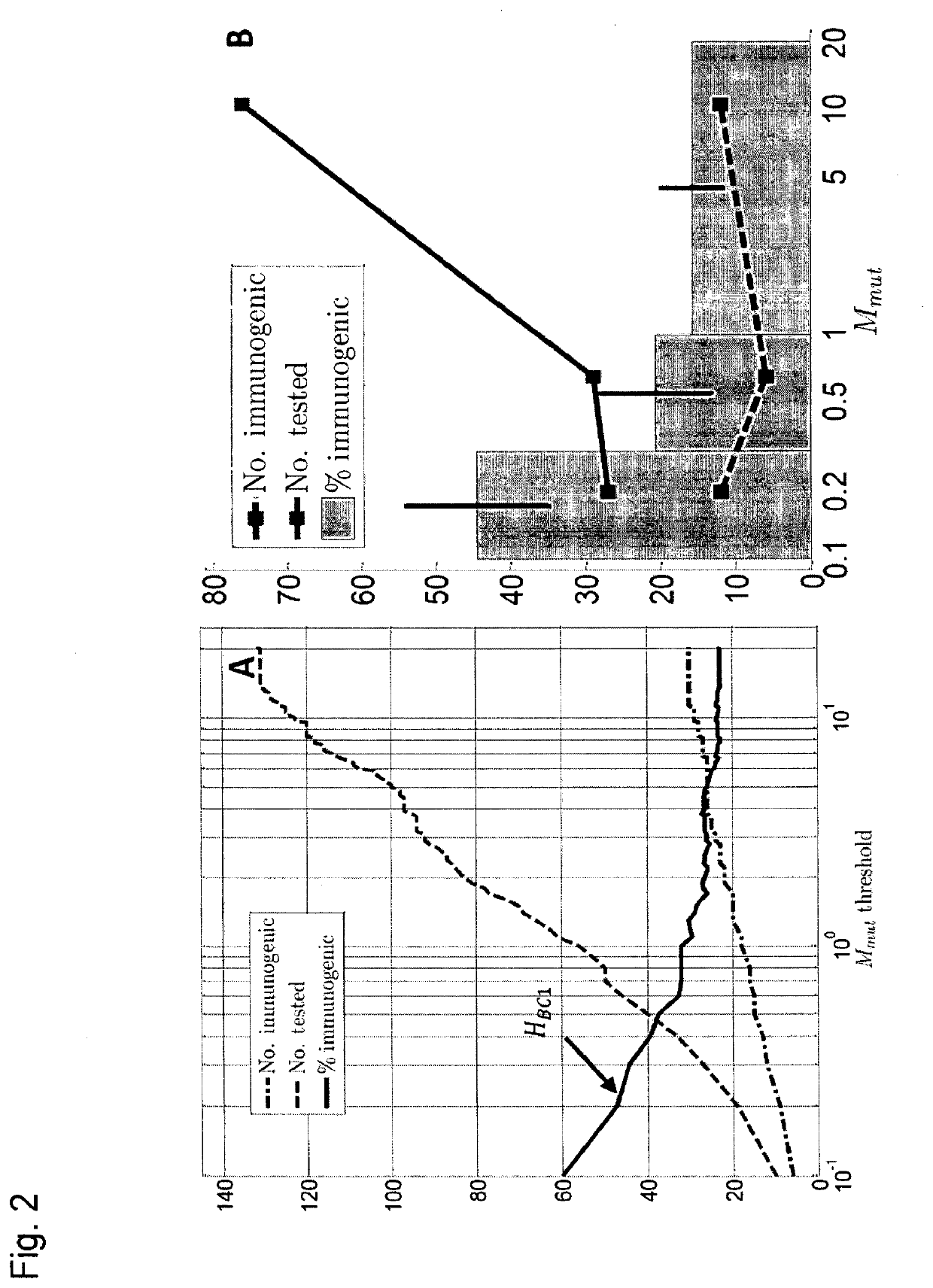
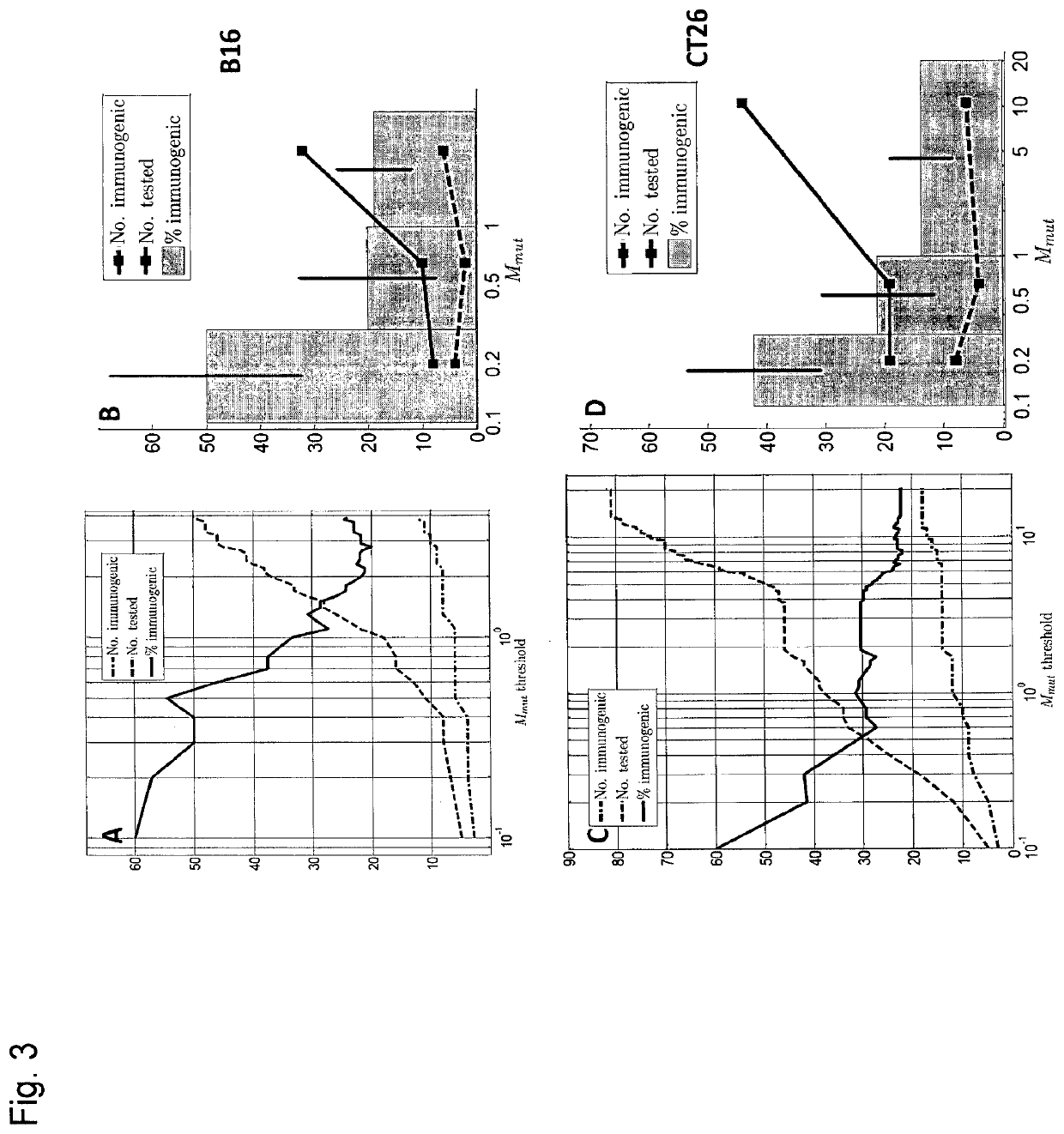
[0270]Previously we explored the immunogenicity of 50 somatic mutations identified in the B16F10 murine melanoma cell line (J. C. Castle et al., Exploiting the mutanome for tumor vaccination. Cancer Research 72, 1081 (2012)). These 50 mutations were selected from a pool of 563 expressed nonsynonymous somatic mutations primarily to maximize MHC class I expression (J. C. Castle et al., Exploiting the mutanome for tumor vaccination. Cancer Research 72, 1081 (2012)) (see also Example 2). For each mutation we predicted the minimal epitope. i.e., the epitope scoring the lowest MHC class I consensus score (Y. Kim et al., Nucleic Acids Research 40, W525 (2012)) (defined here as Mmut) when searching the space of all possible MHC class I alleles, potential epitope lengths and sequence windows (where to position the mutation) (J. C. Castle et al., Exploiting the mutanome for tumor vaccination. Cancer Research 72, 1081 (2012)). Measuri...
example 2
and Methods
[0293]The materials and methods used in Example 1 are described below:
[0294]Animals
[0295]C57BL / 6J and Balb / cJ mice (CRL) were kept in accordance with federal and state policies on animal research at the University of Mainz.
[0296]Cells for Melanoma and Colorectal Murine Tumor Model
