Methods of treatment with cyp3a4 substrate drugs
a technology of cyp3a4 and substrate drugs, which is applied in the direction of drug compositions, cardiovascular disorders, dispersed delivery, etc., can solve the problems that cyp3a4 substrate drugs cannot always be safely administered immediately after a patient has stopped posaconazole treatment, and can have serious and potentially life-threatening side effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
embodiments i
[0274]1. A method of treating a patient who has previously been administered a therapeutically effective regimen of posaconazole, with a CYP3A4 substrate drug contraindicated for concomitant administration with a strong CYP3A4 inhibitor, said method comprising:
[0275]first treating the patient, or prescribing a first treatment to begin, with the CYP3A4 substrate drug at least 2-42 days after stopping administration of posaconazole.
[0276]2. The method of embodiment 1, wherein said CYP3A4 substrate drug is selected from the group consisting of lurasidone, ranolazine, lumacaftor / ivacaftor, venetoclax, trabectedin, ribociclib succinate, deflazacort, cinacalcet hydrochloride, pimavanserin tartrate, aripiprazole lauroxil, cariprazine hydrochloride, simeprevir sodium, everolimus, saxagliptin hydrochloride, saxagliptin / metformin hydrochloride, ticagrelor, vilazodone hydrochloride, apixaban, tofacitinib citrate, eletriptan hydrobromide, nilotinib hydrochloride monohydrate, dronedarone hydroch...
example 1
inetic Studies with Posaconazole and Lurasidone
[0553]Inventors studied 6 obese male and female subjects (ages 18-50, BMI>35) taking Posaconazole oral tablets (300 mg qd) and Lurasidone (20 mg qd). Body weights and BMI measurements for the 6 subjects are provided below in Table 1.
TABLE 1Subject DemographicsSubject #Weight (kg)BMI (kg / m2)101-001111.845101-002136.844.4101-005137.751.2101-007103.736.8101-008122.339.8101-010120.043.9
[0554]Subjects were dosed with Lurasidone alone on Day 1, then subsequently dosed to steady-state Posaconazole levels, with a loading dose of 300 mg twice a day on Day 2 and 300 mg once a day thereafter over a period of 14 days. Posaconazole administration was then stopped and Lurasidone (20 mg qd) administered 2, 4, and 6 days after administration had ceased (studies days 17, 19, and 21 respectively). Lurasidone AUC was measured for 24 hours after each administration. Table 2 shows subject Lurasidone AUC levels 2, 4 and 6 days after Posaconazole was stopped,...
example 2
