Method for purifying anti-HER2 or/and anti-HER3 antibody proteins
A purification method and protein technology, applied in the field of biochemistry, to increase the virus filtration process, reduce production costs, and improve production efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0024] Below in conjunction with specific embodiment, further illustrate the present invention. These examples are only for illustrating the present invention and are not intended to limit the scope of the present invention. For the experimental methods without specific conditions indicated in the following examples, usually follow the conventional conditions or the conditions suggested by the manufacturer. Unless otherwise defined, all professional and scientific terms used herein have the same meanings as commonly understood by those skilled in the art. In addition, any methods and materials similar or equivalent to those described can be applied to the method of the present invention. The preferred implementation methods and materials described herein are for demonstration purposes only.
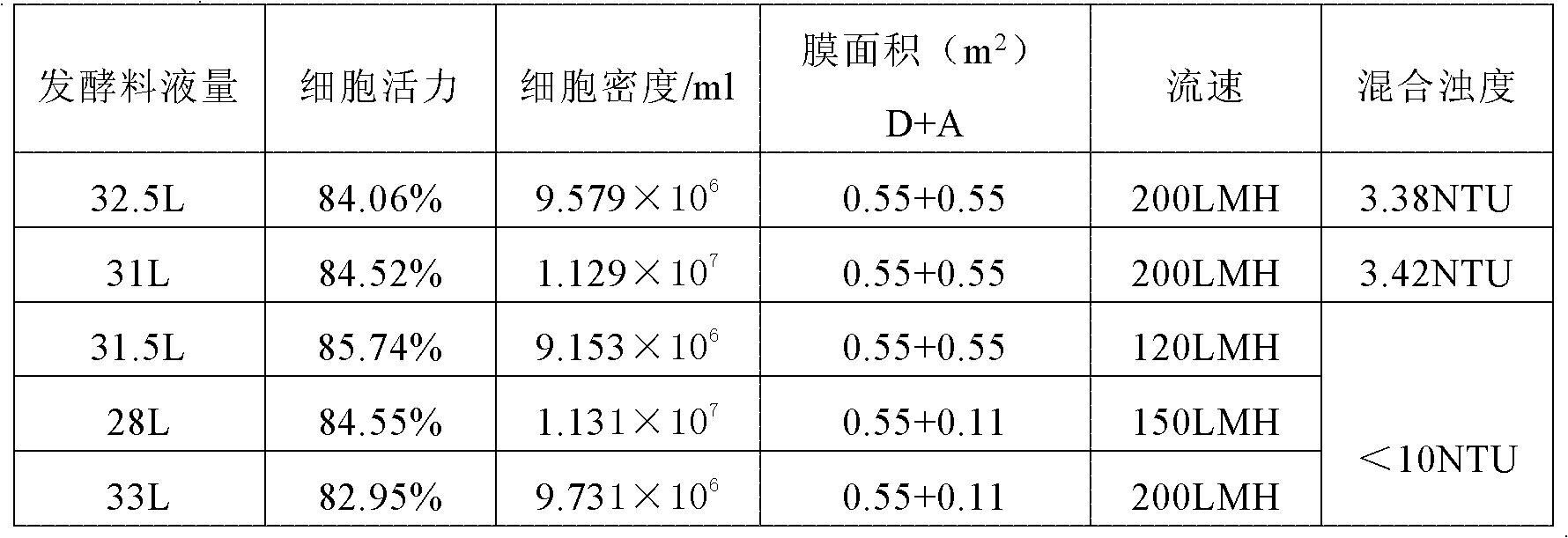
[0025] 1. Harvesting of Fermentation Supernatant
[0026] A two-stage depth filtering method is used.
[0027] Fermentation broth: the fermentation broth containing anti-HER2 monoclon...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More