Ebstine solid oral preparation and its preparing method
An oral preparation, the technology of ebastine, applied in the pharmaceutical field, can solve the problems of people who are not easy to obtain drinking water or tourists, inconvenience and the like
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
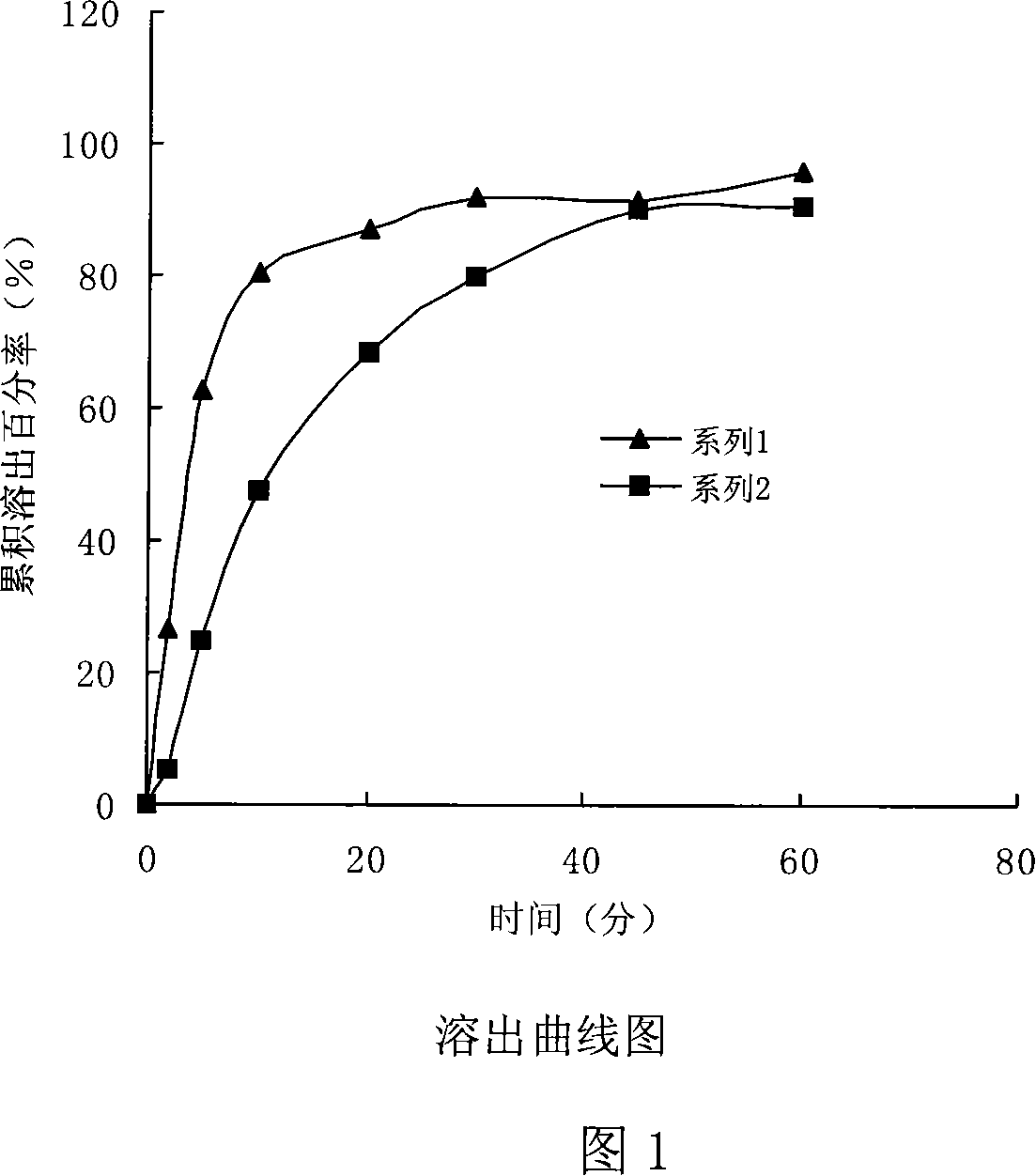
[0085] An ebastine orally disintegrating tablet is made of the following raw materials in parts by weight:
[0086] Example 1
Example 2
Example 3
Example 4
Example 5
parts by weight
parts by weight
parts by weight
parts by weight
parts by weight
95% ethanol solution
80% ethanol solution
Cross-linked CMC
Crospovidone
stevia
Micropowder silica gel
10
40
0
0
0
60
0
40
0
8
0
1
0
0
2
0
10
40
0
5
0
60
0
40
0
8
0
1
0
0
2
0
10
15
0
0
3
0
60
0
30
0
10
0
2
0.5
0
10
10
0
15
0
8 (4 servi...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More