Impairment of Lurasidone Clearance after Discontinuation of Posaconazole, Impact of Obesity, and Implications for Patient Safety
[0560]The following studies were reported by Greenblatt et al., J. Clin. Psychopharmacol., 2018; 38(4):289-295 (doi: 10.1097 / JCP.0000000000000892), which is herein incorporated by reference in its entirety for all purposes.
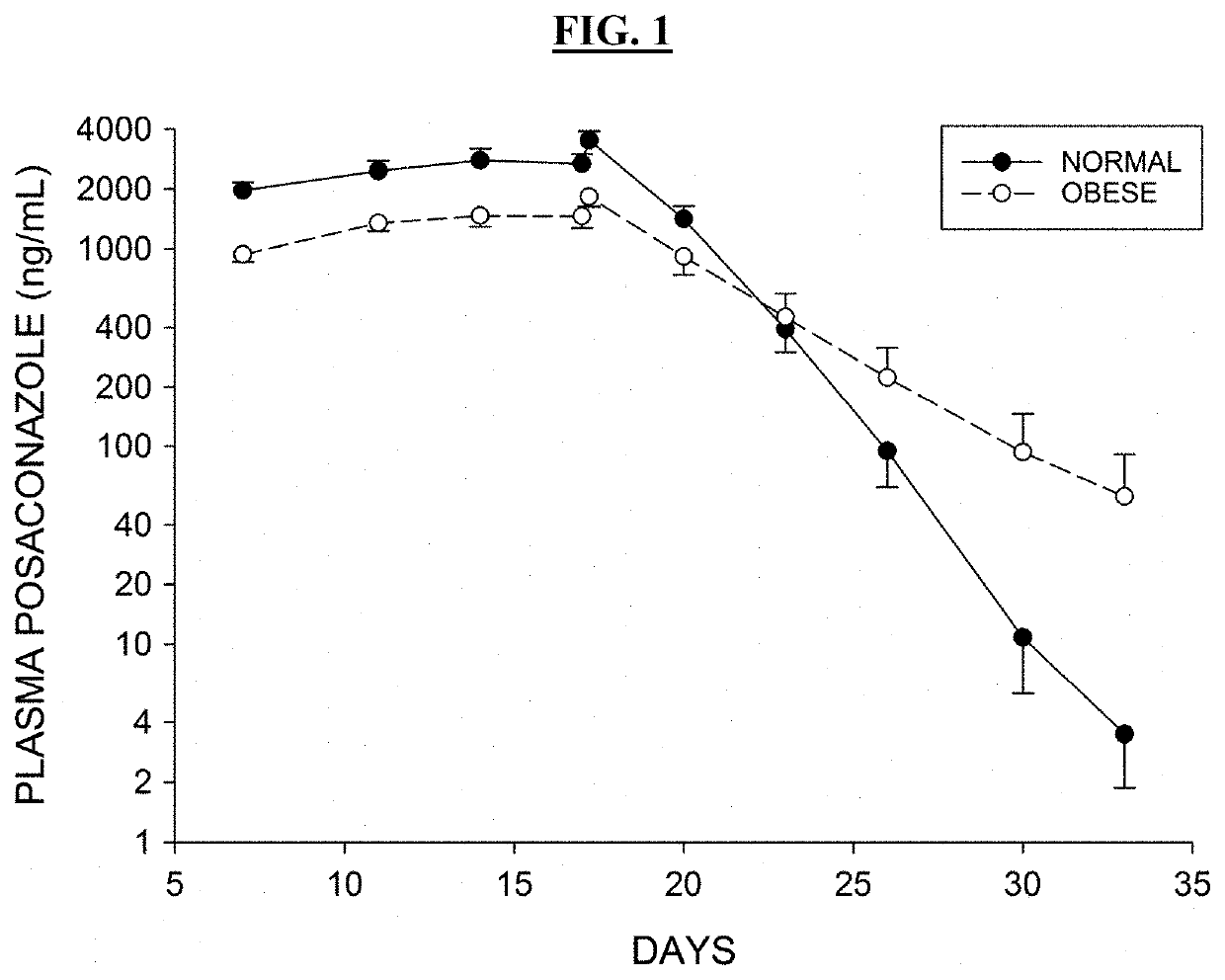
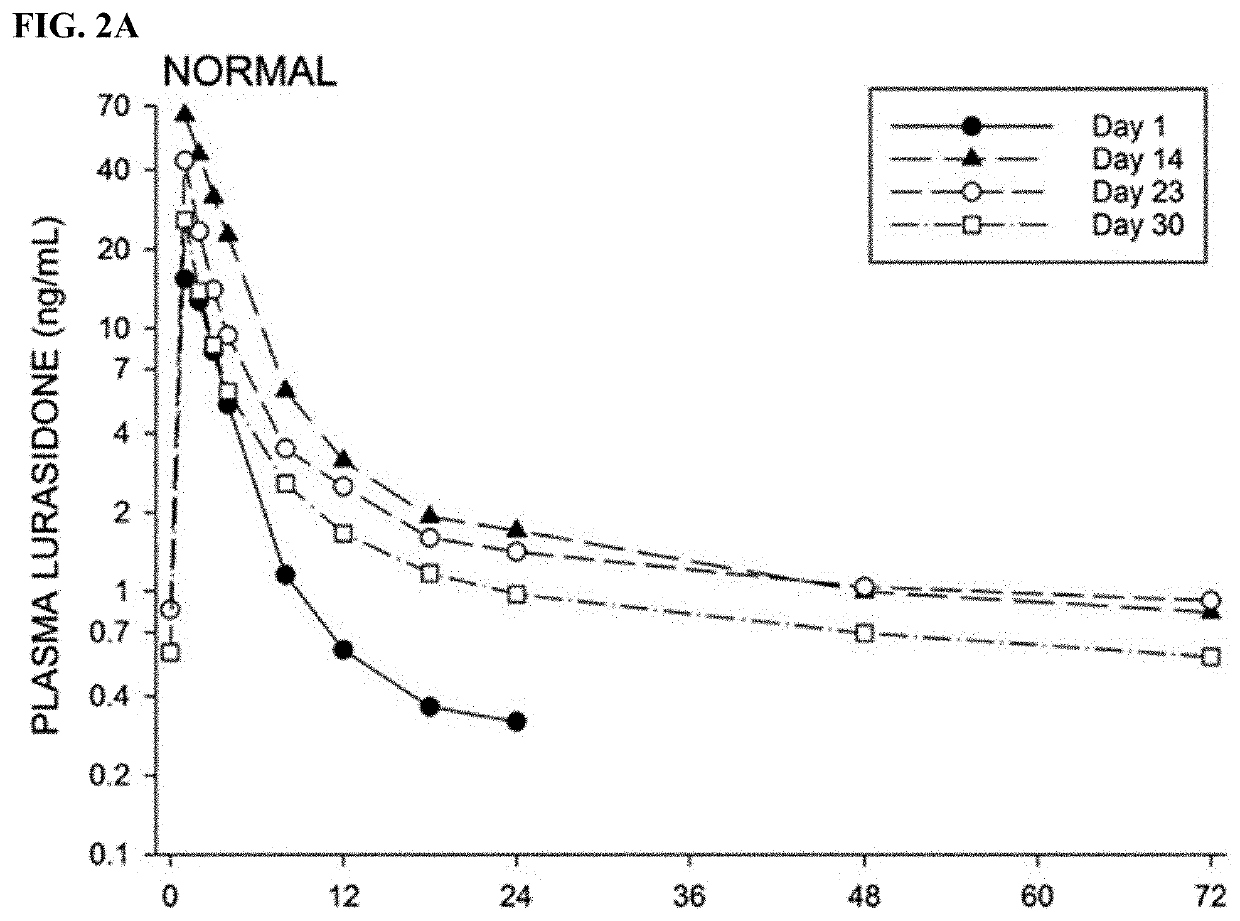
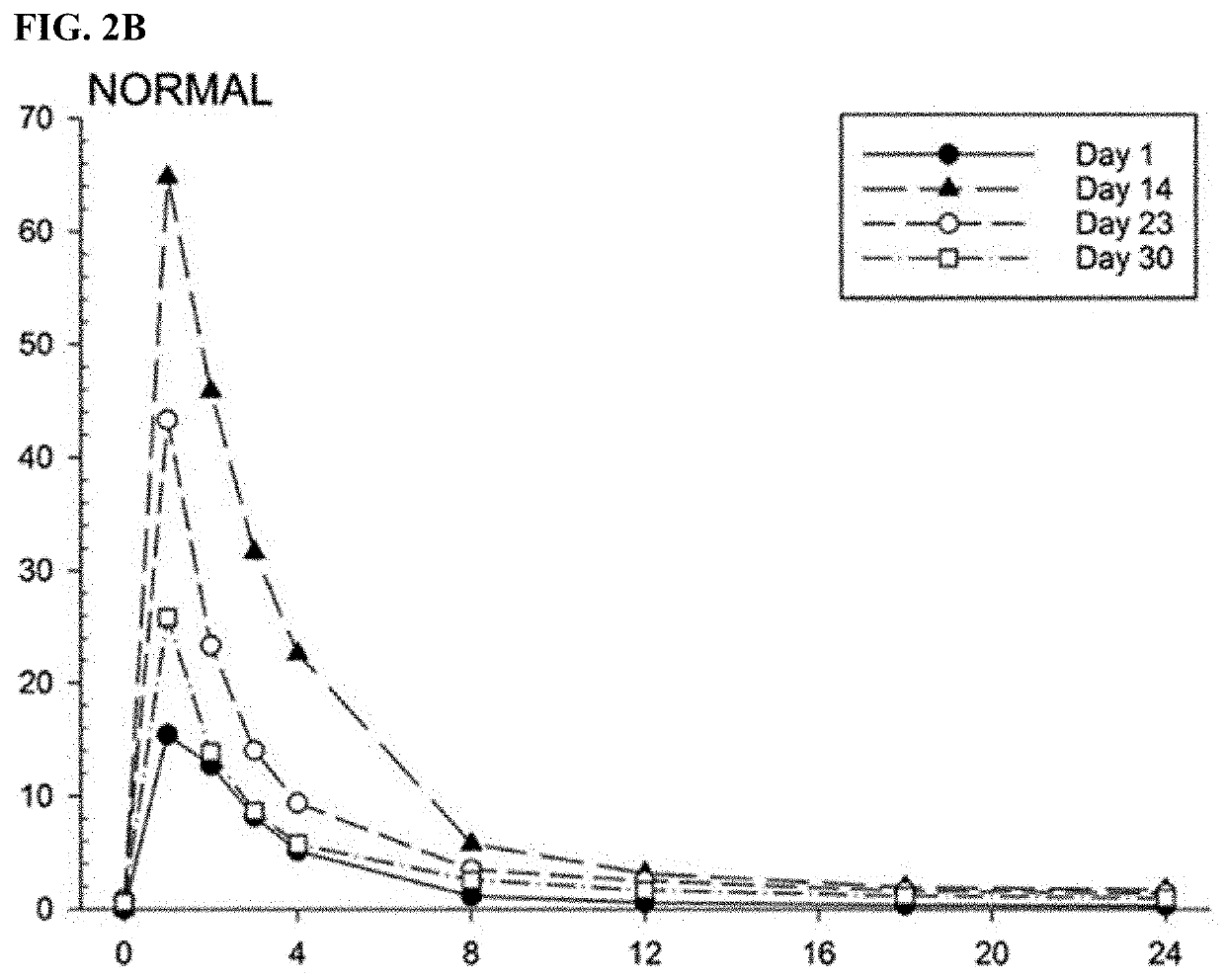
[0561]The antipsychotic agent lurasidone is metabolized by Cytochrome P450-3A (CYP3A) enzymes. Coadministration with strong CYP3A inhibitors (such as ketoconazole, posaconazole, and ritonavir) is contraindicated due to the risk of sedation and movement disorders from high levels of lurasidone. This study evaluated the time-course of recovery from the posaconazole drug interaction, and the effect of obesity on the recovery process.
[0562]With posaconazole coadministration, lurasidone area under the concentration curve (AUC) increased by an arithmetic mean factor of 6.2 in normals, and by 4.9 in obese subjects. Post-treatment washout of posa...
PUM
| Property | Measurement | Unit |
|---|---|---|
| time | aaaaa | aaaaa |
| time | aaaaa | aaaaa |
| size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More