[0297]B16F10 melanoma cell line (Product: ATCC CRL-6475. Lot Number: 58078645) and CT26.WT colon carcinoma cell line (Product: ATCC CRL-2638. Lot Number: 58494154) were purchased in 2010 from the American Type Culture Collection. Early (3rd, 4th) passages of cells were used for sequencing experiments. Cells were routinely tested for Mycoplasma. Re-authentication of cells has not been performed since receipt. MZ7-MEL cell line (established January 1988) and an autologous Epstein-Barr vims-transformed B cell line were obtained from Dr. Thomas Wölfel (Department of Medicine. Hematology Oncology. Johannes Gutenberg University).
[0298]Synthetic Peptides
[0299]Peptides were purchased from Jerini Peptide ...
example 3
f a Scheme for Weighing Mutation Scores to Improve Prioritization of Immunogenic Mutations
[0325]RNA that is injected into the cell, once translated and cleaved into short peptides, can be presented on different HLA types within the cell. Therefore it stands to reason that the more HLA types that are predicted to have a low MHC consensus (or similar) score, the more likely a given mutation will be immunogenic since it can potentially be displayed on more than one HLA type in parallel. Thus, weighing mutations by the number of HLA types for which the mutation is classified as HA and / or HB∪HC or even weighing each mutation simply by the number of HLA types that have a low Mmut score may improve immunogenicity ranking. In the most general solution, when we inject a 27mer RNA or peptide into the cell, there is not just the freedom to select the HLA type, but also the length of the peptide and the position of the mutation within this peptide. Therefore, one can scan all possible HLA types...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More